Abstract
Objective:
As the peroxisome proliferator-activated receptor γ (PPARγ) plays a central role in fat mass regulation, we investigated whether initial subcutaneous PPARγ activity is related to fat mass generation during overfeeding.
Subjects:
Fourteen healthy female subjects (age 25±4 years, BMI 22.1±2.3 kg/m2).
Design and measurements:
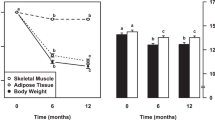
Subjects were overfed with a diet supplying 50% more energy than baseline energy requirements for 14 days. Fasting blood samples were analyzed for leptin, insulin and glucose. Fasting subcutaneous abdominal fat biopsies were obtained for analysis of PPARγ1, PPARγ2, aP2 and UCP2 mRNAs.
Results:
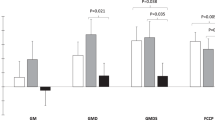
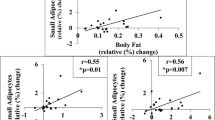
Initial PPARγ1 and 2, aP2 and UCP2 mRNAs were not related to fat gain (P>0.12). However, PPARγ1, PPARγ2 and aP2 mRNA changes were positively related to changes in plasma leptin (P<0.05) and, except aP2 (P=0.06), to fat gain (P<0.05). PPARγ and aP2 mRNA changes were positively related (P<0.01), indicating that PPARγ mRNA levels reflected PPARγ activity.
Conclusion:
These data suggest that the ability to increase PPARγ activity might be involved in the susceptibility to gain weight during a positive energy balance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Bouchard C, Tremblay A, Despres JP, Nadeau A, Lupien PJ, Theriault G et al. The response to long-term overfeeding in identical twins. N Eng J Med 1990; 322: 1477–1482.
Rosen ED, Sarraf P, Troy AE, Bradwin G, Moore K, Milstone DS et al. PPARγ is required for the differentiation of adipose tissue in vivo and in vitro. Mol Cell 1999; 4: 611–617.
Vidal-Puig AJ, Considine RV, Jimenez-Linan M, Werman A, Pories WJ, Caro JF et al. Peroxisome proliferator-activated receptor gene expression in human tissues. J Clin Invest 1997; 99: 2416–2422.
Rosen ED, Walkey CJ, Puigserver P, Spiegelman BM . Transcriptional regulation of adipogenesis. Genes Dev 2000; 14: 1293–1307.
Mueller E, Drori S, Aiyer A, Yie J, Sarraf P, Chen H et al. Genetic analysis of adipogenesis through peroxisome proliferator-activated receptor γ isoforms. J Biol Chem 2002; 277: 41925–41930.
Kliewer SA, Sundseth SS, Jones SA, Brown PJ, Wisely GB, Koble CS et al. Fatty acids and eicosanoids regulate gene expression through direct interactions with peroxisome proliferator-activated receptors α and γ. Proc Natl Acad Sci USA 1997; 94: 4318–4323.
Ross SR, Graves RA, Greenstein A, Platt KA, Shyu HL, Mellovitz B et al. A fat-specific enhancer is the primary determinant of gene expression for adipocyte P2 in vivo. Proc Natl Acad Sci USA 1990; 87: 9590–9594.
Sen A, Lea-Currie YR, Sujkowska D, Franklin DM, Wilkison WO, Halvorsen YD et al. Adipogenic potential of human adipose derived stromal cells from multiple donors is heterogeneous. J Cell Biochem 2001; 81: 312–319.
Kelly LJ, Vicario PP, Thompson GM, Candelore MR, Doebber TW, Ventre J et al. Peroxisome proliferator-activated receptors γ and α mediate in vivo regulation of uncoupling protein (UCP-1, UCP-2, UCP-3) gene expression. Endocrinology 1998; 139: 4920–4927.
Digby JE, Crowley VE, Sewter CP, Whitehead JP, Prins JB, O'Rahilly S . Depot-related and thiazolidinedione-responsive expression of uncoupling protein 2 (UCP2) in human adipocytes. Int J Obes Relat Metab Disord 2000; 24: 585–592.
Medvedev AV, Snedden SK, Raimbault S, Ricquier D, Collins S . Transcriptional regulation of the mouse uncoupling protein-2 gene. Double E-box motif is required for peroxisome proliferator-activated receptor-γ-dependent activation. J Biol Chem 2001; 276: 10817–10823.
Esterbauer H, Schneitler C, Oberkofler H, Ebenbichler C, Paulweber B, Sandhofer F et al. A common polymorphism in the promoter of UCP2 is associated with decreased risk of obesity in middle-aged humans. Nat Genet 2001; 28: 178–183.
Westerterp KR, Wouters L, Marken Lichtenbelt WDV . The Maastricht protocol for the measurement of body composition and energy expenditure with labeled water. Obes Res 1995; 3: 49–57.
Siri WE . Body composition from fluid spaces and density: analysis of methods. National Academy of Science and National Research Council: Washington, DC, 1961, pp 223–244.
Chomczynski P, Sacchi N . Single-step method of RNA isolation by acid guanidinium thiocyanate–phenol–chloroform extraction. Anal Biochem 1987; 162: 156–159.
Deeb SS, Fajas L, Nemoto M, Pihlajamaki J, Mykkanen L, Kuusisto J et al. A Pro12Ala substitution in PPARgamma2 associated with decreased receptor activity, lower body mass index and improved insulin sensitivity. Nat Genet 1998; 20: 284–287.
Masugi J, Tamori Y, Mori H, Koike T, Kasuga M . Inhibitory effect of a proline-to-alanine substitution at codon 12 of peroxisome proliferator-activated receptor-gamma 2 on thiazolidinedione-induced adipogenesis. Biochem Biophys Res Commun 2000; 268: 178–182.
Kolehmainen M, Uusitupa MI, Alhava E, Laakso M, Vidal H . Effect of the Pro12Ala polymorphism in the peroxisome proliferator-activated receptor (PPAR) γ2 gene on the expression of PPARγ target genes in adipose tissue of massively obese subjects. J Clin Endocrinol Metab 2003; 88: 1717–1722.
Yamauchi T, Kamon J, Waki H, Murakami K, Motojima K, Komeda K et al. The mechanisms by which both heterozygous peroxisome proliferator-activated receptor γ (PPARγ) deficiency and PPARγ agonist improve insulin resistance. J Biol Chem 2001; 276: 41245–41254.
Rieusset J, Andreelli F, Auboeuf D, Roques M, Vallier P, Riou JP et al. Insulin acutely regulates the expression of the peroxisome proliferator-activated receptor-γ in human adipocytes. Diabetes 1999; 48: 699–705.
Walder K, Norman RA, Hanson RL, Schrauwen P, Neverova M, Jenkinson CP et al. Association between uncoupling protein polymorphisms (UCP2-UCP3) and energy metabolism/obesity in Pima indians. Hum Mol Genet 1998; 7: 1431–1435.
Nisoli E, Carruba MO, Tonello C, Macor C, Federspil G, Vettor R . Induction of fatty acid translocase/CD36, peroxisome proliferator-activated receptor-γ2, leptin, uncoupling proteins 2 and 3, and tumor necrosis factor-alpha gene expression in human subcutaneous fat by lipid infusion. Diabetes 2000; 49: 319–324.
Acknowledgements
We thank Erwin Thimister and Chris Hukshorn for obtaining blood and fat samples and Jolanda van der Meijde and Esther Moonen-Kornips for mRNA measurements.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Joosen, A., Bakker, A., Zorenc, A. et al. PPARγ activity in subcutaneous abdominal fat tissue and fat mass gain during short-term overfeeding. Int J Obes 30, 302–307 (2006). https://doi.org/10.1038/sj.ijo.0803146
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0803146
Keywords
This article is cited by
-
The interplay of mitophagy, autophagy, and apoptosis in cisplatin-induced kidney injury: involvement of ERK signaling pathway
Cell Death Discovery (2024)
-
Substrate utilization and metabolic profile in response to overfeeding with a high-fat diet in South Asian and white men: a sedentary lifestyle study
International Journal of Obesity (2020)
-
Liver fat accumulation in response to overfeeding with a high-fat diet: a comparison between South Asian and Caucasian men
Nutrition & Metabolism (2015)
-
Moderate alcohol consumption increases insulin sensitivity and ADIPOQ expression in postmenopausal women: a randomised, crossover trial
Diabetologia (2008)