Abstract
OBJECTIVE: To study the effect of simple vs complex carbohydrates (SCHO and CCHO respectively) containing breakfasts on blood parameters, hunger and satiety and mood.
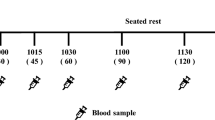
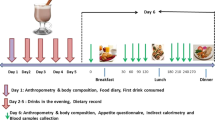
DESIGN: A 2-day, open, randomised, cross-over trial.
SUBJECTS: A total of 26 male subjects (34±6 y; BMI 23.4±2.2 kg m−2).
MEASUREMENTS: Blood glucose, insulin, triacylglycerols (TG), free fatty acids (FFA) and cholecystokinin (CCK) were determined repeatedly for 4 h on both test days after a breakfast containing SCHO or CCHO. Feelings of hunger and satiety were determined at similar time points as well. Mood state was examined 3 h after breakfast consumption.
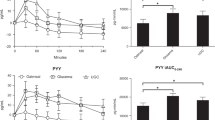
RESULTS: Consumption of a SCHO breakfast resulted in higher glucose and insulin levels at 30 min after breakfast consumption. TG at 180 min, and FFA at 180 and 240 min were higher after SCHO breakfast than after CCHO breakfast. Satiety scores were higher after CCHO breakfast consumption for the first 90 min after intake. Furthermore, the item ‘fatigue’ was scored higher after SCHO breakfast than after CCHO breakfast intake.
CONCLUSION: Consumption of a CCHO breakfast is favourable in comparison to a SCHO breakfast, because of the lower perception of ‘fatigue’ and the higher degree of satiety after consumption.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
World Health Organisation. Obesity: preventing and managing the global epidemic. WHO: Geneva; 1998.
Sørensen TIA . The changing lifestyle in the world. Body weight and what else. Diabetes Care 2000; 23 (Suppl 2): B1–B4.
Seidell JC . Voeding en gezondheid in de volgende eeuw: problemen van grote omvang. Vrije Universiteit Press: Amsterdam; 1999.
Godsland IF, Stevenson JC . Insulin resistance: syndrome or tendency? Lancet 1995; 346: 100–103.
Meigs JB . Invited commentary: insulin resistance syndrome? Syndrome X? Multiple metabolic syndrome? A syndrome at all? Factor analysis reveals patterns in the fabric of correlated metabolic risk factors. Am J Epidemiol 2000; 152: 908–911.
Riccardi G, Rivellese AA . Dietary treatment of the metabolic syndrome—the optimal diet. Br J Nutr 2000; 83(Suppl 1): S143–S148.
Ludwig DS . Dietary glycemic index and obesity. J Nutr 2000; 130: 280S–283S.
Kirk T, Crombie N, Cursiter M . Promotion of dietary carbohydrate as an approach to weight maintenance after initial weight loss: a pilot study. J Hum Nutr Diet 2000; 13: 277–285.
Connor WE, Connor SL, Katan MB, Grundy SM, Willett WC . Should a low-fat, high-carbohydrate diet be recommended for everyone? N Eng J Med 1997; 337: 562–567.
Seidell JC . Optimizing fat intake: does a reduction in fat intake prevent obesity? Eur Heart J Suppl 1999; 1 (Suppl S): S118–S122.
Jenkins DJA, Wolever TMS, Taylor RH, Barker H, Fielden H, Baldwin JM, Bowling AC, Newman HC, Jenkins AL, Goff DV . Glycemic index of foods: a physiological basis for carbohydrate exchange. Am J Clin Nutr 1981; 34: 362–366.
Wolever TMS, Jenkins DJA, Jenkins AL, Josse RG . The glycemic index: methodology and clinical implications. Am J Clin Nutr 1991; 54: 846–854.
Dutch food consumption survey. Zo eet Nederland 1998. Voedingscentrum: Den Haag; 1998.
Markus CR, Klöpping-Ketelaars WI, Pasman WJ, Klarenbeek B, van den Berg H . Dose-dependent effect of α-lactalbumin in combination with two different doses of glucose on the plasma Trp/LNAA ratio. Nutr Neurosci 2000; 3: 345–355.
Jansen JBMJ, Lamers CBHW . Radioimmunoassay of cholecystokinin in human tissue and plasma. Clin Chim Acta 1983; 131: 305–316.
Wald FDM, Mellenbergh GJ . Instrumenteel onderzoek. De verkorte versie van de Nederlandse vertaling van de Profile of Mood States (POMS). Ned Tijdschr voor Psychol 1990; 45: 86–90.
Smith A, Bazzoni C, Beale J, Elliott-Smith J, Tiley M . High fibre breakfast cereals reduce fatigue. Appetite 2001; 37: 249–250.
Roberts SB . High-glycemic index foods, hunger, and obesity: is there a connection? Nutr Rev 2000; 58: 163–169.
Sparti A, Milon H, Di Vetta V, Schneiter P, Tappy L, Jéquier E, Schutz Y . Effects of diets high or low in unavailable and slowly digestible carbohydrates on the pattern of 24-h substrate oxidation and feelings of hunger in humans. Am J Clin Nutr 2000; 72: 1461–1468.
Ludwig DS, Majzoub JA, Al-Zahrani A, Dallal GE, Blanco I, Roberts SB . High glycemic index foods, overeating, and obesity. Pediatrics 1999; 103: E26.
Saris WHM, Astrup A, Prentice AM, Zunft HJF, Formiguera X, Verboeket-van de Venne WPHG, Raben A, Poppitt SD, Seppelt B, Johnston S, Vasilaras TH, Keogh GF . Randomized controlled trial of changes in dietary carbohydrate/fat ratio and simple vs complex carbohydrates on body weight and blood lipids: the CARMEN study. Int J Obes Relat Metab Disord 2000; 24: 1310–1318.
Pasman WJ, Wauters MAJ, Westerterp-Plantenga MS, Saris WHM . Effect of one week of fibre supplementation on hunger and satiety or energy intake. Appetite 1997; 29: 77–87.
Poppitt SD, Keogh GF, Prentice AM, Williams DEM, Sonnemans HMW, Valk EEJ, Robinson E, Wareham NJ . Long-term effects of ad libitum low-fat, high-carbohydrate diets on body weight and serum lipids in overweight subjects with metabolic syndrome. Am J Clin Nutr 2002; 75: 11–20.
Pi-Sunyer FX . Glycemic index. Obes Res 2000; 8(Suppl 1); 1S, I8.
Ludwig DS . The glycemic index. Physiological mechanisms relating to obesity, diabetes, and cardiovascular disease. JAMA 2002; 287: 2414–2423.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pasman, W., Blokdijk, V., Bertina, F. et al. Effect of two breakfasts, different in carbohydrate composition, on hunger and satiety and mood in healthy men. Int J Obes 27, 663–668 (2003). https://doi.org/10.1038/sj.ijo.0802284
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0802284
Keywords
This article is cited by
-
The relation between dietary intakes and psychological disorders in Iranian adults: a population-based study
BMC Psychiatry (2020)
-
Effects of cereal breakfasts on postprandial glucose, appetite regulation and voluntary energy intake at a subsequent standardized lunch; focusing on rye products
Nutrition Journal (2011)
-
Endosperm and whole grain rye breads are characterized by low post-prandial insulin response and a beneficial blood glucose profile
Nutrition Journal (2009)
-
Food intake and plasma ghrelin response during potato-, rice- and pasta-rich test meals
European Journal of Nutrition (2007)