Abstract
OBJECTIVE: Obesity has increased at an alarming rate in recent years and is now a worldwide health problem. We investigated the effects of long-term feeding with tea catechins, which are naturally occurring polyphenolic compounds widely consumed in Asian countries, on the development of obesity in C57BL/6J mice.
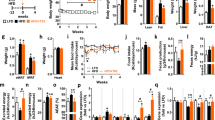
DESIGN: We measured body weight, adipose tissue mass and liver fat content in mice fed diets containing either low-fat (5% triglyceride (TG)), high-fat (30% TG), or high-fat supplemented with 0.1–0.5% (w/w) tea catechins for 11 months. The β-oxidation activities and related mRNA levels were measured after 1 month of feeding.
RESULTS: Supplementation with tea catechins resulted in a significant reduction of high-fat diet-induced body weight gain, visceral and liver fat accumulation, and the development of hyperinsulinemia and hyperleptinemia. Feeding with tea catechins for 1 month significantly increased acyl-CoA oxidase and medium chain acyl-CoA dehydrogenase mRNA expression as well as β-oxidation activity in the liver.
CONCLUSION: The stimulation of hepatic lipid metabolism might be a factor responsible for the anti-obesity effects of tea catechins. The present results suggest that long-term consumption of tea catechins is beneficial for the suppression of diet-induced obesity, and it may reduce the risk of associated diseases including diabetes and coronary heart disease.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Larsson B, Bjorntorp P, Tibblin G . The health consequences of moderate obesity Int J Obes 1981 5: 97–116.
Hartz AJ, Rupley DC Jr, Kalkhoff RD, Rimm AA . Relationship of obesity to diabetes: influence of obesity level and body fat distribution Prev Med 1983 12: 351–357.
Shiota S, Shimizu M, Mizushima T, Ito H, Hatano T, Yoshida T, Tsuchiya T . Marked reduction in the minimum inhibitory concentration (MIC) of beta-lactams in methicillin-resistant Staphylococcus aureus produced by epicatechin gallate, an ingredient of green tea (Camellia sinensis) Biol Pharm Bull 1999 22: 1388–1390.
Yang CS, Wang ZY . Tea and cancer J Natl Cancer Inst 1993 85: 1038–1049.
Muramatsu K, Fukuyo M, Hara Y . Effect of green tea catechins on plasma cholesterol level in cholesterol-fed rats J Nutr Sci Vitaminol 1986 32: 613–622.
Matsumoto N, Ishigaki F, Ishigaki A, Iwashima H, Hara Y . Reduction of blood glucose levels by tea catechin Biosci Biotech Biochem 1993 57: 525–527.
Mukhtar H, Ahmad N . Tea polyphenols: prevention of cancer and optimizing health Am J Clin Nutr 2000 71 (6 Suppl): 1698S–1702S.
Kao YH, Hiipakka RA, Liao S . Modulation of endocrine systems and food intake by green. tea epigallocatechin gallate Endocrinology 2000 141: 980–987.
Kao YH, Hiipakka RA, Liao S . Modulation of obesity by a green tea catechin Am J Clin Nutr 2000 72: 1232–1234.
Dulloo AG, Duret C, Rohrer D, Girardier L, Mensi N, Fathi M, Chantre P, Vandermander J . Efficacy of a green tea extract rich in catechin polyphenols and caffeine in increasing 24-h energy expenditure and fat oxidation in humans Am J Clin Nutr 1999 70: 1040–1045.
Dulloo AG, Seydoux J, Girardier L, Chantre P, Vandermander J . Green tea and thermogenesis: interactions between catechin-polyphenols, caffeine and sympathetic activity Int J Obes Relat Metab Disord 2000 24: 252–258.
Surwit RS, Kuhn CM, Cochrane C, McCubbin JA, Feinglos MN . Diet-induced type II diabetes in C57BL/6J mice Diabetes 1988 37: 1163–1167.
Goto T, Yoshida Y, Kiso M, Nagashima H . Simultaneous analysis of individual catechins and caffeine in green tea J Chromatogr 1996 749: 295–299.
Folch J, Leens M, Sloane-Stanley GH . A simple method for the isolation and purification of total lipids from animal tissue J Biol Chem 1957 226: 497–509.
Murase T, Mizuno T, Omachi T, Onizawa K, Komine Y, Kondo H, Hase T, Tokimitsu I . Dietary diacylglycerol suppresses high-fat and high sucrose diet-induced body fat accumulation in C57BL/6J mice J Lipid Res 2001 42: 372–378.
Singh H, Beckman K, Poulos A . Peroxisomal β-oxidation of branched chain fatty acids in rat liver. Evidence that carnitine palmitoyltransferase I prevents transport of branched chain fatty acids into mitochondria J Biol Chem 1994 269: 9514–9520.
Nakagawa K, Miyazawa T . Absorption and distribution of tea catechin, (−)-epigallocatechin-3-gallate, in the rat J Nutr Sci Vitaminol 1997 43: 679–684.
Suganuma M, Okabe S, Oniyama M, Tada Y, Ito H, Fujiki H . Wide distribution of [3H](−)-epigallocatechin gallate, a cancer preventive tea polyphenol, in mouse tissue Carcinogenesis 1998 19: 1771–1776.
Nemali MR, Usuda N, Reddy MK, Oyasu K, Hashimoto T, Osumi T, Rao MS, Reddy JK . Comparison of constitutive and inducible levels of expression of peroxisomal beta-oxidation and catalase genes in liver and extrahepatic tissues of rat Cancer Res 1988 48: 5316–5324.
Kelly DP, Gordon JI, Alpers R, Strauss AW . The tissue-specific expression and developmental regulation of two nuclear genes encoding rat mitochondrial proteins J Biol Chem 1989 264: 18921–18925.
Nagao T, Meguro S, Soga S, Otsuka A, Tomonobu K, Fumoto S, Chikama A, Mori K, Yuzawa M, Watanabe H, Hase T, Tanaka Y, Tokimitsu I, Shimasaki H, Itakura H . Tea catechins suppress accumulation of body fat in humans J Oleo Sci 2001 50: 717–728.
Scalbert A, Williamson G . Dietary intake and bioavailability of polyphenols J Nutr 2000 130: 2073S–2085S.
Schoonjans K, Staels B, Auwerx J . Role of the peroxisome proliferator-activated receptor (PPAR) in mediating the effects of fibrates and fatty acids on gene expression J Lipid Res 1996 37: 907–925.
Delerive P, De Bosscher K, Besnard S, Vanden Berghe W, Peters JM, Gonzalez FJ, Fruchart JC, Tedgui A, Haegeman G, Staels B . Peroxisome proliferator-activated receptor α negatively regulates the vascular inflammatory gene response by negative cross-talk with transcription factors NF-κB and AP-1 J Biol Chem 1999 274: 32048–32054.
Lin YL, Lin JK . (−)-Epigallocatechin-3-gallate blocks the induction of nitric oxide synthase by down-regulating lipopolysaccharide-induced activity of transcription factor nuclear factor-κB Mol Pharmac 1997 52: 465–472.
Murase T, Kume N, Hase T, Shibuya Y, Nishizawa Y, Tokimitsu I, Kita T . Gallates inhibit cytokine-induced nuclear translocation of NE-κB and expression of leukocyte adhesion molecules in vascular endothelial cells Arterioscler Thromb Vasc Biol 1999 19: 1412–1420.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Murase, T., Nagasawa, A., Suzuki, J. et al. Beneficial effects of tea catechins on diet-induced obesity: stimulation of lipid catabolism in the liver. Int J Obes 26, 1459–1464 (2002). https://doi.org/10.1038/sj.ijo.0802141
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0802141
Keywords
This article is cited by
-
The interaction effect of green tea consumption and exercise training on fat oxidation, body composition and blood lipids in humans: a review of the literature
Sport Sciences for Health (2023)
-
Verification of the folkloric and anecdotal antidiabetic effects of Hypoxis hemerocallidea (Fisch., C.A. Mey. & Avé-Lall) and isolated, β-sitosterol using early-stage type II spontaneous diabetic mutant BKS-Leprdb mice
BMC Complementary Medicine and Therapies (2022)
-
Flavonoids are promising safe therapy against COVID-19
Phytochemistry Reviews (2022)
-
Effect of Yerba Mate (Ilex paraguariensis) on Lipid Levels: A Systematic Review and Meta-Analysis
Plant Foods for Human Nutrition (2022)
-
Dose-dependent Effects of Cacao Polyphenol Intake on Lipid Metabolism in Rats
Plant Foods for Human Nutrition (2021)