Abstract
Objective: To determine reference values for fat-free mass index (FFMI) and fat mass index (FMI) in a large Caucasian group of apparently healthy subjects, as a function of age and gender and to develop percentile distribution for these two parameters.
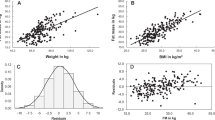
Design: Cross-sectional study in which bioelectrical impedance analysis (50 kHz) was measured (using tetrapolar electrodes and cross-validated formulae by dual-energy X-ray absorptiometry in order to calculate FFMI (fat-free mass/height squared) and FMI (fat mass/height squared).
Subjects: A total of 5635 apparently healthy adults from a mixed non-randomly selected Caucasian population in Switzerland (2986 men and 2649 women), varying in age from 24 to 98 y.
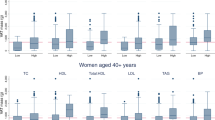
Results: The median FFMI (18–34 y) were 18.9 kg/m2 in young males and 15.4 kg/m2 in young females. No difference with age in males and a modest increase in females were observed. The median FMI was 4.0 kg/m2 in males and 5.5 kg/m2 in females. From young to elderly age categories, FMI progressively rose by an average of 55% in males and 62% in females, compared to an increase in body mass index (BMI) of 9 and 19% respectively.
Conclusions: Reference intervals for FFMI and FMI could be of practical value for the clinical evaluation of a deficit in fat-free mass with or without excess fat mass (sarcopenic obesity) for a given age category, complementing the classical concept of body mass index (BMI) in a more qualitative manner. In contrast to BMI, similar reference ranges seems to be utilizable for FFMI with advancing age, in particular in men.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Kolata G . Obesity declared a disease Science 1985 227: 1019–1020.
James WPT, Ralph A . New understanding in obesity research Proc Nutr Soc 1999 58: 385–393.
Garn SM, Leonard WR, Hawthorne VM . Three limitations of the body mass index Am J Clin Nutr 1986 44: 996–997.
McLaren DS . Three limitations of the body mass index. (Letter.) Am J Clin Nutr 1987 46: 121.
Micozzi MS, Albanes D . Three limitations of the body mass index. (Letter.) Am J Clin Nutr 1987 46: 376–377.
Garrow JS . Three limitations of the body mass index. (letter.) Am J Clin Nutr 1988 47: 553.
Seidell JC, Verschuren WM, van Leer EM, Kromhout D . Overweight, underweight, and mortality. A prospective study of 48,287 men and women Arch Intern Med 1996 156: 958–963.
Kuczmarski RJ, Carroll MD, Flegal KM, Troiano RP . Varying body mass index cutoff points to describe overweight prevalence among U.S. adults: NHANES III (1988–1994) Obes Res 1997 5: 542–548.
Seidell JC, Flegal KM . Assessing obesity: classification and epidemiology Br Med Bull 1997 53: 238–252.
Flegal KM, Carroll MD, Kuczmarski RJ, Johnson CL . Overweight and obesity in the United States: prevalence and trends, 1960–1994 Int J Obes Relat Metab Disord 1998 22: 39–47.
Seidell JC, Visscher TL, Hoogeveen RT . Overweight and obesity in the mortality rate data: current evidence and research issues Med Sci Sports Exerc 1999 31 (Suppl): S597–S601.
Ferraro KF, Both TL . Age, body mass index, and functional illness J Gerontol B Psychol Sci Soc 1999 54: S339–S348.
Kuczmarski RJ, Flegal KM . Criteria for definition of overweight in transition: background and recommendations for the United States Am J Clin Nutr 2000 72: 1074–1081.
Cole TJ, Bellizzi MC, Flegal KM, Dietz WH . Establishing a standard definition for child overweight and obesity worldwide: international survey Br Med J 2000 320: 1240–1243.
Visscher TL, Seidell JC, Menotti A, Blackburn H, Nissinen A, Feskens EJ, Kromhout D . Underweight and overweight in relation to mortality among men aged 40–59 and 50–69 y: the Seven Countries Study Am J Epidemiol 2000 151: 660–666.
Flegal KM, Troiano RP . Changes in the distribution of body mass index of adults and children in the US population Int J Obes Relat Metab Disord 2000 24: 807–818.
Fine JT, Colditz GA, Coakley EH, Moseley G, Manson JE, Willett WC, Kawachi I . A prospective study of weight change and health-related quality of life in women JAMA 1999 282: 2136–2142.
Deurenberg P . Universal cut-off BMI points for obesity are not appropriate Br J Nutr 2001 85: 135–136.
Ferro-Luzzi A, Sette S, Franklin M, James WP . A simplified approach of assessing adult chronic energy deficiency Eur J Clin Nutr 1991 46: 173–186.
Heber D, Ingles S, Ashley JM, Maxwell MH, Lyons RF, Elashoff RM . Clinical detection of sarcopenic obesity by bioelectrical impedance analysis Am J Clin Nutr 1996 64: 472S–477S.
Van Itallie TB, Yang M-U, Heymsfield SB, Funk RC, Boileau R . Height-normalized indices of the body's fat-free mass and fat mass: potentially useful indicators of nutritional status Am J Clin Nutr 1990 52: 953–459.
Deurenberg P, Schutz Y . Body composition: overview of methods and future directions of research Ann Nutr Metab 1995 39: 325–333.
Deurenberg P, Weststrate JA, van der Kooy K . Body composition changes assessed by bioelectrical impedance measurements Am J Clin Nutr 1989 49: 401.
Gray DS . Changes in biolectrical impedance during fasting Am J Clin Nutr 1988 48: 1184–1187.
Kushner RF, Schoeller DA . Estimation of total body water by biolectrical impedance analysis Am J Clin Nutr 1986 44: 417–424.
Lukaski HC . Assessment of fat-free mass using bioelectrical impedance measurements of the human body Am J Clin Nutr 1985 41: 810–817.
Jackson AS, Pollock ML, Graces JE, Mahar MT . Reliability and validity of bioelectrical impedance in determining body composition J Appl Physiol 1988 64: 529–534.
Segal KR . Use of bioelectrical impedance analysis measurements as an evaluation for participating in sports Am J Clin Nutr 1996 64: 469S–471S.
Lukaski HC . Validation of tetrapolar bioelectrical impedance measurements to assess human body composition J Appl Physiol 1986 60: 1327–1332.
Houtkooper LB, Lohman TG, Going SB, Howell WH . Why bioelectrical impedance analysis should be used for estimating adiposity Am J Clin Nutr 1996 64: 436S–448S.
Earthman CP, Matthie JR, Reid PM, Harper IT, Ravussin E, Howell WH . A comparison of bioimpedance methods for detection of body cell mass change in HIV infection J Appl Physiol 2000 88: 944–956.
Kyle UG, Genton L, Karsegard L, Slosman DO, Pichard C . Single prediction equation for bioelectrical impedance analysis in adults aged 20–94 y Nutrition 2001 17: 248–253.
Micozzi MS, Albanes D, Jones DY, Chumlea WC . Correlations of body mass indices with weight, stature, and body composition in men and women in NHANES I and II Am J Clin Nutr 1986 44: 725–731.
Frisancho AR . New standards of weight and body composition by frame size and height for assessment of nutritional status of adults and the elderly Am J Clin Nutr 1984 40: 808–819.
MacDonald SM, Reeder BA, Chen Y, Despres JP . Obesity in Canada: a descriptive analysis. Canadian Heart Health surveys Research Group Can Med Assoc J 1997 157: S3–S9.
Bartlett HL, Puhl SM, Hodgson JL, Buskirk ER . Fat-free mass in relation to stature: ratios of fat-free mass to height in children, adults, and elderly subjects Am J Clin Nutr 1991 53: 1112–1116.
Baarends EM, Schols AMWS, van Marten Lichtenbelt WD, Wouters EFM . Analysis of body water compartments in relation to tissue depletion in clinically stable patients with chronic obstructive pulmonary disease Am J Clin Nutr 1997 65: 88–94.
Forbes GB . Exercise and lean weight: the influence of body weight Nutr Rev 1992 50: 157–161.
Heymsfield SB, Gallagher D, Poehlman ET et al. Menopausal changes in body composition and energy expenditure Exp Gerontol 1994 29: 377–389.
Guo SS, Zeller C, Chumlea WC, Siervogel RM . Aging, body composition, and lifestyle: the Fels Longitudinal Study Am J Clin Nutr 1999 70: 405–411.
Beer-Borst S, Morabia A, Hercberg S, Vitek O, Bernstein MS, Glalan P, Galasso R, Giampaoli S, Houterman S, McCrum E, Panico S, Pannozzo F, Preziosi P, Ribas L, Serra-Majem, Verschuren WMM, Yarnell J, Northridge ME . Obesity and other health determinants across Europe: the EURALIM Project J Epidemiol Commun Health 2000 54: 424–430.
Friedl KE, Moore RJ, Martinez-Lopez LE et al. Lower limit of body fat in healthy active men J Appl Physiol 1994 77: 933–940.
Baumgartner RN, Koehler KM, Gallagher D et al. Epidemiology of sarcopenia among the elderly in New Mexico Am J Epidemiol 1998 147: 755–763.
Deurenberg-Yap M, Schmidt G, van Staveren WA, Deurenberg P . The paradox of low body mass index and high body fat percentage among Chinese, Malays and Indians in Singapore Int J Obes Relat Metab Disord 2000 24: 1011–1017.
Pichard C, Kyle UG, Bracco D, Slosman DO, Morabia A, Schutz Y . Reference values of fat-free and fat masses by bioelectrical impedance analysis in 3393 healthy subjects Nutrition 2000 16: 245–254.
Acknowledgements
We thank the Foundation Nutrition 2000 Plus for its financial support. We are indebted to the dietitians at the Geneva University Hospital for their help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schutz, Y., Kyle, U. & Pichard, C. Fat-free mass index and fat mass index percentiles in Caucasians aged 18–98 y. Int J Obes 26, 953–960 (2002). https://doi.org/10.1038/sj.ijo.0802037
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.ijo.0802037
Keywords
This article is cited by
-
Incident low muscle mass is associated with greater lung disease and lower circulating leptin in a tobacco-exposed longitudinal cohort
Respiratory Research (2023)
-
Irradiation dose to the swallowing apparatus impacts nutritional status in head and neck cancer patients—results from the prospective randomized HEADNUT trial
Strahlentherapie und Onkologie (2023)
-
Body composition assessment in a large cohort of Olympic athletes with different training loads: possible reference values for fat mass and fat-free mass domains
Acta Diabetologica (2023)
-
Effect of cadmium exposure on body composition deterioration: A propensity score-matched cohort study
Environmental Science and Pollution Research (2023)
-
Sarcopenia is associated with cardiovascular risk in men with COPD, independent of adiposity
Respiratory Research (2022)