Abstract
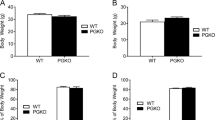
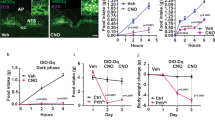
Reduced food intake brings about an adaptive decrease in energy expenditure that contributes to the recidivism of obesity after weight loss. Insulin and leptin inhibit food intake through actions in the central nervous system that are partly mediated by the transcription factor FoxO1. We show that FoxO1 ablation in pro-opiomelanocortin (Pomc)-expressing neurons in mice (here called Pomc-Foxo1−/− mice) increases Carboxypeptidase E (Cpe) expression, resulting in selective increases of α-melanocyte–stimulating hormone (α-Msh) and carboxy-cleaved β-endorphin, the products of Cpe-dependent processing of Pomc. This neuropeptide profile is associated with decreased food intake and normal energy expenditure in Pomc-Foxo1−/− mice. We show that Cpe expression is downregulated by diet-induced obesity and that FoxO1 deletion offsets the decrease, protecting against weight gain. Moreover, moderate Cpe overexpression in the arcuate nucleus phenocopies features of the FoxO1 mutation. The dissociation of food intake from energy expenditure in Pomc-Foxo1−/− mice represents a model for therapeutic intervention in obesity and raises the possibility of targeting Cpe to develop weight loss medications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Yach, D., Stuckler, D. & Brownell, K.D. Epidemiologic and economic consequences of the global epidemics of obesity and diabetes. Nat. Med. 12, 62–66 (2006).
Bray, G.A. Lifestyle and pharmacological approaches to weight loss: efficacy and safety. J. Clin. Endocrinol. Metab. 93, S81–88 (2008).
Schwartz, M.W., Woods, S.C., Porte, D. Jr., Seeley, R.J. & Baskin, D.G. Central nervous system control of food intake. Nature 404, 661–671 (2000).
Plum, L., Belgardt, B.F. & Bruning, J.C. Central insulin action in energy and glucose homeostasis. J. Clin. Invest. 116, 1761–1766 (2006).
Woods, S.C., Lotter, E.C., McKay, L.D. & Porte, D. Jr. Chronic intracerebroventricular infusion of insulin reduces food intake and body weight of baboons. Nature 282, 503–505 (1979).
McGowan, M.K., Andrews, K.M., Fenner, D. & Grossman, S.P. Chronic intrahypothalamic insulin infusion in the rat: behavioral specificity. Physiol. Behav. 54, 1031–1034 (1993).
Benoit, S.C. et al. The catabolic action of insulin in the brain is mediated by melanocortins. J. Neurosci. 22, 9048–9052 (2002).
Accili, D. & Arden, K.C. FoxOs at the Crossroads of Cellular Metabolism, Differentiation, and Transformation. Cell 117, 421–426 (2004).
Kitamura, T. et al. Forkhead protein FoxO1 mediates Agrp-dependent effects of leptin on food intake. Nat. Med. 12, 534–540 (2006).
Kim, M.S. et al. Role of hypothalamic Foxo1 in the regulation of food intake and energy homeostasis. Nat. Neurosci. 9, 901–906 (2006).
Fukuda, M. et al. Monitoring FoxO1 localization in chemically identified neurons. J. Neurosci. 28, 13640–13648 (2008).
Creemers, J.W. et al. Agouti-related protein is posttranslationally cleaved by proprotein convertase 1 to generate agouti-related protein (AGRP)83-132: interaction between AGRP83-132 and melanocortin receptors cannot be influenced by syndecan-3. Endocrinology 147, 1621–1631 (2006).
Nillni, E.A. Regulation of prohormone convertases in hypothalamic neurons: implications for prothyrotropin-releasing hormone and proopiomelanocortin. Endocrinology 148, 4191–4200 (2007).
Pritchard, L.E. & White, A. Neuropeptide processing and its impact on melanocortin pathways. Endocrinology 148, 4201–4207 (2007).
Fox, D.L. & Good, D.J. Nescient helix-loop-helix 2 interacts with signal transducer and activator of transcription 3 to regulate transcription of prohormone convertase 1/3. Mol. Endocrinol. 22, 1438–1448 (2008).
Sanchez, V.C. et al. Regulation of hypothalamic prohormone convertases 1 and 2 and effects on processing of prothyrotropin-releasing hormone. J. Clin. Invest. 114, 357–369 (2004).
Benjannet, S., Rondeau, N., Day, R., Chretien, M. & Seidah, N.G. PC1 and PC2 are proprotein convertases capable of cleaving proopiomelanocortin at distinct pairs of basic residues. Proc. Natl. Acad. Sci. USA 88, 3564–3568 (1991).
Perone, M.J., Ahmed, I., Linton, E.A. & Castro, M.G. Procorticotrophin releasing hormone is endoproteolytically processed by the prohormone convertase PC2 but not by PC1 within stably transfected CHO-K1 cells. Biochem. Soc. Trans. 24, 497S (1996).
Brakch, N. et al. Role of prohormone convertases in pro-neuropeptide Y processing: coexpression and in vitro kinetic investigations. Biochemistry 36, 16309–16320 (1997).
Viale, A. et al. Cellular localization and role of prohormone convertases in the processing of pro-melanin concentrating hormone in mammals. J. Biol. Chem. 274, 6536–6545 (1999).
Allen, R.G. et al. Altered processing of pro-orphanin FQ/nociceptin and pro-opiomelanocortin-derived peptides in the brains of mice expressing defective prohormone convertase 2. J. Neurosci. 21, 5864–5870 (2001).
Furuta, M. et al. Severe defect in proglucagon processing in islet A-cells of prohormone convertase 2 null mice. J. Biol. Chem. 276, 27197–27202 (2001).
Zhu, X. et al. Disruption of PC1/3 expression in mice causes dwarfism and multiple neuroendocrine peptide processing defects. Proc. Natl. Acad. Sci. USA 99, 10293–10298 (2002).
Lloyd, D.J., Bohan, S. & Gekakis, N. Obesity, hyperphagia and increased metabolic efficiency in Pc1 mutant mice. Hum. Mol. Genet. 15, 1884–1893 (2006).
Jackson, R.S. et al. Obesity and impaired prohormone processing associated with mutations in the human prohormone convertase 1 gene. Nat. Genet. 16, 303–306 (1997).
Naggert, J.K. et al. Hyperproinsulinaemia in obese fat/fat mice associated with a carboxypeptidase E mutation which reduces enzyme activity. Nat. Genet. 10, 135–142 (1995).
Chen, H. et al. Missense polymorphism in the human carboxypeptidase E gene alters enzymatic activity. Hum. Mutat. 18, 120–131 (2001).
Che, F.Y. et al. Identification of peptides from brain and pituitary of Cpe(fat)/Cpe(fat) mice. Proc. Natl. Acad. Sci. USA 98, 9971–9976 (2001).
Cawley, N.X. et al. The carboxypeptidase E knockout mouse exhibits endocrinological and behavioral deficits. Endocrinology 145, 5807–5819 (2004).
Balthasar, N. et al. Leptin receptor signaling in POMC neurons is required for normal body weight homeostasis. Neuron. 42, 983–991 (2004).
Belgardt, B.F. et al. PDK1 Deficiency in POMC-expressing cells reveals FOXO1-dependent and -independent pathways in control of energy homeostasis and stress response. Cell. Metab. 7, 291–301 (2008).
Buettner, C. et al. Leptin controls adipose tissue lipogenesis via central, STAT3-independent mechanisms. Nat. Med. 14, 667–675 (2008).
Leibel, R.L., Rosenbaum, M. & Hirsch, J. Changes in energy expenditure resulting from altered body weight. N. Engl. J. Med. 332, 621–628 (1995).
Berman, Y., Mzhavia, N., Polonskaia, A. & Devi, L.A. Impaired prohormone convertases in Cpe(fat)/Cpe(fat) mice. J. Biol. Chem. 276, 1466–1473 (2001).
Miller, R. et al. Obliteration of alpha-melanocyte-stimulating hormone derived from POMC in pituitary and brains of PC2-deficient mice. J. Neurochem. 86, 556–563 (2003).
Zhu, X., Rouille, Y., Lamango, N.S., Steiner, D.F. & Lindberg, I. Internal cleavage of the inhibitory 7B2 carboxyl-terminal peptide by PC2: a potential mechanism for its inactivation. Proc. Natl. Acad. Sci. USA 93, 4919–4924 (1996).
Overton, J.M. & Williams, T.D. Behavioral and physiologic responses to caloric restriction in mice. Physiol. Behav. 81, 749–754 (2004).
Wertz-Lutz, A.E., Daniel, J.A., Clapper, J.A., Trenkle, A. & Beitz, D.C. Prolonged, moderate nutrient restriction in beef cattle results in persistently elevated circulating ghrelin concentrations. J. Anim. Sci. 86, 564–575 (2008).
Nakae, J. et al. The forkhead transcription factor foxo1 regulates adipocyte differentiation. Dev. Cell 4, 119–129 (2003).
Kitamura, T. et al. A Foxo/Notch pathway controls myogenic differentiation and fiber type specification. J. Clin. Invest. 117, 2477–2485 (2007).
Nicolas, P. & Li, C.H. Beta-endorphin-(1-27) is a naturally occurring antagonist to etorphine-induced analgesia. Proc. Natl. Acad. Sci. USA 82, 3178–3181 (1985).
Yanagita, K., Shiraishi, J., Fujita, M. & Bungo, T. Effects of N-terminal fragments of beta-endorphin on feeding in chicks. Neurosci. Lett. 442, 140–142 (2008).
Banks, A.S. et al. SirT1 gain of function increases energy efficiency and prevents diabetes in mice. Cell Metab. 8, 333–341 (2008).
Kitamura, Y.I. et al. FoxO1 protects against pancreatic beta cell failure through NeuroD and MafA induction. Cell Metab. 2, 153–163 (2005).
Bence, K.K. et al. Neuronal PTP1B regulates body weight, adiposity and leptin action. Nat. Med. 12, 917–924 (2006).
Nilaweera, K.N., Barrett, P., Mercer, J.G. & Morgan, P.J. Precursor-protein convertase 1 gene expression in the mouse hypothalamus: differential regulation by ob gene mutation, energy deficit and administration of leptin, and coexpression with prepro-orexin. Neuroscience 119, 713–720 (2003).
Rosenbaum, M. et al. Low-dose leptin reverses skeletal muscle, autonomic, and neuroendocrine adaptations to maintenance of reduced weight. J. Clin. Invest. 115, 3579–3586 (2005).
Paik, J.H. et al. FoxOs are lineage-restricted redundant tumor suppressors and regulate endothelial cell homeostasis. Cell 128, 309–323 (2007).
Plum, L. et al. Enhanced PIP3 signaling in POMC neurons causes KATP channel activation and leads to diet-sensitive obesity. J. Clin. Invest. 116, 1886–1901 (2006).
Plum, L. et al. Enhanced leptin-stimulated Pi3k activation in the CNS promotes white adipose tissue transdifferentiation. Cell Metab. 6, 431–445 (2007).
Woronowicz, A. et al. Absence of carboxypeptidase E leads to adult hippocampal neuronal degeneration and memory deficits. Hippocampus 18, 1051–1063 (2008).
Wardlaw, S.L. Regulation of β-endorphin, corticotropin-like intermediate lobe peptide, and α-melanotropin–stimulating hormone in the hypothalamus by testosterone. Endocrinology 119, 19–24 (1986).
Tsigos, C., Crosby, S.R., Gibson, S., Young, R.J. & White, A. Proopiomelanocortin is the predominant adrenocorticotropin-related peptide in human cerebrospinal fluid. J. Clin. Endocrinol. Metab. 76, 620–624 (1993).
Papadopoulos, A.D. & Wardlaw, S.L. Endogenous MSH modulates the hypothalamic-pituitary-adrenal response to the cytokine interleukin-1β. J. Neuroendocrinol. 11, 315–319 (1999).
Jaffe, S.B., Sobieszczyk, S. & Wardlaw, S.L. Effect of opioid antagonism on β-endorphin processing and proopiomelanocortin-peptide release in the hypothalamus. Brain Res. 648, 24–31 (1994).
Acknowledgements
Supported by Deutsche Forschungsgemeinschaft PL542/1-1 (L.P.), US National Institutes of Health DK57539 and DK58282 (D.A.), DK80003 (S.L.W.) and DK63608 (Columbia Diabetes and Endocrinology Research Center). We thank R. Leibel for insightful discussions, L. Zeltser and S. Padilla for help with in situ hybridization, N. Seidah (Clinical Research Institute of Montreal) and D. Good (University of Massachusetts) for plasmids encoding pCsk1, A. White (University of Manchester) for neuropeptide antisera, and M. Low (Oregon Health Sciences University) for Pomc-Gfp transgenic mice, Y. Liu for technical assistance and members of the Accili and Wardlaw laboratories for stimulating discussions. R.A.D. is an American Cancer Society Research Professor and an Ellison Medical Foundation Senior Scholar and is supported by the Robert A. and Renee E. Belfer Family Institute for Innovative Cancer Science.
Author information
Authors and Affiliations
Contributions
L.P., H.V.L., R.D., J.T., K.S.A., M.M., A.J.K. and S.L.W. performed experiments and analyzed data. N.X.C. and J.-H.P. generated reagents used for experiments. L.P., H.V.L., Y.P.L., R.A.D., S.L.W. and D.A. designed the studies, analyzed the data and wrote the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 and Supplementary Table 1 (PDF 2067 kb)
Rights and permissions
About this article
Cite this article
Plum, L., Lin, H., Dutia, R. et al. The obesity susceptibility gene Cpe links FoxO1 signaling in hypothalamic pro-opiomelanocortin neurons with regulation of food intake. Nat Med 15, 1195–1201 (2009). https://doi.org/10.1038/nm.2026
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.2026
This article is cited by
-
Carboxypeptidase E conditional knockout mice exhibit learning and memory deficits and neurodegeneration
Translational Psychiatry (2023)
-
Hippocampal delivery of neurotrophic factor-α1/carboxypeptidase E gene prevents neurodegeneration, amyloidosis, memory loss in Alzheimer’s Disease male mice
Molecular Psychiatry (2023)
-
Epigenetic regulation of beta-endorphin synthesis in hypothalamic arcuate nucleus neurons modulates neuropathic pain in a rodent pain model
Nature Communications (2023)
-
Signaling pathways in obesity: mechanisms and therapeutic interventions
Signal Transduction and Targeted Therapy (2022)
-
FoxO1 Regulates Neuropeptide Y and Pro-opiomelanocortin in the Hypothalamus of Rat Offspring Small for Gestational Age
Reproductive Sciences (2022)