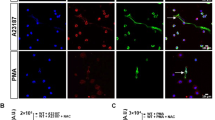
Abstract
Neutrophils encounter and 'prioritize' many chemoattractants in their pursuit of bacteria. Here we tested the possibility that the phosphatase PTEN is responsible for the prioritization of chemoattractants. Neutrophils induced chemotaxis by two separate pathways, the phosphatidylinositol-3-OH kinase (PI(3)K) phosphatase and tensin homolog (PTEN) pathway, and the p38 mitogen-activated protein kinase pathway, with the p38 pathway dominating over the PI(3)K pathway. Pten−/− neutrophils could not prioritize chemoattractants and were 'distracted' by chemokines when moving toward bacterial chemoattractants. In opposing gradients, PTEN became distributed throughout the cell circumference, which inhibited all PI(3)K activity, thus permitting 'preferential' migration toward bacterial products via phospholipase A2 and p38. Such prioritization was defective in Pten−/− neutrophils, which resulted in defective bacterial clearance in vivo. Our data identify a PTEN-dependent mechanism in neutrophils to prioritize, 'triage' and integrate responses to multiple chemotactic cues.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Change history
21 July 2008
In the version of this article initially published, phosphatidylinositol-4,5-bisphosphate (PtdIns(4,5)P2) is incorrectly identified as phosphatidylinositol-3,4-bisphosphate (PtdIns(3,4)P2). The error has been corrected in the HTML and PDF versions of the article.
References
Jin, T., Zhang, N., Long, Y., Parent, C.A. & Devreotes, P.N. Localization of the G protein βγ complex in living cells during chemotaxis. Science 287, 1034–1036 (2000).
Funamoto, S., Meili, R., Lee, S., Parry, L. & Firtel, R.A. Spatial and temporal regulation of 3-phosphoinositides by PI 3-kinase and PTEN mediates chemotaxis. Cell 109, 611–623 (2002).
Chen, L. et al. Two phases of actin polymerization display different dependencies on PI(3,4,5)P3 accumulation and have unique roles during chemotaxis. Mol. Biol. Cell 14, 5028–5037 (2003).
Sun, R. et al. Protein kinase C ζ is required for epidermal growth factor-induced chemotaxis of human breast cancer cells. Cancer Res. 65, 1433–1441 (2005).
Ptasznik, A. et al. A tyrosine kinase signaling pathway accounts for the majority of phosphatidylinositol 3,4,5-trisphosphate formation in chemoattractant-stimulated human neutrophils. J. Biol. Chem. 271, 25204–25207 (1996).
Sasaki, T. et al. Function of PI3Kγ in thymocyte development, T cell activation, and neutrophil migration. Science 287, 1040–1046 (2000).
Stephens, L., Ellson, C. & Hawkins, P. Roles of PI3Ks in leukocyte chemotaxis and phagocytosis. Curr. Opin. Cell Biol. 14, 203–213 (2002).
Huang, Y.E. et al. Receptor-mediated regulation of PI3Ks confines PI(3,4,5)P3 to the leading edge of chemotaxing cells. Mol. Biol. Cell 14, 1913–1922 (2003).
Ferreira, A.M., Isaacs, H., Hayflick, J.S., Rogers, K.A. & Sandig, M. The p110δ isoform of PI3K differentially regulates β1 and β2 integrin-mediated monocyte adhesion and spreading and modulates diapedesis. Microcirculation 13, 439–456 (2006).
Sasaki, A.T. & Firtel, R.A. Regulation of chemotaxis by the orchestrated activation of Ras, PI3K, and TOR. Eur. J. Cell Biol. 85, 873–895 (2006).
Liu, L., Puri, K.D., Penninger, J.M. & Kubes, P. Leukocyte PI3Kγ and PI3Kδ have temporally distinct roles for leukocyte recruitment in vivo. Blood 110, 1191–1198 (2007).
Hirsch, E. et al. Central role for G protein-coupled phosphoinositide 3-kinase gamma in inflammation. Science 287, 1049–1053 (2000).
Heit, B., Liu, L., Colarusso, P., Puri, K.D. & Kubes, P. PI3K accelerates, but is not required for, neutrophil chemotaxis to fMLP. J. Cell Sci. 121, 205–214 (2008).
Maehama, T. & Dixon, J.E. The tumor suppressor, PTEN/MMAC1, dephosphorylates the lipid second messenger, phosphatidylinositol 3,4,5-trisphosphate. J. Biol. Chem. 273, 13375–13378 (1998).
Wu, Y. et al. A requirement of MAPKAPK2 in the uropod localization of PTEN during FMLP-induced neutrophil chemotaxis. Biochem. Biophys. Res. Commun. 316, 666–672 (2004).
Li, Z. et al. Regulation of PTEN by Rho small GTPases. Nat. Cell Biol. 7, 399–404 (2005).
Nishio, M. et al. Control of cell polarity and motility by the PtdIns(3,4,5)P3 phosphatase SHIP1. Nat. Cell Biol. 9, 36–44 (2007).
Subramanian, K.K. et al. Tumor suppressor PTEN is a physiologic suppressor of chemoattractant-mediated neutrophil functions. Blood 109, 4028–4037 (2007).
Damen, J.E. et al. The 145-kDa protein induced to associate with Shc by multiple cytokines is an inositol tetraphosphate and phosphatidylinositol 3,4,5-triphosphate 5-phosphatase. Proc. Natl. Acad. Sci. USA 93, 1689–1693 (1996).
Murdoch, C. & Finn, A. Chemokine receptors and their role in inflammation and infectious diseases. Blood 95, 3032–3043 (2000).
Foxman, E.F., Campbell, J.J. & Butcher, E.C. Multistep navigation and the combinatorial control of leukocyte chemotaxis. J. Cell Biol. 139, 1349–1360 (1997).
Heit, B., Tavener, S., Raharjo, E. & Kubes, P. An intracellular signaling hierarchy determines direction of migration in opposing chemotactic gradients. J. Cell Biol. 159, 91–102 (2002).
Foxman, E.F., Kunkel, E.J. & Butcher, E.C. Integrating conflicting chemotactic signals. The role of memory in leukocyte navigation. J. Cell Biol. 147, 577–588 (1999).
Khan, A.I., Heit, B., Andonegui, G., Colarusso, P. & Kubes, P. Lipopolysaccharide: a p38 MAPK-dependent disrupter of neutrophil chemotaxis. Microcirculation 12, 421–432 (2005).
Chen, L. et al. PLA2 and PI3K/PTEN pathways act in parallel to mediate chemotaxis. Dev. Cell 12, 603–614 (2007).
Bae, Y.S. et al. Differential signaling of formyl peptide receptor-like 1 by Trp-Lys-Tyr-Met-Val-Met-CONH2 or lipoxin A4 in human neutrophils. Mol. Pharmacol. 64, 721–730 (2003).
Lee, J.O. et al. Crystal structure of the PTEN tumor suppressor: implications for its phosphoinositide phosphatase activity and membrane association. Cell 99, 323–334 (1999).
Marino, S. et al. PTEN is essential for cell migration but not for fate determination and tumourigenesis in the cerebellum. Development 129, 3513–3522 (2002).
Tkalcevic, J. et al. Impaired immunity and enhanced resistance to endotoxin in the absence of neutrophil elastase and cathepsin G. Immunity 12, 201–210 (2000).
Ditzel, H.J. The K/BxN mouse: a model of human inflammatory arthritis. Trends Mol. Med. 10, 40–45 (2004).
Kouskoff, V. et al. Organ-specific disease provoked by systemic autoimmunity. Cell 87, 811–822 (1996).
Maccioni, M. et al. Arthritogenic monoclonal antibodies from K/BxN mice. J. Exp. Med. 195, 1071–1077 (2002).
Weljie, A.M., Dowlatabadi, R., Miller, B.J., Vogel, H.J. & Jirik, F.R. An inflammatory arthritis-associated metabolite biomarker pattern revealed by 1H NMR spectroscopy. J. Proteome Res. 6, 3456–3464 (2007).
Solomon, S. et al. Transmission of antibody-induced arthritis is independent of complement component 4 (C4) and the complement receptors 1 and 2 (CD21/35). Eur. J. Immunol. 32, 644–651 (2002).
Iijima, M. & Devreotes, P. Tumor suppressor PTEN mediates sensing of chemoattractant gradients. Cell 109, 599–610 (2002).
Herbert, D.R. et al. Alternative macrophage activation is essential for survival during schistosomiasis and downmodulates T helper 1 responses and immunopathology. Immunity 20, 623–635 (2004).
Hagenbeek, T.J. et al. The loss of PTEN allows TCR αβ lineage thymocytes to bypass IL-7 and pre-TCR-mediated signaling. J. Exp. Med. 200, 883–894 (2004).
Georgel, P. et al. A toll-like receptor 2-responsive lipid effector pathway protects mammals against skin infections with gram-positive bacteria. Infect. Immun. 73, 4512–4521 (2005).
Acknowledgements
We thank C. Benoist (Harvard Medical School) for the KRN mouse line; T.W. Mak (University Health Network) for the loxP-flanked Pten mouse line; and M. Gaestel (Martin Luther University) for the MK2-deficient mice. Supported by the Canadian Institutes of Health Research (P.K.), the Arthritis Society of Canada (F.R.J.), the Alberta Heritage Foundation for Medical Research (P.K.), the Canada Research Chairs Program (P.K. and F.R.J.) and the Calvin, Phoebe and Joan Snyder Chair in Critical Care Research (P.K.).
Author information
Authors and Affiliations
Contributions
B.H. did all experiments unless otherwise noted; C.D., B.J.M. and F.R.J. did the arthritis modeling and managed the bioimaging facility; S.R. provided molecular biology expertise and technical input; Z.G. did all p38 blotting; P.C. managed the imaging facility and aided in the development of the imaging assays; P.K. managed the study, and all experiments except the arthritis model were done in the laboratory of P.K.; and B.H. and P.K. prepared the manuscript with input from the other authors.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–5 and Supplementary Methods (PDF 3347 kb)
Rights and permissions
About this article
Cite this article
Heit, B., Robbins, S., Downey, C. et al. PTEN functions to 'prioritize' chemotactic cues and prevent 'distraction' in migrating neutrophils. Nat Immunol 9, 743–752 (2008). https://doi.org/10.1038/ni.1623
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1623
This article is cited by
-
Electric signals counterbalanced posterior vs anterior PTEN signaling in directed migration of Dictyostelium
Cell & Bioscience (2021)
-
A double-edged sword of immuno-microenvironment in cardiac homeostasis and injury repair
Signal Transduction and Targeted Therapy (2021)
-
The principles of directed cell migration
Nature Reviews Molecular Cell Biology (2021)
-
Regulatory mechanisms of neutrophil migration from the circulation to the airspace
Cellular and Molecular Life Sciences (2021)
-
Anti-inflammatory cellular targets on neutrophils elucidated using a novel cell migration model and confocal microscopy: a clinical supplementation study
Journal of Inflammation (2018)