Abstract
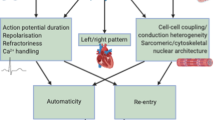
Atrial fibrillation is the most common cardiac arrhythmia, and it increases in prevalence with advancing age to about 6% in people older than 65 years. The chance of developing atrial fibrillation at age 40 years or older is about 25% in men and women. This arrhythmia accounts for about one-third of all strokes, and 30% of all patients with atrial fibrillation have a family history of the disease. In 1997, Brugada et al. identified the first locus for familial atrial fibrillation on chromosome 10q22–24 in three different Spanish families. Since that time, seven further loci have been mapped and four relevant genes identified. All these genes encode potassium-channel subunits. The mechanism of action by which all four genes induce atrial fibrillation is via shortening of the action potential duration and atrial effective refractory period. The consistency of the mechanism of action beckons the development of therapy specifically targeted to prevent these molecular events. In addition to monogenic diseases, patients with structural heart disease are predisposed to atrial fibrillation by inherited DNA polymorphisms. The development of chips with hundreds of thousands of single-nucleotide polymorphisms to perform genome-wide scans will elucidate over the next few years the single-nucleotide polymorphisms that predispose to atrial fibrillation. Within the next decade, most of the genes responsible for atrial fibrillation and the single-nucleotide polymorphisms that confer predisposition will probably be identified, and therapies will be developed on the basis of individuals' genomic profiles. In this review I provide an overview of the understanding of the relevant genetic mutations that have been identified so far, and briefly discuss what implications this information might have for practice.
Key Points
-
Atrial fibrillation is the most common atrial arrhythmia and accounts for over one-third of all strokes
-
A significant proportion of atrial fibrillation has a genetic origin or predisposition
-
Four genes have been identified to be responsible for atrial fibrillation and the loci (chromosomal position) of three other genes have been mapped, but they have not yet been identified
-
The four genes currently identified as responsible for familial atrial fibrillation encode for a cardiac potassium channel and the defect is due to a missense mutation
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Rensma P et al. (1988) Length of excitation wave and susceptibility to reentrant atrial arrhythmias in normal conscious dogs. Circ Res 62: 395–410
Nattel S et al. (2000) Basic mechanisms of atrial fibrillation—very new insights into very old ideas. Annu Rev Physiol 62: 51–77
Nattel S (1999) Atrial electrophysiological remodeling caused by rapid atrial activation: underlying mechanisms and clinical relevance to atrial fibrillation. Cardiovasc Res 42: 298–308
Feinberg WM et al. (1995) Prevalence, age distribution, and gender of patients with atrial fibrillation. Analysis and implications. Arch Intern Med 155: 469–473
Go AS et al. (2001) Prevalence of diagnosed atrial fibrillation in adults: national implications for rhythm management and stroke prevention: the AnTicoagulation and Risk Factors in Atrial Fibrillation (ATRIA) Study. JAMA 285: 2370–2375
Lloyd-Jones DM et al. (2004) Lifetime risk for development of atrial fibrillation: the Framingham Heart Study. Circulation 110: 1042–1046
Rubenstein JJ et al. (1972) Clinical spectrum of the sick sinus syndrome. Circulation 46: 5–13
Terent A and Andersson B (1981) The prognosis for patients with cerebrovascular stroke and transient ischemic attacks. Ups J Med Sci 86: 63–74
Wolf PA et al. (1987) Atrial fibrillation: a major contributor to stroke in the elderly: the Framingham Study. Arch Intern Med 147: 1561–1564
The Boston Area Anticoagulation trial for Atrial Fibrillation Investigators (1990) The effect of low-dose warfarin on the risk of stroke in patients with nonrheumatic atrial fibrillation. N Engl J Med 323: 1505–1511
Darbar D et al. (2003) Familial atrial fibrillation is a genetically heterogeneous disorder. J Am Cardiol 41: 2185–2192
Kopecky SL et al. (1987) The natural history of lone atrial fibrillation. A population-based study over three decades. N Engl J Med 317: 669–674
Wolff L (1943) Familiar auricular fibrillation. N Engl J Med 229: 396–397
Brugada R (2005) Is atrial fibrillation a genetic disease? J Cardiovasc Electrophysiol 16: 553–556
Fox CS et al. (2004) Parental atrial fibrillation as a risk factor for atrial fibrillation in offspring. JAMA 291: 2851–2855
Brugada R et al. (1997) Identification of a genetic locus for familial atrial fibrillation. N Engl J Med 336: 905–911
Chen YH et al. (2003) KCNQ1 gain-of-function mutation in familial atrial fibrillation. Science 299: 251–254
Barhanin J et al. (1996) KVLQT1 and IsK (minK) proteins associate to form the IKs cardiac potassium current. Nature 384: 78–80
Sanguinetti MC et al. (1996) Coassembly of KVLQT1 and minK (IsK) proteins to form cardiac IKs potassium channel. Nature 384: 80–83
Tinel N et al. (2000) KCNE2 confers background current characteristics to the cardiac KCNQ1 potassium channel. Embo J 19: 6326–6330
Schroeder BC et al. (2000) A constitutively open potassium channel formed by KCNQ1 and KCNE3. Nature 403: 196–199
Grunnet M et al. (2002) KCNE4 is an inhibitory subunit to the KCNQ1 channel. J Physiol 542: 119–130
Ellinor PT et al. (2004) Mutations in the long QT gene, KCNQ1, are an uncommon cause of atrial fibrillation. Heart 90: 1487–1488
Yang Y et al. (2004) Identification of a KCNE2 gain-of-function mutation in patients with familial atrial fibrillation. Am J Hum Genet 75: 899–905
Xia M et al. (2005) A Kir2.1 gain-of-function mutation underlies familial atrial fibrillation. Biochem Biophys Res Commun 332: 1012–1019
Li J et al. (2004) Transgenic upregulation of IK1 in the mouse heart leads to multiple abnormalities of cardiac excitability. Am J Physiol Heart Circ Physiol 287: H2790–H2802
Dobrev D et al. (2001) Molecular basis of downregulation of G-protein-coupled inward rectifying K+ current (IK,ACh) in chronic human atrial fibrillation: decrease in GIRK4 mRNA correlates with reduced IK,ACh and muscarinic receptor-mediated shortening of action potentials. Circulation 104: 2551–2557
Dobrev D et al. (2002) Human inward rectifier potassium channels in chronic and postoperative atrial fibrillation. Cardiovasc Res 54: 397–404
Van Wagoner DR et al. (1997) Outward K+ current densities and Kv1.5 expression are reduced in chronic human atrial fibrillation. Circ Res 80: 772–781
Van Wagoner DR (2003) Electrophysiological remodeling in human atrial fibrillation. Pacing Clin Electrophysiol 26: 1572–1575
Gussak I et al. (2000) Idiopathic short QT interval: a new clinical syndrome? Cardiology 94: 99–102
Hong K et al. (2005) Short QT syndrome and atrial fibrillation caused by mutation in KCNH2. J Cardiovasc Electrophysiol 16: 394–396
Oberti C et al. (2004) Genome-wide linkage scan identifies a novel genetic locus on chromosome 5p13 for neonatal atrial fibrillation associated with sudden death and variable cardiomyopathy. Circulation 110: 3753–3759
Riordan JR et al. (1989) Identification of the cystic fibrosis gene: cloning and characterization of complementary DNA. Science 245: 1066–1073
Venter JC et al. (2001) The sequence of the human genome. Science 291: 1304–1351
Hille B (2001) Ionic Channels of Excitable Membranes, edn 3. Sunderland: Sinauer Associates
Gargus J (2003) Unraveling monogenic channelopathies and their implications for complex polygenic disease. Am J Hum Genet 72: 785–803
Gruver EJ et al. (1999) Familial hypertrophic cardiomyopathy and atrial fibrillation caused by Arg663His beta-cardiac myosin heavy chain mutation. Am J Cardiol 83: 13H–18H
Lundquist AL et al. (2005) Expression of multiple KCNE genes in human heart may enable variable modulation of IKs . J Mol Cell Cardiol 38: 277–287
Marian AJ and Roberts R (2001) The molecular genetic basis for hypertrophic cardiomyopathy. J Mol Cell Cardiol 33: 655–670
Ahmad F et al. (2000) Identification and characterization of a novel gene (C4orf5) located on human chromosome 4q with specific expression in cardiac and skeletal muscle. Genomics 70: 347–353
Gollob MH et al. (2001) Identification of a gene responsible for familial Wolff–Parkinson–White syndrome. N Engl J Med 344: 1823–1831
Sidhu J et al. (2005) A transgeneic mouse model of ventricular preexcitation and atrioventricular reentrant tachycardia induced by an AMP-activated kinase loss-of-function mutation responsible for Wolff–Parkinson–White syndrome. Circulation 111: 21–29
Arad M et al. (2002) Constitutively active AMP kinase mutations cause glycogen storage disease mimicking hypertrophic cardiomyopathy. J Clin Invest 109: 357–362
Tsai CT et al. (2004) Renin–angiotensin system gene polymorphisms and atrial fibrillation. Circulation 109: 1640–1646
Firouzi M et al. (2004) Association of human connexin40 gene polymorphisms with atrial vulnerability as a risk factor for idiopathic atrial fibrillation. Circ Res 95: e29–e33
Lai L-P et al. (2002) Association of the human minK gene 38G allele with atrial fibrillation: evidence of possible genetic control on the pathogenesis of atrial fibrillation. Am Heart J 144: 485–490
Kuo A et al. (2003) Crystal structure of the potassium channel KirBac1.1 in the closed state. Science 300: 1922–1926
Acknowledgements
The assistance of Susan Slater and Fran Baas on the preparation of this manuscript is greatly appreciated. This work is supported by grants from the National Heart, Lung and Blood Institute; Specialized Centers of Research; the National Institutes of Health Training Center in Molecular Cardiology; and the Canadian Institutes of Health Research.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Roberts, R. Mechanisms of Disease: genetic mechanisms of atrial fibrillation. Nat Rev Cardiol 3, 276–282 (2006). https://doi.org/10.1038/ncpcardio0509
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpcardio0509
This article is cited by
-
Points to Consider: Genetic Evaluation of the Cardiology Patient
Journal of Cardiovascular Translational Research (2009)
-
KATP channel mutation confers risk for vein of Marshall adrenergic atrial fibrillation
Nature Clinical Practice Cardiovascular Medicine (2007)
-
The worldwide social burden of atrial fibrillation: What should be done and where do we go?
Journal of Interventional Cardiac Electrophysiology (2007)
-
Reversible atrial fibrillation secondary to a mega-oesophagus
BMC Ear, Nose and Throat Disorders (2006)
-
The worldwide social burden of atrial fibrillation: What should be done and where do we go?
Journal of Interventional Cardiac Electrophysiology (2006)