Abstract
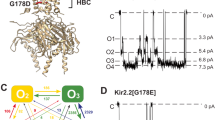
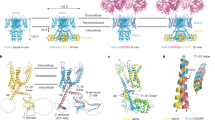
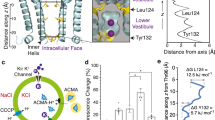
Inwardly rectifying potassium (Kir) channels are gated by the membrane phospholipid phosphatidylinositol-4,5-bisphosphate (PtdIns(4,5)P2). Among them, Kir3 requires additional molecules, such as the βγ subunits of G proteins or intracellular sodium, for channel gating. Using an interactive computational-experimental approach, we show that sodium sensitivity of Kir channels involves the side chains of an aspartate and a histidine located across from each other in a crucial loop in the cytosolic domain, as well as the backbone carbonyls of two more residues and a water molecule. The location of the coordination site in the vicinity of a conserved arginine shown to affect channel–PtdIns(4,5)P2 interactions suggests that sodium triggers a structural switch that frees the crucial arginine. Mutations of the aspartate and the histidine that affect sodium sensitivity also enhance the channel's sensitivity to PtdIns(4,5)P2. Furthermore, on the basis of the molecular characteristics of the coordination site, we identify and confirm experimentally a sodium-sensitive phenotype in Kir5.1.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Accession codes
References
Tamargo, J., Caballero, R., Gómez, R., Valenzuela, C. & Delpón, E. Pharmacology of cardiac potassium channels. Cardiovasc. Res. 62, 9–33 (2004).
Isomoto, S., Kondo, C. & Kurachi, Y. Inwardly rectifying potassium channels: their molecular heterogeneity and function. Jpn. J. Physiol. 47, 11–39 (1997).
Nerbonne, J.M., Nichols, C.G., Schwarz, T.L. & Escande, D. Genetic manipulation of cardiac K+ channel function in mice: what have we learned, and where do we go from here? Circ. Res. 89, 944–956 (2001).
Hilgemann, D.W., Feng, S. & Nasuhoglu, C. The complex and intriguing lives of PIP2 with ion channels and transporters. Sci. STKE doi: 10.1126/stke.2001.111.re19 (4 December 2001).
Lopes, C.M.B., Zhang, H., Rohacs, T., Yang, J. & Logothetis, D.E. Alterations in conserved interactions between PIP2 and Kir channels underlie channelopathies. Neuron 34, 933–944 (2002).
Krapivinsky, G. et al. The G-protein-gated atrial K+ channel IKACh is a heteromultimer of two inwardly rectifying K+-channel proteins. Nature 374, 135–141 (1995).
Chan, K.W. et al. A recombinant inwardly rectifying potassium channel coupled to GTP-binding proteins. J. Gen. Physiol. 107, 381–397 (1996).
Hedin, K.E., Lim, N.F. & Clapham, D.E. Cloning of a Xenopus laevis inwardly rectifying K+ channel subunit that permits GIRK1 expression of IKACh currents in oocytes. Neuron 16, 423–429 (1996).
Chan, K.W., Sui, J.L., Vivaudou, M. & Logothetis, D.E. Control of channel activity through a unique amino acid residue of a G protein-gated inwardly rectifying K+ channel subunit. Proc. Natl. Acad. Sci. USA 93, 14193–14198 (1996b).
Vivaudou, M. et al. Probing the G-protein regulation of GIRK1 and GIRK4, the two subunits of the KACh channel, using functional homomeric mutants. J. Biol. Chem. 272, 31553–31560 (1997).
Sui, J.L., Petit-Jacques, J. & Logothetis, D.E. Activation of the atrial KACh channel by the βγ subunits of G proteins or intracellular Na+ ions depends on the presence of phosphatidylinositol phosphates. Proc. Natl. Acad. Sci. USA 95, 1307–1312 (1998).
Lesage, F. et al. Molecular properties of neuronal G-protein-activated inwardly rectifying K+ channels. J. Biol. Chem. 270, 28660–28667 (1995).
Ho, I.H. & Murrell-Lagnado, R.D. Molecular determinants for sodium-dependent activation of G protein-gated K+ channels. J. Biol. Chem. 274, 8639–8648 (1999).
Ho, I.H. & Murrell-Lagnado, R.D. Molecular mechanism for sodium-dependent activation of G protein-gated K+ channels. J. Physiol. (Lond.) 520, 645–651 (1999).
Sui, J.L., Chan, K.W. & Logothetis, D.E. Na+ activation of the muscarinic K+ channel by a G-protein-independent mechanism. J. Gen. Physiol. 108, 381–391 (1996).
Zhang, H., He, C., Yan, X., Mirshahi, T. & Logothetis, D.E. Activation of inwardly rectifying K+ channels by distinct PtdIns(4,5)P2 interactions. Nat. Cell Biol. 1, 183–188 (1999).
Tristani-Firouzi, M. et al. Functional and clinical characterization of KCNJ2 mutations associated with LQT7 (Andersen syndrome). J. Clin. Invest. 110, 381–388 (2002).
Nishida, M. & MacKinnon, R. Structural basis of inward rectification: cytoplasmic pore of the G protein-gated inward rectifier GIRK1 at 1.8 Å resolution. Cell 111, 957–965 (2002).
Pegan, S. et al. Cytoplasmic domain structures of Kir2.1 and Kir3.1 show sites for modulating gating and rectification. Nat. Neurosci. 8, 279–287 (2005).
Stevens, E.B., Woodward, R., Ho, I.H. & Murrell-Lagnado, R. Identification of regions that regulate the expression and activity of G protein-gated inward rectifier K+ channels in Xenopus oocytes. J. Physiol. (Lond.) 503, 547–562 (1997).
Di Cera, E. et al. The Na+ binding site of thrombin. J. Biol. Chem. 270, 22089–22092 (1995).
Glusker, J.P. Structural aspects of metal liganding to functional groups in proteins. Adv. Protein Chem. 42, 1–76 (1991).
Karpen, M.E., Tobias, D.J. & Brooks, C.L., III. Statistical clustering techniques for the analysis of long molecular dynamics trajectories: analysis of 2.2-ns trajectories of YPGDV. Biochemistry 32, 412–420 (1993).
Carpenter, G.A. & Grossberg, S. ART 2: self-organization of stable category recognition codes for analog input patterns. Appl. Opt. 26, 4919–4930 (1987).
Pao, Y.-H. Adaptive Pattern Recognition and Neural Networks (Addison-Wesley, New York, 1989).
Brooks, B.R. et al. CHARMM: a program for macromolecular energy, minimization, and dynamics calculations. J. Comput. Chem. 4, 187–217 (1983).
Beglov, D. & Roux, B. Dominant solvation effects from primary shell of hydration: approximation for molecular dynamics simulations. Biopolymers 35, 171–178 (1995).
Zerangue, N., Schwappach, B., Jan, Y.N. & Jan, L.Y. A new ER trafficking signal regulates the subunit stoichiometry of plasma membrane KATP channels. Neuron 22, 537–548 (1999).
Lamoureux, G. & Roux, B. Absolute hydration free energy scale for alkali and halide ions established from simulations with a polarizable force field. J. Phys. Chem. B 110, 3308–3322 (2006).
Wang, P., Wesdemiotis, C., Kapota, C. & Ohanessian, G. The sodium ion affinities of simple di-, tri-, and tetrapeptides. J. Am. Soc. Mass Spectrom. 18, 541–552 (2007).
Kapota, C. & Ohanessian, G. The low energy tautomers and conformers of the dipeptides HisGly and GlyHis and of their sodium ion complexes in the gas phase. Phys. Chem. Chem. Phys. 7, 3744–3755 (2005).
Yu, X.M. The role of intracellular sodium in the regulation of NMDA-receptor-mediated channel activity and toxicity. Mol. Neurobiol. 33, 63–80 (2006).
Dryer, S.E. Na+-activated K+ channels: A new family of large-conductance ion channels. Trends Neurosci. 17, 155–160 (1994).
Salkoff, L., Butler, A., Ferreira, G., Santi, C. & Wei, A. High-conductance potassium channels of the SLO family. Nat. Rev. Neurosci. 7, 921–931 (2006).
Komwatana, P., Dinudom, A., Young, J.A. & Cook, D.I. Cytosolic Na+ controls and epithelial Na+ channel via the Go guanine nucleotide-binding regulatory protein. Proc. Natl. Acad. Sci. USA 93, 8107–8111 (1996).
Harding, M.M. The architecture of metal coordination groups in proteins. Acta Crystallogr. D Biol. Crystallogr. 60, 849–859 (2004).
Logothetis, D.E., Jin, T., Lupyan, D. & Rosenhouse-Dantsker, A. Phosphoinositide-mediated gating of inwardly rectifying K+ channels. Pflugers Arch. 455, 83–95 (2007a).
Logothetis, D.E., Lupyan, D. & Rosenhouse-Dantsker, A. Diverse Kir modulators act in close proximity to residues implicated in phosphoinositide binding. J. Physiol. (Lond.) 582, 953–965 (2007b).
Nishida, M., Cadene, M., Chait, B.T. & Mackinnon, R. Crystal structure of a Kir3.1-prokaryotic Kir channel chimera. EMBO J. 26, 4005–4015 (2007).
Inanobe, A., Matsuura, T., Nakagawa, A. & Kurachi, Y. Structural diversity in the cytoplasmic region of G protein-gated inward rectifier K+ channels. Channels 1, 39–45 (2007).
Tucker, S.J., Imbrici, P., Salvatore, L., D'Adamo, M.C. & Pessia, M. pH dependence of the inwardly rectifying potassium channel, Kir5.1, and localization in renal tubular epithelia. J. Biol. Chem. 275, 16404–16407 (2000).
Wu, J., Xu, H., Shen, W. & Jiang, C. Expression and coexpression of CO2-sensitive Kir channels in brainstem neurons of rats. J. Membr. Biol. 197, 179–191 (2004).
Schreiber, H. & Steinhauser, O. Cutoff size does strongly influence molecular dynamics results on solvated polypeptides. Biochemistry 31, 5856–5860 (1992).
Chan, K.W., Sui, J.L., Vivaudou, M. & Logothetis, D.E. Specific regions of heteromeric subunits involved in enhancement of G protein-gated K+ channel activity. J. Biol. Chem. 272, 6548–6555 (1997).
He, C. et al. Identification of critical residues controlling G protein-gated inwardly rectifying K+ channel activity through interactions with the βγ subunits of G proteins. J. Biol. Chem. 277, 6088–6096 (2002).
Hilgemann, D.W. The giant membrane patch, in Single-Channel Recording (eds. Sakmann, B. & Neher, E.) 307–327 (Plenum, New York, 1995).
DeLano, W.L. The PyMOL Molecular Graphics System (DeLano Scientific, San Carlos, California, USA, 2002).
Acknowledgements
We thank E. Findeis, T. Borges, V. Petrou and H. Vaananen for oocyte preparation. This work was supported by a US National Institutes of Health grant (HL-59949) to D.E.L.
Author information
Authors and Affiliations
Contributions
A.R.-D. designed and performed the molecular modeling studies, the mutagenesis and the two-electrode voltage-clamp experiments, wrote the manuscript and revised the manuscript to address the reviewers' comments. J.L.S. (Kir3.4*, Kir2.1), Q.Z. (Kir4.1/Kir5.1) and R.R. (Kir3.1/Kir3.2, Kir4.1) performed the inside-out macropatch recordings. A.A.R.-M. carried out the PtdIns(4,5)P2 dose-response curve and Z.Z. performed the Na+ dose-response curve. D.E.L. initiated and supervised the work, edited the manuscript produced by A.R.-D., and revised the final form of the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Table 1 (PDF 850 kb)
Rights and permissions
About this article
Cite this article
Rosenhouse-Dantsker, A., Sui, J., Zhao, Q. et al. A sodium-mediated structural switch that controls the sensitivity of Kir channels to PtdIns(4,5)P2. Nat Chem Biol 4, 624–631 (2008). https://doi.org/10.1038/nchembio.112
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio.112