Abstract
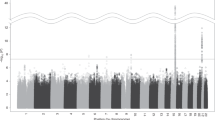
Twin studies document substantial heritability for successful abstinence from smoking. A genome-wide association study has identified markers whose allele frequencies differ with nominal P<0.005 in nicotine-dependent clinical trial participants who were successful vs unsuccessful in abstaining from smoking; many of these results are also supported by data from two additional samples. More study is required to precisely determine the variance in quitting success that can be accounted for by the single-nucleotide polymorphisms that are currently identified and to precisely classify individuals who may display varying degrees of genetic vs environmental effects into quitters or nonquitters. However, the data at hand do allow us to model the effects of genotypic stratification in smoking cessation trials. We identify relationships between the costs of identifying and genotyping prospective trial participants vs the costs of performing the clinical trials. We quantitate the increasing savings that result from genetically stratified designs as recruiting/genotyping costs go down and trial costs increase. This model helps to define the circumstances in which genetically stratified designs may enhance power and reduce costs for smoking cessation clinical trials.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Uhl GR, Elmer GI, Labuda MC, Pickens RW . Genetic influences in drug abuse. In: Gloom FE, Kupfer DJ (eds). Psychopharmacology: The Fourth Generation of Progress. Raven Press: New York, 1995, pp 1793–2783.
Tsuang MT, Lyons MJ, Meyer JM, Doyle T, Eisen SA, Goldberg J et al. Co-occurrence of abuse of different drugs in men: the role of drug-specific and shared vulnerabilities. Arch Gen Psychiatry 1998; 55: 967–972.
Karkowski LM, Prescott CA, Kendler KS . Multivariate assessment of factors influencing illicit substance use in twins from female-female pairs. Am J Med Genet 2000; 96: 665–670.
True WR, Heath AC, Scherrer JF, Xian H, Lin N, Eisen SA et al. Interrelationship of genetic and environmental influences on conduct disorder and alcohol and marijuana dependence symptoms. Am J Med Genet 1999; 88: 391–397.
Xian H, Scherrer JF, Madden PA, Lyons MJ, Tsuang M, True WR et al. The heritability of failed smoking cessation and nicotine withdrawal in twins who smoked and attempted to quit. Nicotine Tob Res 2003; 5: 245–254.
Broms U, Silventoinen K, Madden PA, Heath AC, Kaprio J . Genetic architecture of smoking behavior: a study of Finnish adult twins. Twin Res Hum Genet 2006; 9: 64–72.
Carmelli D, Swan GE, Robinette D, Fabsitz R . Genetic influence on smoking—a study of male twins. N Engl J Med 1992; 327: 829–833.
Morley KI, Lynskey MT, Madden PA, Treloar SA, Heath AC, Martin NG . Exploring the inter-relationship of smoking age-at-onset, cigarette consumption and smoking persistence: genes or environment? Psychol Med 2007; 37: 1357–1367.
Tonnesen P, Norregaard J, Simonsen K, Sawe U . A double-blind trial of a 16-h transdermal nicotine patch in smoking cessation. N Engl J Med 1991; 325: 311–315.
Kenford SL, Fiore MC, Jorenby DE, Smith SS, Wetter D, Baker TB . Predicting smoking cessation. Who will quit with and without the nicotine patch. JAMA 1994; 271: 589–594.
Jones RL, Nguyen A, Man SF . Nicotine and cotinine replacement when nicotine nasal spray is used to quit smoking. Psychopharmacology (Berl) 1998; 137: 345–350.
Uhl GR, Liu QR, Drgon T, Johnson C, Walther D, Rose JE . Molecular genetics of nicotine dependence and abstinence: whole genome association using 520 000 SNPs. BMC Genet 2007; 8: 10.
Uhl GR, Liu QR, Drgon T, Johnson C, Walther D, Rose JE et al. Molecular genetics of successful smoking cessation: convergent genome-wide association study results. Arch Gen Psychiatry 2008; 65: 683–693.
Cornuz J, Gilbert A, Pinget C, McDonald P, Slama K, Salto E et al. Cost-effectiveness of pharmacotherapies for nicotine dependence in primary care settings: a multinational comparison. Tob Control 2006; 15: 152–159.
Hall SM, Lightwood JM, Humfleet GL, Bostrom A, Reus VI, Munoz R . Cost-effectiveness of bupropion, nortriptyline, and psychological intervention in smoking cessation. J Behav Health Serv Res 2005; 32: 381–392.
Hughes JR, Giovino GA, Klevens RM, Fiore MC . Assessing the generalizability of smoking studies. Addiction 1997; 92: 469–472.
Acknowledgements
We acknowledge the support given by the NIH IRP (NIDA), DHSS, unrestricted support for studies of adult smoking cessation to the Duke Center for Nicotine and Smoking Cessation Research from Philip Morris USA Inc. and advice on the article and statistical approaches from Dr Greg Samsa.
Author information
Authors and Affiliations
Corresponding author
Additional information
Duality of interest
None declared.
Rights and permissions
About this article
Cite this article
Uhl, G., Drgon, T., Johnson, C. et al. Nicotine abstinence genotyping: assessing the impact on smoking cessation clinical trials. Pharmacogenomics J 9, 111–115 (2009). https://doi.org/10.1038/tpj.2008.10
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/tpj.2008.10
Keywords
This article is cited by
-
Smoking quit success genotype score predicts quit success and distinct patterns of developmental involvement with common addictive substances
Molecular Psychiatry (2014)
-
Biomarkers for Smoking Cessation
Clinical Pharmacology & Therapeutics (2013)
-
Genetics of dopamine receptors and drug addiction
Human Genetics (2012)
-
Early Human Screening of Medications to Treat Drug Addiction: Novel Paradigms and the Relevance of Pharmacogenetics
Clinical Pharmacology & Therapeutics (2011)