Abstract
Study design:
Retrospective examination.
Objective:
To define the clinical characteristics and response to therapy of spinal multiple sclerosis (MS).
Setting:
Italy.
Methods:
Retrospective review was performed on 563 patients with clinical definite MS. Selection criteria were two or more spinal cord lesions in the presence of normal magnetic resonance imaging of the brain.
Results:
Spinal MS was diagnosed in 13 patients (2.3%) out of 563 with clinical definite MS. There were seven female and six male patients; nine had a relapsing–remitting (RR) and four, a primary progressive (PP) course. All patients were treated with immunosoppressive or immunomodulatory therapy. Mean disease duration in patients with RR–MS was 13.1±10.1 years with a mean age at onset of 29.5±14.3 years; the mean Expanded Disability Status Scale (EDSS) at the time of the study was 3.5±2.5 with a progression index of 0.28. Mean disease duration in patients with PP course was 7±6.2 years with a mean age at onset of 56.7±10.4 years; the mean EDSS at the time of the study was 6.2±2.0 with a progression index of 1.48.
Conclusions:
Patients with spinal RR–MS are characterised by an early disease onset with minimal or moderate disability progression; patients with spinal PP–MS show a late disease onset and more rapid disability progression. In our series of spinal MS patients, disability progression seems to be mainly due to the disease course and age at onset rather than to the site of lesion.
Similar content being viewed by others
Introduction
Multiple sclerosis (MS) is a chronic inflammatory disease of the central nervous system (CNS) characterised by white matter hyperintense lesions on magnetic resonance imaging (MRI) T2-weighted images localised either in the brain and the spinal cord, or in the brain only, or, in a few patients, in the spinal cord only. Exclusive spinal cord involvement is reported in 2–10% of patients with MS.1, 2
The importance of spinal cord lesions involving clinically eloquent pathways (pyramidal tracts, spinothalamic tracts and posterior columns) lies in their presumed greater potential to result in clinical symptoms and disability than brain white matter lesions.3
The aim of this study was to analyse the clinical and radiological characteristics of patients with clinical definite MS with typical lesions visible only in spinal cord MRI.
Patients and methods
Patients were selected from a total of 563, with clinical definite MS according to McDonald's guidelines.4 Selection criteria were two or more spinal cord lesions in the presence of normal brain MRI. Each patient underwent two or more serial MR imaging of the brain and spinal cord. Individual case reports include clinical, cerebro-spinal fluid (presence of oligoclonal banding) electrophysiological (visual, motor and sensory-evoked potentials) and neuroimaging (number and extension of spinal cord lesions, presence of gadolinium enhancement) data and treatment.
Disease course was classified as: relapsing–remitting (RR), when there was a history of relapses and remissions without progressive deterioration; primary progressive (PP), when there was progressive deterioration from onset with no relapses or remissions; secondary progressive, when, after an initial RR course, there was progressive deterioration, with or without superimposed relapses. Disability was graded according to Kurtzke's Expanded Disability Status Scale (EDSS).5 Brain and spinal imaging was performed in all cases, every year. MRI data were acquired with high resolution 1.5 Tesla system (5 mm slice thickness). Scanning sessions included proton density (echo time (TE) 20/repetition time (TR) 2500), T1-weighted (TE 17/TR 600) and T2-weighted (TE 80/TR 2500) images, fluid attenuated inversion recovery (FLAIR) sequence. The T1-weighted images were acquired before and 10 min after an intravenous injection of gadolinium-diethylenetriaminepentaacetic acid (0.1 mmol/kg) (Gd-DTPA). FLAIR imaging was not acquired in spinal cord because it was not sensitive to spinal MS lesions.6 The EDSS score was assessed at each visit, and the annual mean increase in disability or progression index (EDSS/disease duration) was calculated.
Results
In total, 13 patients (2.3%) fulfilled the inclusion criteria. Seven of them were females and six males, with age at onset being from 12 to 71 years (mean 37.9±18.3). Nine patients had a RR and four a PP course.
Motor symptoms were the most common neurological presentation at onset (76.9%).
Oligoclonal bands (OCB) were found in the CSF in nine, out of the 13 patients (all the ones with PP course had OCB). Two had abnormal visual evoked potentials, 11 had abnormal motor evoked potentials, 10 had abnormal sensory evoked potentials.
In patients with RR course, disease duration ranged from 2 to 34 years (mean 13.1±10.1) with age at onset being from 12 to 54 years (mean 29.5±14.3). Disability at onset varied from EDSS 1 to EDSS 6.5 (mean EDSS 3). All the patients were treated with intravenous metilprednisolone (20 mg/kg/day) for 5 days at relapses. Five patients were also treated with immunomodulatory drugs, three with IFN-beta (IFN-beta 1a, 44 μg three times a week subcutaneously), one with methotrexate (2.5 mg three times a week per os) and one with azathioprine (2.5 mg/kg/day). One patient showed a secondary progressive course after 21 years from disease onset and was treated with mitoxantrone (12 mg/m2 every 3 months). All RR–MS patients showed minimal or moderate disability progression (mean EDSS at present 3.5): the mean progression index was 0.28.
In patients with PP course, disease duration ranged from 4 to 16 years (mean 7±6.2) with age at onset being from 48 to 71 years (mean 56.7±10.4). All four patients were treated with azathioprine with no beneficial effects. They showed a severe disability progression (mean EDSS at time of the study 6.2): the mean progression index was 1.48.
In all patients, follow-up MR examinations showed normal brain and one or more new hyperintense lesions on T2-weighted images in the spinal cord, clearly demonstrating a dissemination in time and space. All lesions were localised in lateral and posterior white-matter columns, mainly in the cervical tract. These lesions typically occupied less than 50% of the cross-sectional area of the cord and did not usually cause focal swelling. All PP–MS patients and the one with SP–MS showed spinal cord atrophy.
The clinical and MRI details are summarised in Table 1 (see Figure 1).
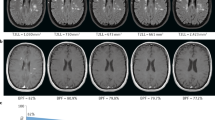
Sagittal FSE T2-W MRI image of the cervical and thoracic spinal cord and axial FSE T2-W images of the brain corresponding to case 4 in the table. Three ill defined areas of high signal are seen within the spinal cord at C2–C3, C7 (a), D10–D11 levels (b). No abnormalities are seen in the white matter of both cerebral hemispheres (c)
Discussion
MS is a disseminated disease of the CNS rarely affecting spinal cord only. In agreement with other authors,1, 2 we found that only 2.3% of our patients with clinical definite MS had two or more spinal cord lesions concomitantly with normal brain MRI. The age range of disease onset was similar to classical MS forms although there was no preponderance of female patients. Most of the younger patients showed a RR course of disease, while a primary progressive course was observed in older ones.
In accordance with previous studies,7, 8 the most common finding in our series consisted of two or more spinal lesions (max five lesions) with typical localisation and size. Like other authors,9, 10 we found that spinal cord atrophy was present mainly in patients with SP–MS and PP–MS.
The patients with a RR course showed a low progression index (0.28). The patients with PP course had a more disability progression (progression index 1.48) than RR patients. Some authors11 found that higher levels of disability (measured by EDSS) were present in patients with spinal cord atrophy, compared with patients without atrophy. Atrophy and progression in our PP patients may reflect the extent and severity of the demyelinating lesions. However, it cannot be excluded the presence of diffuse damage in normal-appearing tissue undetectable by conventional MRI.12 In fact, in cervical spinal cord, changes in quantitative MR imaging parameters seem to be mainly determined by demyelination12 while MR spectroscopy can provide more information on axonal loss within normal-appearing white matter.13 Nevertheless, a more widespread damage in normal-appearing tissue may also be present in the brain of spinal MS patients. Thus, further studies with nonconventional MRI techniques are needed for this group of patients.
References
Thorpe JW et al. Spinal MRI in patients with suspected multiple sclerosis and negative brain MRI. Brain 1996; 119: 709–714.
Trop I et al. Multiple sclerosis of the spinal cord: diagnosis and follow-up with contrast-enhanced MR and correlation with clinical activity. Am J Neuroradiol 1998; 19: 1025–1033.
O’Riordan JI et al. Asymptomatic spinal cord lesions in clinically isolated optic nerve, brain stem, and spinal cord syndrome suggestive of demyelination. J Neurol Neurosurg Psychiatry 1998; 64: 353–357.
McDonald WI et al. Recommended diagnostic criteria for multiple sclerosis: guidelines from the International Panel on the diagnosis of multiple sclerosis. Ann Neurol 2000; 50: 121–127.
Kurtzke JF . Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 1983; 33: 1444–1452.
Keiper MD, Grossman RI, Brunson JC, Schnall MD . The low sensitivity of fluid-attenuated inversion-recovery MR in the detection of multiple sclerosis of the spinal cord. Am J Neuroradiol 1997; 18: 1035–1039.
Lycklama G et al. Spinal-cord MRI in multiple sclerosis. Lancet Neurol 2003; 2: 555–562.
Tartaglino LM, Friedman DP, Flanders AE, Lublin FD, Knobler RL, Liem M . Multiple sclerosis in the spinal cord: MR appearance and correlation with clinical parameters. Radiology 1995; 19: 725–732.
Stevenson VL et al. Primary and transitional progressive MS: a clinical and MRI cross-sectional study. Neurology 1999; 52: 839–845.
Stevenson VL et al. One year follow-up study of primary and transitional progressive multiple sclerosis. J Neurol Neurosurg Psychiatry 2000; 68: 713–718.
Zivadinov R, Bakshi R . Central nervous system atrophy and clinical status in multiple sclerosis. J Neuroimaging 2004; 14: 27S–35S.
Bot JC et al. The spinal cord in multiple sclerosis: relationship of high-spatial-quantitative MR imaging findings to histopathologic results. Radiology 2004; 233: 531–540.
Kendi AT, Tan FU, Kendi M, Yilmaz S, Huvaj S, Tellioglu S . MR spectroscopy of cervical spinal cord in patients with multiple sclerosis. Neuroradiology 2004; 16: 1270–1277.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nociti, V., Cianfoni, A., Mirabella, M. et al. Clinical characteristics, course and prognosis of spinal multiple sclerosis. Spinal Cord 43, 731–734 (2005). https://doi.org/10.1038/sj.sc.3101798
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.sc.3101798
Keywords
This article is cited by
-
An IFNγ/CXCL2 regulatory pathway determines lesion localization during EAE
Journal of Neuroinflammation (2018)
-
Progressive ascending myelopathy: atypical forms of multiple sclerosis or what else?
Journal of Neurology (2011)
-
Comparison of LEP and QST and their contribution to standard sensory diagnostic assessment of spinal lesions: a pilot study
Neurological Sciences (2011)