Abstract
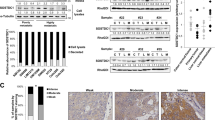
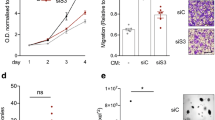
S100A4 is implicated in metastasis and chronic inflammation, but its function remains uncertain. Here we establish an S100A4-dependent link between inflammation and metastatic tumor progression. We found that the acute-phase response proteins serum amyloid A (SAA) 1 and SAA3 are transcriptional targets of S100A4 via Toll-like receptor 4 (TLR4)/nuclear factor-κB signaling. SAA proteins stimulated the transcription of RANTES (regulated upon activation normal T-cell expressed and presumably secreted), G-CSF (granulocyte-colony-stimulating factor) and MMP2 (matrix metalloproteinase 2), MMP3, MMP9 and MMP13. We have also shown for the first time that SAA stimulate their own transcription as well as that of proinflammatory S100A8 and S100A9 proteins. Moreover, they strongly enhanced tumor cell adhesion to fibronectin, and stimulated migration and invasion of human and mouse tumor cells. Intravenously injected S100A4 protein induced expression of SAA proteins and cytokines in an organ-specific manner. In a breast cancer animal model, ectopic expression of SAA1 or SAA3 in tumor cells potently promoted widespread metastasis formation accompanied by a massive infiltration of immune cells. Furthermore, coordinate expression of S100A4 and SAA in tumor samples from colorectal carcinoma patients significantly correlated with reduced overall survival. These data show that SAA proteins are effectors for the metastasis-promoting functions of S100A4, and serve as a link between inflammation and tumor progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Langley RR, Fidler IJ . The seed and soil hypothesis revisited—the role of tumor–stroma interactions in metastasis to different organs. Int J Cancer 2011; 128: 2527–2535.
DeNardo DG, Johansson M, Coussens LM . Immune cells as mediators of solid tumor metastasis. Cancer Metast Rev 2008; 27: 11–18.
Fridman WH, Galon J, Pagès F, Tartour E, Sautès-Fridman C, Kroemer G . Prognostic and predictive impact of intra- and peritumoral immune infiltrates. Cancer Res 2011; 71: 5601–5605.
Malle E, Sodin-Semrl S, Kovacevic A . Serum amyloid A: an acute-phase protein involved in tumour pathogenesis. Cell Mol Life Sci 2009; 66: 9–26.
Larson MA, Wei SH, Weber A, Weber AT, McDonald TL . Induction of human mammary-associated serum amyloid A3 expression by prolactin or lipopolysaccharide. Biochem Biophys Res Commun 2003; 301: 1030–1037.
Furlaneto CJ, Campa A . A novel function of serum amyloid A: a potent stimulus for the release of tumor necrosis factor-alpha, interleukin-1beta, and interleukin-8 by human blood neutrophil. Biochem Biophys Res Commun 2000; 268: 405–408.
He R, Shepard LW, Chen J, Pan ZK, Ye RD . Serum amyloid A is an endogenous ligand that differentially induces IL-12 and IL-23. J Immunol 2006; 177: 4072–4079.
Song C, Hsu K, Yamen E, Yan W, Fock J, Witting PK et al. Serum amyloid A induction of cytokines in monocytes/macrophages and lymphocytes. Atherosclerosis 2009; 207: 374–383.
He RL, Zhou J, Hanson CZ, Chen J, Cheng N, Ye RD . Serum amyloid A induces G-CSF expression and neutrophilia via Toll-like receptor 2. Blood 2009; 113: 429–437.
Yamada T . Serum amyloid A (SAA): a concise review of biology, assay methods and clinical usefulness. Clin Chem Lab Med 1999; 37: 381–388.
Lee HY, Kim MK, Park KS, Bae YH, Yun J, Park JI et al. Serum amyloid A stimulates matrix metalloproteinase-9 upregulation via formyl peptide receptor like-1-mediated signaling in human monocytic cells. Biochem Biophys Res Commun 2005; 330: 989–998.
Zhao Y, Zhou S, Heng CK . Celecoxib inhibits serum amyloid a-induced matrix metalloproteinase-10 expression in human endothelial cells. J Vasc Res 2009; 46: 64–72.
Okamoto H, Katagiri Y, Kiire A, Momohara S, Kamatani N . Serum amyloid A activates nuclear factor-kappaB in rheumatoid synovial fibroblasts through binding to receptor of advanced glycation endproducts. J Rheumatol 2008; 35: 752–756.
Hiratsuka S, Watanabe A, Sakurai Y, Akashi-Takamura S, Ishibashi S, Miyake K et al. The S100A8-serum amyloid A3-TLR4 paracrine cascade establishes a pre-metastatic phase. Nat Cell Biol 2008; 10: 1349–1355.
Kaplan RN, Riba RD, Zacharoulis S, Bramley AH, Vincent L, Costa C et al. VEGFR1-positive haematopoietic bone marrow progenitors initiate the pre-metastatic niche. Nature 2005; 438: 820–827.
Grigorian M, Ambartsumian N, Lukanidin E . Metastasis-inducing S100A4 protein: implication in non-malignant human pathologies. Curr Mol Med 2008; 8: 492–496.
Zibert JR, Skov L, Thyssen JP, Jacobsen GK, Grigorian M . Significance of the S100A4 protein in psoriasis. J Invest Dermatol 2010; 130: 150–160.
Cerezo LA, Kuncová K, Mann H, Tomcík M, Zámecník J, Lukanidin E et al. The metastasis promoting protein S100A4 is increased in idiopathic inflammatory myopathies. Rheumatology (Oxford) 2011; 50: 1766–1772.
Marenholz I, Heizmann CW, Fritz G . S100 proteins in mouse and man: from evolution to function and pathology (including an update of the nomenclature). Biochem Biophys Res Commun 2004; 322: 1111–1122.
Fritz G, Botelho HM, Morozova-Roche LA, Gomes CM . Natural and amyloid self-assembly of S100 proteins: structural basis of functional diversity. FEBS J 2010; 277: 4578–4590.
Ebralidze A, Tulchinsky E, Grigorian M, Afanasyeva A, Senin V, Revazova E et al. Isolation and characterization of a gene specifically expressed in different metastatic cells and whose deduced gene product has a high degree of homology to a Ca2þ-binding protein family. Genes Dev 1989; 3: 1086–1093.
Grigorian M, Ambartsumian N, Lykkesfeldt AE, Bastholm L, Elling F, Georgiev G et al. Effect of mts1 (S100A4) expression on the progression of human breast cancer cells. Int J Cancer 1996; 67: 831–841.
Maelandsmo GM, Hovig E, Skrede M, Engebraaten O, Flørenes VA, Myklebost O et al. Reversal of the in vivo metastatic phenotype of human tumor cells by an anti-CAPL (mts1) ribozyme. Cancer Res 1996; 56: 5490–5498.
Ambartsumian NS, Grigorian MS, Larsen IF, Karlstrøm O, Sidenius N, Rygaard J et al. Metastasis of mammary carcinomas in GRS/A hybrid mice transgenic for the mts1 gene. Oncogene 1996; 13: 1621–1630.
Davies MP, Rudland PS, Robertson L, Parry EW, Jolicoeur P, Barraclough R . Expression of the calcium-binding protein S100A4 (p9Ka) in MMTV-neu transgenic mice induces metastasis of mammary tumours. Oncogene 1996; 13: 1631–1637.
Schmidt-Hansen B, Klingelhöfer J, Grum-Schwensen B, Christensen A, Andresen S, Kruse C et al. Functional significance of metastasis-inducing S100A4 (Mts1) in tumor–stroma interplay. J Biol Chem 2004; 279: 24498–24504.
Helfman DM, Kim E, Lukanidin E, Grigorian M . The metastasis associated protein S100A4. Br J Cancer 2005; 92: 1955–1958.
Grum-Schwensen B, Klingelhöfer J, Grigorian M, Almholt K, Nielsen BS, Lukanidin E et al. Lung metastasis fails in MMTV-PyMT oncomice lacking S100A4 due to a T-cell deficiency in primary tumors. Cancer Res 2010; 70: 936–947.
Klingelhöfer J, Grum-Schwensen B, Beck MK, Knudsen RS, Grigorian M, Lukanidin E et al. Anti-S100A4 antibody suppresses metastasis formation by blocking stroma cell invasion. Neoplasia 2012; 14: 1260–1268.
Lin Y, Rajala MW, Berger JP, Moller DE, Barzilai N, Scherer PE . Hyperglycemia-induced production of acute phase reactants in adipose tissue. J Biol Chem 2001; 276: 42077–42083.
Ehrchen JM, Sunderkötter C, Foell D, Vogl T, Roth J . The endogenous Toll-like receptor 4 agonist S100A8/S100A9 (calprotectin) as innate amplifier of infection, autoimmunity, and cancer. J Leukoc Biol 2009; 86: 557–566.
O'Neill LA . Signal transduction pathways activated by the IL-1 receptor/toll-like receptor superfamily. Curr Top Microbiol Immunol 2002; 270: 47–61.
Klingelhöfer J, Møller HD, Sumer EU, Berg CH, Ambartsumian N, Grigorian M et al. EGFR ligands as new extracellular targets for the metastasis-promoting S100A4 protein. FEBS J 2009; 276: 5936–5948.
Lukanidin E, Sleeman JP . Building the niche: The role of the S100 proteins in metastatic growth. Semin Cancer Biol 2012; 22: 216–225.
Gregory AD, Houghton AM . Tumor-associated neutrophils: new targets for cancer therapy. Cancer Res 2011; 71: 2411–2416.
Schmid MC, Varner JA . Myeloid cells in the tumor microenvironment: modulation of tumor angiogenesis and tumor inflammation. J Oncol 2010; 2010: 201026.
Tan W, Zhang W, Strasner A, Grivennikov S, Cheng JQ, Hoffman RM et al. Tumour-infiltrating regulatory T cells stimulate mammary cancer metastasis through RANKL-RANK signalling. Nature 2011; 470: 548–553.
Aguirre-Ghiso JA, Liu D, Mignatti A, Kovalski K, Ossowski L . Urokinase receptor and fibronectin regulate the ERK(MAPK) to p38(MAPK) activity ratios that determine carcinoma cell proliferation or dormancy in vivo. Mol Biol Cell 2001; 12: 863–879.
Cabezón T, Celis JE, Skibshøj I, Klingelhöfer J, Grigorian M, Gromov P et al. Expression of S100A4 by a variety of cell types present in the tumor microenvironment of human breast cancer. Int J Cancer 2007; 121: 1433–1444.
Klingelhofer J, Šenolt L, Baslund B, Nielsen GH, Skibshøj I, Pavelka K et al. Up-regulation of metastasis-promoting S100A4 (Mts1) in rheumatoid arthritis: putative involvement in the pathogenesis of RA. Arthritis Rheum 2007; 56: 779–789.
Sung HJ, Ahn JM, Yoon YH, Rhim TY, Park CS, Park JY et al. Identification and validation of SAA as a potential lung cancer biomarker and its involvement in metastatic pathogenesis of lung cancer. J Proteome Res 2011; 10: 1383–1395.
Cunnane G . Amyloid precursors and amyloidosis in inflammatory arthritis. Curr Opin Rheumatol 2001; 13: 67–73.
Mullan RH, Bresnihan B, Golden-Mason L, Markham T, O'Hara R, FitzGerald O et al. Acute-phase serum amyloid A stimulation of angiogenesis, leukocyte recruitment, and matrix degradation in rheumatoid arthritis through an NF-kappaB-dependent signal transduction pathway. Arthritis Rheum 2006; 54: 105–114.
Grigorian M, Tulchinsky E, Burrone O, Tarabykina S, Georgiev G, Lukanidin E . Modulation of mts1 expression in mouse and human normal and tumor cells. Electrophoresis 1994; 15: 463–468.
Pear W, Nolan GP, Scott ML, Baltimore D . Production of high titer helper-free retroviruses by transient transfection. Proc Natl Acad Sci USA 1993; 90: 8392–8396.
Møller HD, Ralfkjær U, Cremers N, Frankel M, Pedersen RT, Klingelhöfer J et al. Role of fibulin-5 in metastatic organ colonization. Mol Cancer Res 2011; 9: 553–563.
Novitskaya V, Grigorian M, Kriajevska M, Tarabykina S, Bronstein I, Berezin V et al. Oligomeric forms of the metastasis-related Mts1 (S100A4) protein stimulate neuronal differentiation in cultures of rat hippocampal neurons. J Biol Chem 2000; 275: 41278–41286.
Bang CA, Bro S, Bartels ED, Pedersen TX, Nielsen LB. . Effect of uremia on HDL composition, vascular inflammation, and atherosclerosis in wild-type mice. Am J Physiol Renal Physiol 2007; 293: 1325–1331.
Baumann P, Cremers N, Kroese F, Orend G, Chiquet-Ehrismann R, Uede T et al. CD24 expression causes the acquisition of multiple cellular properties associated with tumor growth and metastasis. Cancer Res 2005; 65: 10783–10793.
Forst B, Hansen MT, Klingelhöfer J, Møller HD, Nielsen GH, Grum-Schwensen B et al. Metastasis-inducing S100A4 and RANTES cooperate in promoting tumor progression in mice. PLoS One 2010; 5: e10374.
Ambartsumian N, Christensen CRL, Lukanidin E . Endothelial cell invasion assay. Cell Biology, Vol. 1.: Academic Press: Waltham, MA, USA 2005, pp 363–366.
Schmidt-Hansen B, Klingelhöfer J, Grum-Schwensen B, Christensen A, Andresen S, Kruse C et al. Functional significance of metastasis-inducing S100A4(Mts1) in tumor–stroma interplay. J Biol Chem 2004; 279: 24498–24504.
Stein U, Walther W, Arlt F, Schwabe H, Smith J, Fichtner I et al. MACC1, a newly identified key regulator of HGF-MET signaling, predicts colon cancer metastasis. Nat Med 2009; 15: 59–67.
Acknowledgements
We thank Ekaterina Dulina, Inge Skibshøj and Lene Bregnholt Larsen for careful technical assistance. We gratefully acknowledge funding by the European Union (TuMIC, Health-F2-2008-201662) and INARMERA (FP7-INCO-2010-6), the Danish Cancer Society and the Dansk Kræftforsknings Fond.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Hansen, M., Forst, B., Cremers, N. et al. A link between inflammation and metastasis: serum amyloid A1 and A3 induce metastasis, and are targets of metastasis-inducing S100A4. Oncogene 34, 424–435 (2015). https://doi.org/10.1038/onc.2013.568
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2013.568
Keywords
This article is cited by
-
Claudin-18.2 mediated interaction of gastric Cancer cells and Cancer-associated fibroblasts drives tumor progression
Cell Communication and Signaling (2024)
-
Hepatocyte SGK1 activated by hepatic ischemia-reperfusion promotes the recurrence of liver metastasis via IL-6/STAT3
Journal of Translational Medicine (2023)
-
Pulmonary interleukin 1 beta/serum amyloid A3 axis promotes lung metastasis of hepatocellular carcinoma by facilitating the pre-metastatic niche formation
Journal of Experimental & Clinical Cancer Research (2023)
-
Sex differences in renal cell carcinoma: a single-cell analysis reveals exhausted CD8+ T-cells highly infiltrated in males
Biology of Sex Differences (2023)
-
Acute-phase serum amyloid A for early detection of hepatocellular carcinoma in cirrhotic patients with low AFP level
Scientific Reports (2022)