Abstract
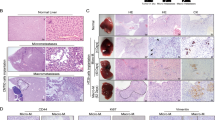
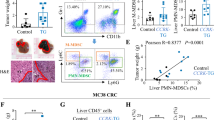
Liver metastasis is the predominant cause of colorectal cancer (CRC)-related mortality in developed countries. Carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM1) is a cell adhesion molecule with reduced expression in early phases of CRC development and thus functions as a tumor growth inhibitor. However, CEACAM1 is upregulated in metastatic colon cancer, suggesting a bimodal role in CRC progression. To investigate the role of this protein in the host metastatic environment, Ceacam1−/− mice were injected intrasplenically with metastatic MC38 mouse CRC cells. A significant reduction in metastatic burden was observed in Ceacam1−/− compared with wild-type (WT) livers. Intravital microscopy showed decreased early survival of MC38 cells in Ceacam1−/− endothelial environment. Metastatic cell proliferation within the Ceacam1−/− livers was also diminished. Bone marrow-derived cell recruitment, attenuation of immune infiltrates and diminished CCL2, CCL3 and CCL5 chemokine production participated in the reduced Ceacam1−/− metastatic phenotype. Transplantations of WT bone marrow (BM) into Ceacam1−/− mice fully rescued metastatic development, whereas Ceacam1−/− BM transfer into WT mice showed reduced metastatic burden. Chimeric immune cell profiling revealed diminished recruitment of CD11b+Gr1+ myeloid-derived suppressor cells (MDSCs) to Ceacam1−/− metastatic livers and adoptive transfer of MDSCs confirmed the involvement of these immune cells in reduction of liver metastasis. CEACAM1 may represent a novel metastatic CRC target for treatment.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bamberger AM, Riethdorf L, Nollau P, Naumann M, Erdmann I, Gotze J et al. (1998). Dysregulated expression of CD66a (BGP, C-CAM), an adhesion molecule of the CEA family, in endometrial cancer. Am J Pathol 152: 1401–1406.
Beauchemin N, Draber P, Dveksler G, Gold P, Gray-Owen S, Grunert F et al. (1999). Redefined nomenclature for members of the carcinoembryonic antigen family. Exp Cell Res 252: 243–249.
Busch C, Hanssen TA, Wagener C, Obrink B . (2002). Down-regulation of CEACAM1 in human prostate cancer: correlation with loss of cell polarity, increased proliferation rate, and Gleason grade 3 to 4 transition. Hum Pathol 33: 290–298.
Chen T, Zimmermann W, Parker J, Chen I, Maeda A, Bolland S . (2001). Biliary glycoprotein (BGPa, CD66a, CEACAM1) mediates inhibitory signals. J Leukoc Biol 70: 335–340.
Chen Z, Chen L, Baker K, Olszak T, Zeissig S, Huang YH et al. (2011). CEACAM1 dampens anti-tumor immunity by downregulation of NKG2D ligand expression on tumor cells and NKG2D function on NK cells. J Exp Med 208: 2633–2640.
Dango S, Sienel W, Schreiber M, Stremmel C, Kirschbaum A, Pantel K et al. (2008). Elevated expression of carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM-1) is associated with increased angiogenic potential in non-small-cell lung cancer. Lung Cancer 60: 426–433.
Ebrahimnejad A, Streichert T, Nollau P, Horst AK, Wagener C, Bamberger AM et al. (2004). CEACAM1 enhances invasion and migration of melanocytic and melanoma cells. Am J Pathol 165: 1781–1787.
Gabrilovich DI, Nagaraj S . (2009). Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol 9: 162–174.
Giavazzi R, Campbell DE, Jessup JM, Cleary K, Fidler IJ . (1986). Metastatic behavior of tumor cells isolated from primary and metastatic human colorectal carcinomas implanted into different sites in nude mice. Cancer Res 46: 1928–1933.
Gout S, Huot J . (2008). Role of cancer microenvironment in metastasis: focus on colon cancer. Cancer Microenviron 1: 69–83.
Gray-Owen SD, Blumberg RS . (2006). CEACAM1: contact-dependent control of immunity. Nat Rev Immunol 6: 433–446.
Hanahan D, Weinberg RA . (2011). Hallmarks of cancer: the next generation. Cell 144: 646–674.
Hemmila E, Turbide C, Olson M, Jothy S, Holmes KV, Beauchemin N . (2004). Ceacam1a-/- mice are completely resistant to infection by murine coronavirus MHV-A59. J Virol 78: 10156–10165.
Horst A, Bickert T, Brewig N, Schumacher U, Beauchemin N, Fleischer B et al. (2009). CEACAM1 expression on macrophage precursors affects early lymphangiogenesis in vivo. Blood 113: 6726–6736.
Horst AK, Wulf DI, Dabelstein J, Schumacher U, Sander H, Turbide C et al. (2006). Carcinoembryonic antigen-related cell adhesion molecule 1 modulates vascular remodeling in vitro and in vivo. J Clin Investig 116: 1596–1605.
Ieda J, Yokoyama S, Tamura K, Takifuji K, Hotta T, Matsuda K et al. (2011). Re-expression of CEACAM1 long cytoplasmic domain isoform is associated with invasion and migration of colorectal cancer. Int J Cancer 129: 1351–1361.
Kang WY, Chen WT, Wu MT, Chai CY . (2007). Expression of CD66a and possible roles in colorectal adenoma and adenocarcinoma. Int J Colorectal Dis 22: 869–874.
Kaplan RN, Rafii S, Lyden D . (2006). Preparing the “soil”: the premetastatic niche. Cancer Res 66: 11089–11093.
Kirshner J, Chen CJ, Liu P, Huang J, Shively JE . (2003). CEACAM1-4S, a cell-cell adhesion molecule, mediates apoptosis and reverts mammary carcinoma cells to a normal morphogenic phenotype in a 3D culture. Proc Natl Acad Sci USA 100: 521–526.
Laurie NA, Comegys MM, Carreiro MP, Brown JF, Flanagan DL, Brilliant KE et al. (2005). Carcinoembryonic antigen-related cell adhesion molecule 1a-4L suppression of rat hepatocellular carcinomas. Cancer Res 65: 11010–11017.
Leung N, Turbide C, Balachandra B, Marcus V, Beauchemin N . (2008). Intestinal tumor progression is promoted by decreased apoptosis and dysregulated Wnt signaling in Ceacam1(-/-) mice. Oncogene 27: 4943–4953.
Leung N, Turbide C, Marcus V, Jothy S, Beauchemin N . (2006). Carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM1) contributes to progression of colon tumors. Oncogene 25: 5527–5536.
Liu W, Wei W, Winer D, Bamberger AM, Bamberger C, Wagener C et al. (2007). CEACAM1 impedes thyroid cancer growth but promotes invasiveness: a putative mechanism for early metastases. Oncogene 26: 2747–2758.
Loberg RD, Ying C, Craig M, Day LL, Sargent E, Neeley C et al. (2007). Targeting CCL2 with systemic delivery of neutralizing antibodies induces prostate cancer tumor regression in vivo. Cancer Res 67: 9417–9424.
Ma G, Pan PY, Eisenstein S, Divino CM, Lowell CA, Takai T et al. (2011). Paired-immunoglobulin-like receptor-B regulates the suppressive function and fate of myeloid-derived suppressor cells. Immunity 34: 385–395.
Movahedi K, Guilliams M, Van den Bossche J, Van den Bergh R, Gysemans C, Beschin A et al. (2008). Identification of discrete tumor-induced myeloid-derived suppressor cell subpopulations with distinct T cell-suppressive activity. Blood 111: 4233–4244.
Nagaishi T, Pao L, Lin SH, Iijima H, Kaser A, Qiao SW et al. (2006). SHP1 phosphatase-dependent T cell inhibition by CEACAM1 adhesion molecule isoforms. Immunity 25: 769–781.
Neumaier M, Paululat S, Chan A, Matthaes P, Wagener C . (1993). Biliary glycoprotein, a potential human cell adhesion molecule, is down-regulated in colorectal carcinomas. Proc Natl Acad Sci USA 90: 10744–10748.
Nittka S, Gunther J, Ebisch C, Erbersdobler A, Neumaier M . (2004). The human tumor suppressor CEACAM1 modulates apoptosis and is implicated in early colorectal tumorigenesis. Oncogene 23: 9306–9313.
Niwa Y, Akamatsu H, Niwa H, Sumi H, Ozaki Y, Abe A . (2001). Correlation of tissue and plasma RANTES levels with disease course in patients with breast or cervical cancer. Clin Cancer Res 7: 285–289.
Nollau P, Prall F, Helmchen U, Wagener C, Neumaier M . (1997). Dysregulation of carcinoembryonic antigen group members CGM2, CD66a (biliary glycoprotein), and nonspecific cross-reacting antigen in colorectal carcinomas. Comparative analysis by northern blot and in situ hybridization. Am J Pathol 151: 521–530.
Nouvion AL, Oubaha M, LeBlanc S, Davis EC, Jastrow H, Kammerer R et al. (2010). CEACAM1: a key regulator of vascular permeability. J Cell Sci 123: 4221–4230.
Obrink B . (2008). On the role of CEACAM1 in cancer. Lung Cancer 60: 309–312.
Oliveira-Ferrer L, Tilki D, Ziegeler G, Hauschild J, Loges S, Irmak S et al. (2004). Dual role of carcinoembryonic antigen-related cell adhesion molecule 1 in angiogenesis and invasion of human urinary bladder cancer. Cancer Res 64: 8932–8938.
Pan H, Shively JE . (2010). Carcinoembryonic antigen-related cell adhesion molecule-1 regulates granulopoiesis by inhibition of granulocyte colony-stimulating factor receptor. Immunity 33: 620–631.
Popivanova BK, Kostadinova FI, Furuichi K, Shamekh MM, Kondo T, Wada T et al. (2009). Blockade of a chemokine, CCL2, reduces chronic colitis-associated carcinogenesis in mice. Cancer Res 69: 7884–7892.
Poy MN, Yang Y, Rezaei K, Fernstrom MA, Lee AD, Kido Y et al. (2002). CEACAM1 regulates insulin clearance in liver. Nat Genet 30: 270–276.
Qian BZ, Li J, Zhang H, Kitamura T, Zhang J, Campion LR et al. (2011). CCL2 recruits inflammatory monocytes to facilitate breast-tumour metastasis. Nature 475: 222–225.
Rosenberg M, Nédellec P, Jothy S, Fleiszer D, Turbide C, Beauchemin N . (1993). The expression of mouse biliary glycoprotein, a carcinoembryonic antigen-related gene, is down-regulated in malignant mouse tissues. Cancer Res 53: 4938–4945.
Serra S, Asa SL, Bamberger AM, Wagener C, Chetty R . (2009). CEACAM1 expression in pancreatic endocrine tumors. Appl Immunohistochem Mol Morphol 17: 286–293.
Skalli O, Pelte MF, Peclet MC, Gabbiani G, Gugliotta P, Bussolati G et al. (1989). Alpha-smooth muscle actin, a differentiation marker of smooth muscle cells, is present in microfilamentous bundles of pericytes. J Histochem Cytochem 37: 315–321.
Song JH, Cao Z, Yoon JH, Nam SW, Kim SY, Lee JY et al. (2011). Genetic alterations and expression pattern of CEACAM1 in colorectal adenomas and cancers. Pathol Oncol Res 17: 67–74.
Starke RD, Ferraro F, Paschalaki KE, Dryden NH, McKinnon TA, Sutton RE et al. (2011). Endothelial von Willebrand factor regulates angiogenesis. Blood 117: 1071–1080.
Wu Y, Brodt P, Sun H, Mejia W, Novosyadlyy R, Nunez N et al. (2010). Insulin-like growth factor-I regulates the liver microenvironment in obese mice and promotes liver metastasis. Cancer Res 70: 57–67.
Xu E, Leung N, Dubois MJ, Charbonneau A, Streichert T, Elcheby M et al. (2009). Targeted disruption of Ceacam1 promotes diet-induced hepatic steatosis and insulin resistance. Endocrinol 150: 3503–3512.
Yeatman TJ, Mao W, Karl RC . (1997). Biliary glycoprotein is overexpressed in human colon cancer cells with high metastatic potential. J Gastrointest Surg 1: 292–298.
Yoshidome H, Kohno H, Shida T, Kimura F, Shimizu H, Ohtsuka M et al. (2009). Significance of monocyte chemoattractant protein-1 in angiogenesis and survival in colorectal liver metastases. Int J Oncol 34: 923–930.
Zhou CJ, Liu B, Zhu KK, Zhang QH, Zhang TG, Xu WH et al. (2009). The different expression of carcinoembryonic antigen-related cell adhesion molecule 1 (CEACAM1) and possible roles in gastric carcinomas. Pathol Res Pract 205: 483–489.
Acknowledgements
We wish to thank Drs Janusz Rak, Peter Siegel and Julie St-Pierre (McGill University) for their helpful corrections and comments on the manuscript. We are also indebted to Dr Russell Jones (McGill University) for help with bone marrow transplantations and Dr Kathryn V Holmes (University of Colorado) for the monoclonal CC1 antibody. This work was supported by grants from the Canadian Institutes for Health Research (NB, LF and PB) and the Canadian Cancer Society Research Institute (LF). AA and ALN were recipients of post-doctoral fellowships from the Fonds de la Recherche en Santé du Québec (FRSQ). AA and CC received post-doctoral awards from the McGill Integrated Cancer Research Training Program funded by the Canadian Institutes for Health Research and the Fonds de la Recherche en Santé du Québec (FRSQ).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Arabzadeh, A., Chan, C., Nouvion, AL. et al. Host-related carcinoembryonic antigen cell adhesion molecule 1 promotes metastasis of colorectal cancer. Oncogene 32, 849–860 (2013). https://doi.org/10.1038/onc.2012.112
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2012.112
Keywords
This article is cited by
-
Roles of CEACAM1 in cell communication and signaling of lung cancer and other diseases
Cancer and Metastasis Reviews (2015)
-
CEACAM1 induces B-cell survival and is essential for protective antiviral antibody production
Nature Communications (2015)
-
AMP-activated protein kinase activation mediates CCL3-induced cell migration and matrix metalloproteinase-2 expression in human chondrosarcoma
Cell Communication and Signaling (2013)
-
Carcinoembryonic antigen-related cell adhesion molecules (CEACAMs) in cancer progression and metastasis
Cancer and Metastasis Reviews (2013)