Abstract
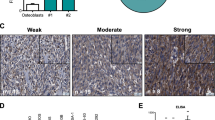
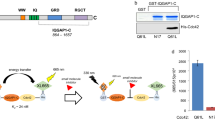
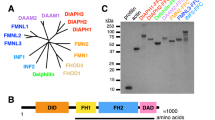
Ezrin is a multifunctional protein that connects the actin cytoskeleton to the extracellular matrix through transmembrane proteins. High ezrin expression is associated with lung metastasis and poor survival in cancer. We screened small molecule libraries for compounds that directly interact with ezrin protein using surface plasmon resonance to identify lead compounds. The secondary functional assays used for lead compound selection included ezrin phosphorylation as measured by immunoprecipitation and in vitro kinase assays, actin binding, chemotaxis, invasion into an endothelial cell monolayer, zebrafish and Xenopus embryonic development, mouse lung organ culture and an in vivo lung metastasis model. Two molecules, NSC305787 and NSC668394, that directly bind to ezrin with low micromolar affinity were selected based on inhibition of ezrin function in multiple assays. They inhibited ezrin phosphorylation, ezrin–actin interaction and ezrin-mediated motility of osteosarcoma (OS) cells in culture. NSC305787 mimicked the ezrin morpholino phenotype, and NSC668394 caused a unique developmental defect consistent with reduced cell motility in zebrafish. Following tail vein injection of OS cells into mice, both molecules inhibited lung metastasis of ezrin-sensitive cells, but not ezrin-resistant cells. The small molecule inhibitors NSC305787 and NSC668394 demonstrate a novel targeted therapy that directly inhibits ezrin protein as an approach to prevent tumor metastasis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Bacci G, Longhi A, Cesari M, Versari M, Bertoni F . (2006). Influence of local recurrence on survival in patients with extremity osteosarcoma treated with neoadjuvant chemotherapy: the experience of a single institution with 44 patients. Cancer 106: 2701–2706.
Bayly C, Cieplak P, Cornell WD, Kollman PA . (1993). A well-behaved electrostatic potential based method using charge restraints for deriving atomic charges: the RESP model. J Phys Chem 97: 10269–10280.
Boger DL, Yasuda M, Mitscher LA, Drake SD, Kitos PA, Thompson SC . (1987). Streptonigrin and lavendamycin partial structures. Probes for the minimum, potent pharmacophore of streptonigrin, lavendamycin, and synthetic quinoline-5,8-diones. J Med Chem 30: 1918–1928.
Bretscher A, Edwards K, Fehon RG . (2002). ERM proteins and merlin: integrators at the cell cortex. Nat Rev Mol Cell Biol 3: 586–599.
Case DA, Darden TA, Cheatham III TE, Simmerling CL, Wang J, Duke RE et al. (2004). AMBER 8. University of California at San Francisco: San Francisco.
Chen K, Fallen S, Abaan HO, Hayran M, Gonzalez C, Wodajo F et al. (2008). Wnt10b induces chemotaxis of osteosarcoma and correlates with reduced survival. Pediatr Blood Cancer 51: 349–355.
Dunn D, Dehner LP . (1977). Metastatic osteosarcoma to lung: a clinicopathologic study of surgical biopsies and resections. Cancer 40: 3054–3064.
Elliott BE, Meens JA, SenGupta SK, Louvard D, Arpin M . (2005). The membrane cytoskeletal crosslinker ezrin is required for metastasis of breast carcinoma cells. Breast Cancer Res 7: R365–R373.
Elzagheid A, Korkeila E, Bendardaf R, Buhmeida A, Heikkila S, Vaheri A et al. (2008). Intense cytoplasmic ezrin immunoreactivity predicts poor survival in colorectal cancer. Hum Pathol 39: 1737–1743.
Fehon RG, McClatchey AI, Bretscher A . (2010). Organizing the cell cortex: the role of ERM proteins. Nat Rev Mol Cell Biol 11: 276–287.
Ferrari S, Palmerini E . (2007). Adjuvant and neoadjuvant combination chemotherapy for osteogenic sarcoma. Curr Opin Oncol 19: 341–346.
Fievet B, Louvard D, Arpin M . (2007). ERM proteins in epithelial cell organization and functions. Biochim Biophys Acta 1773: 653–660.
Fivash M, Towler EM, Fisher RJ . (1998). BIAcore for macromolecular interaction. Curr Opin Biotechnol 9: 97–101.
Gary R, Bretscher A . (1995). Ezrin self-association involves binding of an N-terminal domain to a normally masked C-terminal domain that includes the F-actin binding site. Mol Biol Cell 6: 1061–1075.
Gautreau A, Louvard D, Arpin M . (2000). Morphogenic effects of ezrin require a phosphorylation-induced transition from oligomers to monomers at the plasma membrane. J Cell Biol 150: 193–203.
Gautreau A, Poullet P, Louvard D, Arpin M . (1999). Ezrin, a plasma membrane-microfilament linker, signals cell survival through the phosphatidylinositol 3-kinase/Akt pathway. Proc Natl Acad Sci USA 96: 7300–7305.
Geiger KD, Stoldt P, Schlote W, Derouiche A . (2000). Ezrin immunoreactivity is associated with increasing malignancy of astrocytic tumors but is absent in oligodendrogliomas. Am J Pathol 157: 1785–1793.
Hirao M, Sato N, Kondo T, Yonemura S, Monden M, Sasaki T et al. (1996). Regulation mechanism of ERM (ezrin/radixin/moesin) protein/plasma membrane association: possible involvement of phosphatidylinositol turnover and Rho-dependent signaling pathway. J Cell Biol 135: 37–51.
Khanna C, Khan J, Nguyen P, Prehn J, Caylor J, Yeung C et al. (2001). Metastasis-associated differences in gene expression in a murine model of osteosarcoma. Cancer Res 61: 3750–3759.
Khanna C, Wan X, Bose S, Cassaday R, Olomu O, Mendoza A et al. (2004). The membrane-cytoskeleton linker ezrin is necessary for osteosarcoma metastasis. Nat Med 10: 182–186.
Kimmel CB, Ballard WW, Kimmel SR, Ullmann B, Schilling TF . (1995). Stages of embryonic development of the zebrafish. Dev Dyn 203: 253–310.
Kobel M, Gradhand E, Zeng K, Schmitt WD, Kriese K, Lantzsch T et al. (2006). Ezrin promotes ovarian carcinoma cell invasion and its retained expression predicts poor prognosis in ovarian carcinoma. Int J Gynecol Pathol 25: 121–130.
Legg JW, Isacke CM . (1998). Identification and functional analysis of the ezrin-binding site in the hyaluronan receptor, CD44. Curr Biol 8: 705–708.
Link V, Carvalho L, Castanon I, Stockinger P, Shevchenko A, Heisenberg CP . (2006). Identification of regulators of germ layer morphogenesis using proteomics in zebrafish. J Cell Sci 119: 2073–2083.
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ . (2001). Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv Drug Deliv Rev 46: 3–26.
Longhi A, Errani C, De Paolis M, Mercuri M, Bacci G . (2006). Primary bone osteosarcoma in the pediatric age: state of the art. Cancer Treat Rev 32: 423–436.
Louvet-Vallee S . (2000). ERM proteins: from cellular architecture to cell signaling. Biol Cell 92: 305–316.
Makitie T, Carpen O, Vaheri A, Kivela T . (2001). Ezrin as a prognostic indicator and its relationship to tumor characteristics in uveal malignant melanoma. Invest Ophthalmol Vis Sci 42: 2442–2449.
Malmqvist M . (1999). BIACORE: an affinity biosensor system for characterization of biomolecular interactions. Biochem Soc Trans 27: 335–340.
Matsui T, Maeda M, Doi Y, Yonemura S, Amano M, Kaibuchi K et al. (1998). Rho-kinase phosphorylates COOH-terminal threonines of ezrin/radixin/moesin (ERM) proteins and regulates their head-to-tail association. J Cell Biol 140: 647–657.
Mendoza A, Hong SH, Osborne T, Khan MA, Campbell K, Briggs J et al. (2010). Modeling metastasis biology and therapy in real time in the mouse lung. J Clin Invest 120: 2979–2988.
Ohnmacht CJ, Patel AR, Lutz RE . (1971). Antimalarials. 7. Bis(trifluoromethyl)- -(2-piperidyl)-4-quinolinemethanols. J Med Chem 14: 926–928.
Pang ST, Fang X, Valdman A, Norstedt G, Pousette A, Egevad L et al. (2004). Expression of ezrin in prostatic intraepithelial neoplasia. Urology 63: 609–612.
Park HR, Jung WW, Bacchini P, Bertoni F, Kim YW, Park YK . (2006). Ezrin in osteosarcoma: comparison between conventional high-grade and central low-grade osteosarcoma. Pathol Res Pract 202: 509–515.
Reczek D, Berryman M, Bretscher A . (1997). Identification of EBP50: A PDZ-containing phosphoprotein that associates with members of the ezrin-radixin-moesin family. J Cell Biol 139: 169–179.
Ren L, Hong SH, Cassavaugh J, Osborne T, Chou AJ, Kim SY et al. (2009). The actin-cytoskeleton linker protein ezrin is regulated during osteosarcoma metastasis by PKC. Oncogene 28: 792–802.
Rosen G, Caparros B, Huvos AG, Kosloff C, Nirenberg A, Cacavio A et al. (1982). Preoperative chemotherapy for osteogenic sarcoma: selection of postoperative adjuvant chemotherapy based on the response of the primary tumor to preoperative chemotherapy. Cancer 49: 1221–1230.
Ten Klooster JP, Jansen M, Yuan J, Oorschot V, Begthel H, Di Giacomo V et al. (2009). Mst4 and ezrin induce brush borders downstream of the Lkb1/Strad/Mo25 polarization complex. Dev Cell 16: 551–562.
Wan X, Kim SY, Guenther LM, Mendoza A, Briggs J, Yeung C et al. (2009). Beta4 integrin promotes osteosarcoma metastasis and interacts with ezrin. Oncogene 28: 3401–3411.
Wang J, Wolf RM, Caldwell JW, Kollman PA, Case DA . (2004). Development and testing of a general amber force field. J Comput Chem 25: 1157–1174.
Weng WH, Ahlen J, Astrom K, Lui WO, Larsson C . (2005). Prognostic impact of immunohistochemical expression of ezrin in highly malignant soft tissue sarcomas. Clin Cancer Res 11: 6198–6204.
Westerfield M . (1993). The Zebrafish book: a guide for the laboratory use of zebrafish (Brachydanio rerio). University of Oregon Press: Eugene Oregon.
Yu Y, Khan J, Khanna C, Helman L, Meltzer PS, Merlino G . (2004). Expression profiling identifies the cytoskeletal organizer ezrin and the developmental homeoprotein six-1 as key metastatic regulators. Nat Med 10: 175–181.
Zhang P, Yang Y, Zweidler-McKay PA, Hughes DP . (2008). Critical role of notch signaling in osteosarcoma invasion and metastasis. Clin Cancer Res 14: 2962–2969.
Acknowledgements
Support for our work came from the Children's Cancer Foundation of Baltimore (AÜ), US Department of Defense (W81XWH-10-1-0137, AÜ), Brandon Carrington Lee Foundation (G.K., AÜ), Go4theGoal, Dani's Foundation, Alex's Lemonade Stand Foundation, Liddy Shriver Sarcoma Initiative, Burroughs Wellcome Clinical Scientist Award in Translational Research (JT), and the NIH R01CA133662 (JT), R01CA138212 (JT) and Cancer Center Support Grant P30 CA051008 for use of Biacore Molecular Interaction Shared Resource. We would like to thank to Dr Anthony Bretscher (Cornell University) for providing the WT Ezrin construct, Developmental Therapeutics Program, NCI for providing the small molecule libraries.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Bulut, G., Hong, SH., Chen, K. et al. Small molecule inhibitors of ezrin inhibit the invasive phenotype of osteosarcoma cells. Oncogene 31, 269–281 (2012). https://doi.org/10.1038/onc.2011.245
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2011.245
Keywords
This article is cited by
-
Osteosarcoma
Nature Reviews Disease Primers (2022)
-
NSC305787, a pharmacological ezrin inhibitor, exhibits antineoplastic activity in pancreatic cancer cells
Investigational New Drugs (2022)
-
Aggressive nonfunctioning pituitary neuroendocrine tumors
Brain Tumor Pathology (2022)
-
EZR promotes pancreatic cancer proliferation and metastasis by activating FAK/AKT signaling pathway
Cancer Cell International (2021)
-
Identification of key genes as predictive biomarkers for osteosarcoma metastasis using translational bioinformatics
Cancer Cell International (2021)