Abstract
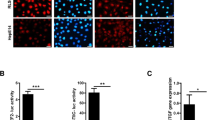
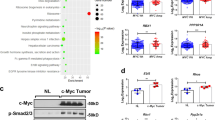
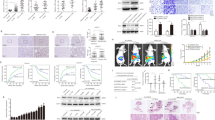
Yes-associated protein (YAP) is a downstream effector of the Hippo signaling pathway, which controls organ expansion and tissue development. We have recently defined the tumorigenic potential and clinical significance of the YAP1 oncogene in human hepatocellular carcinoma (HCC). The present study aims to define the tumorigenic properties of YAP in HCC and elucidate the related downstream signaling mechanism. In a gain-of-function study, we demonstrated that ectopic increased expression of YAP in the immortalized non-tumorigenic hepatocyte cell line MIHA confers tumorigenic and metastatic potentials, as evidenced by (1) enhanced aptitudes in cell viability, anchorage-independent growth, migration and invasion; (2) tumor formation in a xenograft mouse model; and (3) induction of HCC biomarker α-fetoprotein and activation of mitogen-activated protein kinase. Furthermore, we have identified AXL, a receptor tyrosine kinase, as a key downstream target that drives YAP-dependent oncogenic functions. RNAi-mediated knockdown of AXL expression decreased the ability of YAP-expressing MIHA cells and of the primary HCC cell line to proliferate and invade. These results indicate that AXL is a mediator of YAP-dependent oncogenic activities and implicates it as a potential therapeutic target for HCC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Bandura JL, Edgar BA . (2008). Yorkie and Scalloped: partners in growth activation. Dev Cell 14: 315–316.
Berclaz G, Altermatt HJ, Rohrbach V, Kieffer I, Dreher E, Andres AC . (2001). Estrogen dependent expression of the receptor tyrosine kinase axl in normal and malignant human breast. Ann Oncol 12: 819–824.
Bittner M, Meltzer P, Chen Y, Jiang Y, Seftor E, Hendrix M et al. (2000). Molecular classification of cutaneous malignant melanoma by gene expression profiling. Nature 406: 536–540.
Bueno L, de Alwis DP, Pitou C, Yingling J, Lahn M, Glatt S et al. (2008). Semi-mechanistic modelling of the tumour growth inhibitory effects of LY2157299, a new type I receptor TGF-beta kinase antagonist, in mice. Eur J Cancer 44: 142–150.
Buttitta LA, Edgar BA . (2007). How size is controlled: from Hippos to Yorkies. Nat Cell Biol 9: 1225–1227.
Camargo FD, Gokhale S, Johnnidis JB, Fu D, Bell GW, Jaenisch R et al. (2007). YAP1 increases organ size and expands undifferentiated progenitor cells. Curr Biol 17: 2054–2060.
Cao X, Pfaff SL, Gage FH . (2008). YAP regulates neural progenitor cell number via the TEA domain transcription factor. Genes Dev 22: 3320–3334.
Chan SW, Lim CJ, Loo LS, Chong YF, Huang C, Hong W . (2009). TEADs Mediate Nuclear Retention of TAZ to Promote Oncogenic Transformation. J Biol Chem 284: 14347–14358.
Craven RJ, Xu LH, Weiner TM, Fridell YW, Dent GA, Srivastava S et al. (1995). Receptor tyrosine kinases expressed in metastatic colon cancer. Int J Cancer 60: 791–797.
Demarchi F, Verardo R, Varnum B, Brancolini C, Schneider C . (2001). Gas6 anti-apoptotic signaling requires NF-kappa B activation. J Biol Chem 276: 31738–31744.
Dong J, Feldmann G, Huang J, Wu S, Zhang N, Comerford SA et al. (2007). Elucidation of a universal size-control mechanism in Drosophila and mammals. Cell 130: 1120–1133.
Edgar BA . (2006). From cell structure to transcription: Hippo forges a new path. Cell 124: 267–273.
Goruppi S, Ruaro E, Schneider C . (1996). Gas6, the ligand of Axl tyrosine kinase receptor, has mitogenic and survival activities for serum starved NIH3T3 fibroblasts. Oncogene 12: 471–480.
Hafizi S, Dahlback B . (2006). Signalling and functional diversity within the Axl subfamily of receptor tyrosine kinases. Cytokine Growth Factor Rev 17: 295–304.
Hao Y, Chun A, Cheung K, Rashidi B, Yang X . (2008). Tumor suppressor LATS1 is a negative regulator of oncogene YAP. J Biol Chem 283: 5496–5509.
Hasanbasic I, Cuerquis J, Varnum B, Blostein MD . (2004). Intracellular signaling pathways involved in Gas6-Axl-mediated survival of endothelial cells. Am J Physiol Heart Circ Physiol 287: H1207–H1213.
Huang J, Wu S, Barrera J, Matthews K, Pan D . (2005). The Hippo signaling pathway coordinately regulates cell proliferation and apoptosis by inactivating Yorkie, the Drosophila homolog of YAP. Cell 122: 421–434.
Lay JD, Hong CC, Huang JS, Yang YY, Pao CY, Liu CH et al. (2007). Sulfasalazine suppresses drug resistance and invasiveness of lung adenocarcinoma cells expressing AXL. Cancer Res 67: 3878–3887.
Lei QY, Zhang H, Zhao B, Zha ZY, Bai F, Pei XH et al. (2008). TAZ promotes cell proliferation and epithelial-mesenchymal transition and is inhibited by the hippo pathway. Mol Cell Biol 28: 2426–2436.
Liu AM, Xu MZ, Chen J, Poon RT, Luk JM . (2010). Targeting YAP and Hippo signaling pathway in liver cancer. Expert Opin Ther Targets 14: 855–868.
Liu LX, Lee NP, Chan VW, Xue W, Zender L, Zhang C et al. (2009). Targeting cadherin-17 inactivates Wnt signaling and inhibits tumor growth in liver carcinoma. Hepatology 50: 1453–1463.
Luk JM, Wang PP, Lee CK, Wang JH, Fan ST . (2005). Hepatic potential of bone marrow stromal cells: development of in vitro co-culture and intra-portal transplantation models. J Immunol Methods 305: 39–47.
Oh H, Irvine KD . (2010). Yorkie: the final destination of Hippo signaling. Trends Cell Biol 20: 410–417.
Overholtzer M, Zhang J, Smolen GA, Muir B, Li W, Sgroi DC et al. (2006). Transforming properties of YAP, a candidate oncogene on the chromosome 11q22 amplicon. Proc Natl Acad Sci USA 103: 12405–12410.
Pan D . (2007). Hippo signaling in organ size control. Genes Dev 21: 886–897.
Peeper D, Berns A . (2006). Cross-species oncogenomics in cancer gene identification. Cell 125: 1230–1233.
Sainaghi PP, Castello L, Bergamasco L, Galletti M, Bellosta P, Avanzi GC . (2005). Gas6 induces proliferation in prostate carcinoma cell lines expressing the Axl receptor. J Cell Physiol 204: 36–44.
Sawabu T, Seno H, Kawashima T, Fukuda A, Uenoyama Y, Kawada M et al. (2007). Growth arrest-specific gene 6 and Axl signaling enhances gastric cancer cell survival via Akt pathway. Mol Carcinog 46: 155–164.
Shieh YS, Lai CY, Kao YR, Shiah SG, Chu YW, Lee HS et al. (2005). Expression of axl in lung adenocarcinoma and correlation with tumor progression. Neoplasia 7: 1058–1064.
Steinhardt AA, Gayyed MF, Klein AP, Dong J, Maitra A, Pan D et al. (2008). Expression of Yes-associated protein in common solid tumors. Hum Pathol 39: 1582–1589.
Tai KY, Shieh YS, Lee CS, Shiah SG, Wu CW . (2008). Axl promotes cell invasion by inducing MMP-9 activity through activation of NF-kappaB and Brg-1. Oncogene 27: 4044–4055.
Thompson BJ, Cohen SM . (2006). The Hippo pathway regulates the bantam microRNA to control cell proliferation and apoptosis in Drosophila. Cell 126: 767–774.
Tsou AP, Wu KM, Tsen TY, Chi CW, Chiu JH, Lui WY et al. (1998). Parallel hybridization analysis of multiple protein kinase genes: identification of gene expression patterns characteristic of human hepatocellular carcinoma. Genomics 50: 331–340.
Wong BW, Luk JM, Ng IO, Hu MY, Liu KD, Fan ST . (2003). Identification of liver-intestine cadherin in hepatocellular carcinoma--a potential disease marker. Biochem Biophys Res Commun 311: 618–624.
Wu S, Liu Y, Zheng Y, Dong J, Pan D . (2008). The TEAD/TEF family protein Scalloped mediates transcriptional output of the Hippo growth-regulatory pathway. Dev Cell 14: 388–398.
Xu MZ, Yao TJ, Lee NP, Ng IO, Chan YT, Zender L et al. (2009). Yes-associated protein is an independent prognostic marker in hepatocellular carcinoma. Cancer 115: 4576–4585.
Yin F, Pan D . (2007). Fat flies expanded the hippo pathway: a matter of size control. Sci STKE 2007: pe12.
Zender L, Lowe SW . (2008). Integrative oncogenomic approaches for accelerated cancer-gene discovery. Curr Opin Oncol 20: 72–76.
Zender L, Spector MS, Xue W, Flemming P, Cordon-Cardo C, Silke J et al. (2006). Identification and validation of oncogenes in liver cancer using an integrative oncogenomic approach. Cell 125: 1253–1267.
Zender L, Xue W, Zuber J, Semighini CP, Krasnitz A, Ma B et al. (2008). An oncogenomics- based in vivo RNAi screen identifies tumor suppressors in liver cancer. Cell 135: 852–864.
Zeng Q, Hong W . (2008). The emerging role of the hippo pathway in cell contact inhibition, organ size control, and cancer development in mammals. Cancer Cell 13: 188–192.
Zhang YX, Knyazev PG, Cheburkin YV, Sharma K, Knyazev YP, Orfi L et al. (2008). AXL is a potential target for therapeutic intervention in breast cancer progression. Cancer Res 68: 1905–1915.
Zhao B, Lei QY, Guan KL . (2008a). The Hippo-YAP pathway: new connections between regulation of organ size and cancer. Curr Opin Cell Biol 20: 638–646.
Zhao B, Li L, Tumaneng K, Wang CY, Guan KL . (2010). A coordinated phosphorylation by Lats and CK1 regulates YAP stability through SCF(beta-TRCP). Genes Dev 24: 72–85.
Zhao B, Wei X, Li W, Udan RS, Yang Q, Kim J et al. (2007). Inactivation of YAP oncoprotein by the Hippo pathway is involved in cell contact inhibition and tissue growth control. Genes Dev 21: 2747–2761.
Zhao B, Ye X, Yu J, Li L, Li W, Li S et al. (2008b). TEAD mediates YAP-dependent gene induction and growth control. Genes Dev 22: 1962–1971.
Acknowledgements
We thank Dr Yuk-Tat Chan (Queen Mary Hospital, HK) for the AFP assay and Dr Stella Sun (University of Hong Kong)) for technical assistance on cell line maintenance.
Sources of support: The work was supported by grants to JML from the Biomedical Research Grants Council of Singapore and by the National University Cancer Institute (NCIS) Centre Grant; WH was supported by the A*STAR and MX by the National Natural Science Foundation of China (Grant No. 81000880). SWL is an investigator in the Howard Hughes Medical Institute and Dr Lowe's work is supported by a program grant (CA13106) from the National Cancer Institute of NIH.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Xu, M., Chan, S., Liu, A. et al. AXL receptor kinase is a mediator of YAP-dependent oncogenic functions in hepatocellular carcinoma. Oncogene 30, 1229–1240 (2011). https://doi.org/10.1038/onc.2010.504
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.504
Keywords
This article is cited by
-
Targeting AXL induces tumor-intrinsic immunogenic response in tyrosine kinase inhibitor-resistant liver cancer
Cell Death & Disease (2024)
-
Loss-of-function mutations of SOX17 lead to YAP/TEAD activation-dependent malignant transformation in endometrial cancer
Oncogene (2023)
-
Gas6 in chronic liver disease—a novel blood-based biomarker for liver fibrosis
Cell Death Discovery (2023)
-
CYT387, a potent IKBKE inhibitor, suppresses human glioblastoma progression by activating the Hippo pathway
Journal of Translational Medicine (2021)
-
RGS12 is a novel tumor suppressor in osteosarcoma that inhibits YAP-TEAD1-Ezrin signaling
Oncogene (2021)