Abstract
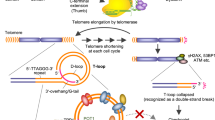
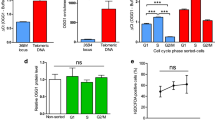
New anti-telomere strategies represent important goals for the development of selective cancer therapies. In this study, we reported that uncapped telomeres, resulting from pharmacological stabilization of quadruplex DNA by RHPS4 (3,11-difluoro-6,8,13-trimethyl-8H-quino[4,3,2-kl]acridinium methosulfate), trigger specific recruitment and activation of poly-adenosine diphosphate (ADP) ribose polymerase I (PARP1) at the telomeres, forming several ADP-ribose polymers that co-localize with the telomeric repeat binding factor 1 protein and are inhibited by selective PARP(s) inhibitors or PARP1-specific small interfering RNAs. The knockdown of PARP1 prevents repairing of RHPS4-induced telomere DNA breaks, leading to increases in chromosome abnormalities and eventually to the inhibition of tumor cell growth both in vitro and in xenografts. More interestingly, the integration of a TOPO1 inhibitor on the combination treatment proved to have a high therapeutic efficacy ensuing a complete regression of the tumor as well as a significant increase in overall survival and cure of mice even when treatments started at a very late stage of tumor growth. Overall, this work reveals the unexplored link between the PARP1 and G-quadruplex ligands and demonstrates the excellent efficacy of a multi-component strategy based on the use of PARP inhibitors in telomere-based therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Amé JC, Rolli V, Schreiber V, Niedergang C, Apiou F, Decker P et al. (1999). PARP-2, a novel mammalian DNA damage-dependent poly(ADP-ribose) polymerase. J Biol Chem 274: 17860–17868.
Amé JC, Spenlehauer C, de Murcia G . (2004). The PARP superfamily. Bioessays 26: 882–893.
Burkle A (eds). (2005). Poly(ADP-ribosyl)ation. Landes Biosciences: Austin, TX, USA.
Brunori M, Mathieu N, Ricoul M, Bauwens S, Koering CE, Roborel de Climens A et al. (2006). TRF2 inhibition promotes anchorage-independent growth of telomerase-positive human fibroblasts. Oncogene 25: 990–997.
Biroccio A, Amodei S, Benassi B, Scarsella M, Cianciulli A, Mottolese M et al. (2002). Reconstitution of hTERT restores tumorigenicity in melanoma-derived c-Myc low-expressing clones. Oncogene 21: 3011–3019.
Bakondi E, Bai P, Szabó EE, Hunyadi J, Gergely P, Szabó C et al. (2002). Detection of poly(ADP-ribose) polymerase activation in oxidatively stressed cells and tissues using biotinylated NAD substrate. J Histochem Cytochem 50: 91–98.
Cook BD, Dynek JN, Chang W, Shostak G, Smith S . (2002). Role for the related poly(ADP-ribose) polymerases tankyrase 1 and 2 at human telomeres. Mol Cell Biol 22: 332–342.
Dantzer F, Giraud-Panis MJ, Jaco I, Amé JC, Schultz I, Blasco M et al. (2004). Functional interaction between poly(ADP-ribose) polymerase 2 (PARP-2) and TRF2: PARP activity negatively regulates TRF2. Mol Cell Biol 24: 1595–1607.
d'Adda di Fagagna F, Reaper PM, Clay-Farrace L, Fiegler H, Carr P, Von Zglinicki T et al. (2003). A DNA damage checkpoint response in telomere-initiated senescence. Nature 426: 194–198.
de Lange T . (2005). Shelterin: the protein complex that shapes and safeguards human telomeres. Genes Dev 19: 2100–2110.
Donigian JR, de Lange T . (2007). The role of the poly(ADP-ribose) polymerase tankyrase1 in telomere length control by the TRF1 component of the shelterin complex. J Biol Chem 282: 22662–22667.
Farmer H, McCabe N, Lord CJ, Tutt AN, Johnson DA, Richardson TB et al. (2005). Targeting the DNA repair defect in BRCA mutant cells as a therapeutic strategy. Nature 434: 917–921.
Gavathiotis E, Heald RA, Stevens MF, Searle MS . (2003). Drug recognition and stabilisation of the parallel-stranded DNA quadruplex d(TTAGGGT)4 containing the human telomeric repeat. J Mol Biol 334: 25–36.
Gagné JP, Hendzel MJ, Droit A, Poirier GG . (2006). The expanding role of poly(ADP-ribose) metabolism: current challenges and new perspectives. Curr Opin Cell Biol 18: 145–151.
Geran RI, Greenberg NH, Macdonald MM, Shumacher AM, Abbott BJ . (1972). Protocols for screening chemical agents and natural products against animal tumors and other biological systems, 3rd edn. Cancer Chemother Rep 3: 1–88.
Gomez M, Wu J, Schreiber V, Dunlap J, Dantzer F, Wang Y et al. (2006). PARP1 is a TRF2-associated poly(ADP-ribose)polymerase and protects eroded telomeres. Mol Biol Cell 17: 1686–1696.
Griffith JD, Comeau L, Rosenfield S, Stansel RM, Bianchi A, Moss H et al. (1999). Mammalian telomeres end in a large duplex loop. Cell 97: 503–514.
Heald RA, Modi C, Cookson JC, Hutchinson I, Laughton CA, Gowan SM et al. (2002). Antitumor polycyclic acridines 8 synthesis and telomerase-inhibitory activity of methylated pentacyclic acridinium salts. J Med Chem 45: 590–597.
Kaminker PG, Kim SH, Taylor RD, Zebarjadian Y, Funk WD, Morin GB et al. (2001). TANK2, a new TRF1-associated poly(ADP-ribose) polymerase, causes rapid induction of cell death upon overexpression. J Biol Chem 276: 35891–35899.
Kelland L . (2007). Targeting the limitless replicative potential of cancer: the telomerase/telomere pathway. Clin Cancer Res 13: 4960–4963.
Lenain C, Bauwens S, Amiard S, Brunori M, Giraud-Panis MJ, Gilson E . (2006). The Apollo 5′ exonuclease functions together with TRF2 to protect telomeres from DNA repair. Curr Biol 16: 1303–1310.
Leonetti C, Scarsella M, Riggio G, Rizzo A, Salvati E, D′Incalci M et al. (2008). G-quadruplex ligand RHPS4 potentiates the antitumor activity of camptothecins in preclinical models of solid tumors. Clin Cancer Res 14: 7284–7291.
Lord CJ, Ashworth A . (2008). Targeted therapy for cancer using PARP inhibitors. Curr Opin Pharmacol 8: 363–369.
Lindahl T, Satoh MS, Poirier GG, Klungland A . (1995). Post-translational modification of poly(ADP-ribose) polymerase induced by DNA strand breaks. Trends Biochem Sci 20: 405–411.
Makarov VL, Hirose Y, Langmore JP . (1997). Long G tails at both ends of human chromosomes suggest a C strand degradation mechanism for telomere shortening. Cell 88: 657–666.
Malanga M, Althaus FR . (2005). The role of poly(ADP-ribose) in the DNA damage signaling network. Biochem Cell Biol 83: 354–364.
Marcellini M, De Luca N, Riccioni T, Ciucci A, Orecchia A, Lacal PM et al. (2006). Increased melanoma growth and metastasis spreading in mice overexpressing placenta growth factor. Am J Pathol 169: 643–654.
Oganesian L, Bryan TM . (2007). Physiological relevance of telomeric G-quadruplex formation: a potential drug target. Bioessays 29: 155–165.
Orlandi A, Di Lascio A, Francesconi A, Scioli MG, Arcuri G, Ferlosio A et al. (2008). Stem cell marker expression and proliferation and apoptosis of vascular smooth muscle cells. Cell Cycle 7: 3889–3897.
Pleschke JM, Kleczkowska HE, Strohm M, Althaus FR . (2000). Poly(ADP-ribose) binds to specific domains in DNA damage checkpoint proteins. J Biol Chem 275: 40974–44080.
Plummer ER, Calvert H . (2007). Targeting poly(ADP-ribose) polymerase: a two-armed strategy for cancer therapy. Clin Cancer Res 13: 6252–6256.
Pisano C, De Cesare M, Beretta GL, Zuco V, Pratesi G, Penco S et al. (2008). Preclinical profile of antitumor activity of a novel hydrophilic campothecin, ST 1968. Mol Cancer Ther 7: 2051–2059.
Ratnam K, Low JA . (2007). Current development of clinical inhibitors of poly(ADP-ribose) polymerase in oncology. Clin Cancer Res 13: 1383–1388.
Rippmann JF, Damm K, Schnapp A . (2002). Functional characterization of the poly(ADP-ribose) polymerase activity of tankyrase 1, a potential regulator of telomere length. J Mol Biol 323: 217–224.
Rizzo A, Salvati E, Porru M, D′Angelo C, Stevens MF, D′Incalci M et al. (2009). Stabilization of quadruplex DNA perturbs telomere replication leading to the activation of an ATR-dependent ATM signaling pathway. Nucleic Acids Res 37: 5353–5364.
Schreiber V, Amé JC, Dollé P, Schultz I, Rinaldi B, Fraulob V et al. (2002). Poly(ADP-ribose) polymerase-2 (PARP-2) is required for efficient base excision DNA repair in association with PARP-1 and XRCC1. J Biol Chem 277: 23028–23036.
Salvati E, Leonetti C, Rizzo A, Scarsella M, Mottolese M, Galati R et al. (2007). Telomere damage induced by the G-quadruplex ligand RHPS4 has an antitumor effect. J Clin Invest 117: 3236–3247.
Smith S, de Lange T . (1999). Cell cycle dependent localization of the telomeric PARP, tankyrase, to nuclear pore complexes and centrosomes. J Cell Sci 112 (Pt 21): 3649–3656.
Sugimura K, Takebayashi S, Taguchi H, Takeda S, Okumura K . (2008). PARP-1 ensures regulation of replication fork progression by homologous recombination on damaged DNA. J Cell Biol 183: 1203–1212.
Takai H, Smogorzewska A, de Lange T . (2003). DNA damage foci at dysfunctional telomeres. Curr Biol 13: 1549–1556.
Tentori L, Graziani G . (2005). Chemopotentiation by PARP inhibitors in cancer therapy. Pharmacol Res 52: 25–33.
Tentori L, Leonetti C, Scarsella M, Muzi A, Mazzon E, Vergati M et al. (2006). Inhibition of poly(ADP-ribose) polymerase prevents irinotecan-induced intestinal damage and enhances irinotecan/temozolomide efficacy against colon carcinoma. FASEB J 20: 1709–1711.
Wright WE, Tesmer VM, Huffman KE, Levene SD, Shay JW . (1997). Normal human chromosomes have long G-rich telomeric overhangs at one end. Genes Dev 11: 2801–2809.
Acknowledgements
This work was supported by grants from the Italian Association for Cancer Research (AIRC), Ministero della Salute and Ligue Nationale contre le Cancer (EG ‘équipe labellisée’). ES and MP are recipients of fellowships from the Italian Foundation for Cancer Research (FIRC). We thank Mrs Carmen D’Angelo for generating HT29 cells that express the luciferase gene and Mrs Adele Petricca for her helpful assistance in typing the manuscript. We are also grateful to Tania Merlino for the English revision.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Salvati, E., Scarsella, M., Porru, M. et al. PARP1 is activated at telomeres upon G4 stabilization: possible target for telomere-based therapy. Oncogene 29, 6280–6293 (2010). https://doi.org/10.1038/onc.2010.344
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.344
Keywords
This article is cited by
-
PARP2 promotes Break Induced Replication-mediated telomere fragility in response to replication stress
Nature Communications (2024)
-
PARP1 allows proper telomere replication through TRF1 poly (ADP-ribosyl)ation and helicase recruitment
Communications Biology (2023)
-
Functions of ADP-ribose transferases in the maintenance of telomere integrity
Cellular and Molecular Life Sciences (2022)
-
G-quadruplex binding properties of a potent PARP-1 inhibitor derived from 7-azaindole-1-carboxamide
Scientific Reports (2021)
-
The regulation and functions of DNA and RNA G-quadruplexes
Nature Reviews Molecular Cell Biology (2020)