Abstract
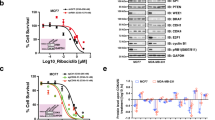
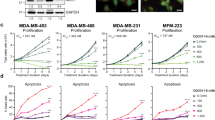
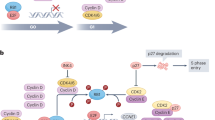
A hallmark of cancer is the deregulation of cell-cycle machinery, ultimately facilitating aberrant proliferation that fuels tumorigenesis and disease progression. Particularly, in breast cancers, cyclin D1 has a crucial role in the development of disease. Recently, a highly specific inhibitor of CDK4/6 activity (PD-0332991) has been developed that may have efficacy in the treatment of breast cancer. To interrogate the utility of PD-0332991 in treating breast cancers, therapeutic response was evaluated on a panel of breast cancer cell lines. These analyses showed that the chronic loss of Rb is specifically associated with evolution to a CDK4/6-independent state and, ultimately, resistance to PD-0332991. However, to interrogate the functional consequence of Rb directly, knockdown experiments were performed in models that represent immortalized mammary epithelia and multiple subtypes of breast cancer. These studies showed a highly specific role for Rb in mediating the response to CDK4/6 inhibition that was dependent on transcriptional repression manifest through E2F, and the ability to attenuate CDK2 activity. Acquired resistance to PD-03322991 was specifically associated with attenuation of CDK2 inhibitors, indicating that redundancy in CDK functions represents a determinant of therapeutic failure. Despite these caveats, in specific models, PD-0332991 was a particularly effective therapy, which induced Rb-dependent cytostasis. Combined, these findings indicate the critical importance of fully understanding cell-cycle regulatory pathways in directing the utilization of CDK inhibitors in the clinic.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Abukhdeir AM, Park BH . (2008). P21 and p27: roles in carcinogenesis and drug resistance. Expert Rev Mol Med 10: e19.
Alle KM, Henshall SM, Field AS, Sutherland RL . (1998). Cyclin D1 protein is overexpressed in hyperplasia and intraductal carcinoma of the breast. Clin Cancer Res 4: 847–854.
Ariazi EA, Ariazi JL, Cordera F, Jordan VC . (2006). Estrogen receptors as therapeutic targets in breast cancer. Curr Top Med Chem 6: 181–202.
Barker S . (2003). Anti-estrogens in the treatment of breast cancer: current status and future directions. Curr Opin Investig Drugs 4: 652–657.
Borg A, Zhang QX, Alm P, Olsson H, Sellberg G . (1992). The retinoblastoma gene in breast cancer: allele loss is not correlated with loss of gene protein expression. Cancer Res 52: 2991–2994.
Brenner AJ, Paladugu A, Wang H, Olopade OI, Dreyling MH, Aldaz CM . (1996). Preferential loss of expression of p16(INK4a) rather than p19(ARF) in breast cancer. Clin Cancer Res 2: 1993–1998.
Buzdar AU . (2009). Role of biologic therapy and chemotherapy in hormone receptor- and HER2-positive breast cancer. Ann Oncol 20: 993–999.
Dannenberg JH, te Riele HP . (2006). The retinoblastoma gene family in cell cycle regulation and suppression of tumorigenesis. Results Probl Cell Differ 42: 183–225.
Diehl JA, Zindy F, Sherr CJ . (1997). Inhibition of cyclin D1 phosphorylation on threonine-286 prevents its rapid degradation via the ubiquitin-proteasome pathway. Genes Dev 11: 957–972.
Donovan JC, Milic A, Slingerland JM . (2001). Constitutive MEK/MAPK activation leads to p27(Kip1) deregulation and antiestrogen resistance in human breast cancer cells. J Biol Chem 276: 40888–40895.
Dyson N . (1998). The regulation of E2F by pRB-family proteins. Genes Dev 12: 2245–2262.
Encarnacion CA, Ciocca DR, McGuire WL, Clark GM, Fuqua SA, Osborne CK . (1993). Measurement of steroid hormone receptors in breast cancer patients on tamoxifen. Breast Cancer Res Treat 26: 237–246.
Fry DW, Harvey PJ, Keller PR, Elliott WL, Meade M, Trachet E et al. (2004). Specific inhibition of cyclin-dependent kinase 4/6 by PD 0332991 and associated antitumor activity in human tumor xenografts. Mol Cancer Ther 3: 1427–1438.
Geng Y, Whoriskey W, Park MY, Bronson RT, Medema RH, Li T et al. (1999). Rescue of cyclin D1 deficiency by knockin cyclin E. Cell 97: 767–777.
Gillett C, Fantl V, Smith R, Fisher C, Bartek J, Dickson C et al. (1994). Amplification and overexpression of cyclin D1 in breast cancer detected by immunohistochemical staining. Cancer Res 54: 1812–1817.
Halaban R, Miglarese MR, Smicun Y, Puig S . (1998). Melanomas, from the cell cycle point of view (review). Int J Mol Med 1: 419–425.
Hanahan D, Weinberg RA . (2000). The hallmarks of cancer. Cell 100: 57–70.
Harari D, Yarden Y . (2000). Molecular mechanisms underlying ErbB2/HER2 action in breast cancer. Oncogene 19: 6102–6114.
Jemal A, Siegel R, Ward E, Hao Y, Xu J, Thun MJ . (2009). Cancer statistics, 2009. CA Cancer J Clin 59: 225–249.
Jensen EV, Jordan VC . (2003). The estrogen receptor: a model for molecular medicine. Clin Cancer Res 9: 1980–1989.
Keyomarsi K, O'Leary N, Molnar G, Lees E, Fingert HJ, Pardee AB . (1994). Cyclin E, a potential prognostic marker for breast cancer. Cancer Res 54: 380–385.
Keyomarsi K, Pardee AB . (1993). Redundant cyclin overexpression and gene amplification in breast cancer cells. Proc Natl Acad Sci USA 90: 1112–1116.
Knudsen ES, Knudsen KE . (2008). Tailoring to RB: tumour suppressor status and therapeutic response. Nat Rev Cancer 8: 714–724.
LaBaer J, Garrett MD, Stevenson LF, Slingerland JM, Sandhu C, Chou HS et al. (1997). New functional activities for the p21 family of CDK inhibitors. Genes Dev 11: 847–862.
Loden M, Stighall M, Nielsen NH, Roos G, Emdin SO, Ostlund H et al. (2002). The cyclin D1 high and cyclin E high subgroups of breast cancer: separate pathways in tumorogenesis based on pattern of genetic aberrations and inactivation of the pRb node. Oncogene 21: 4680–4690.
Malumbres M, Barbacid M . (2001). To cycle or not to cycle: a critical decision in cancer. Nat Rev Cancer 1: 222–231.
Malumbres M, Barbacid M . (2005). Mammalian cyclin-dependent kinases. Trends Biochem Sci 30: 630–641.
Mittnacht S . (1998). Control of pRB phosphorylation. Curr Opin Genet Dev 8: 21–27.
Nielsen NH, Emdin SO, Cajander J, Landberg G . (1997). Deregulation of cyclin E and D1 in breast cancer is associated with inactivation of the retinoblastoma protein. Oncogene 14: 295–304.
Okamoto A, Demetrick DJ, Spillare EA, Hagiwara K, Hussain SP, Bennett WP et al. (1994). p16INK4 mutations and altered expression in human tumors and cell lines. Cold Spring Harb Symp Quant Biol 59: 49–57.
Ormandy CJ, Musgrove EA, Hui R, Daly RJ, Sutherland RL . (2003). Cyclin D1, EMS1 and 11q13 amplification in breast cancer. Breast Cancer Res Treat 78: 323–335.
Ortega S, Malumbres M, Barbacid M . (2002). Cyclin D-dependent kinases, INK4 inhibitors and cancer. Biochim Biophys Acta 1602: 73–87.
Otterson GA, Kratzke RA, Coxon A, Kim YW, Kaye FJ . (1994). Absence of p16INK4 protein is restricted to the subset of lung cancer lines that retains wildtype RB. Oncogene 9: 3375–3378.
Pietilainen T, Lipponen P, Aaltomaa S, Eskelinen M, Kosma VM, Syrjanen K . (1995). Expression of retinoblastoma gene protein (Rb) in breast cancer as related to established prognostic factors and survival. Eur J Cancer 31A: 329–333.
Planas-Silva MD, Weinberg RA . (1997). Estrogen-dependent cyclin E-cdk2 activation through p21 redistribution. Mol Cell Biol 17: 4059–4069.
Poon RY, Toyoshima H, Hunter T . (1995). Redistribution of the CDK inhibitor p27 between different cyclin.CDK complexes in the mouse fibroblast cell cycle and in cells arrested with lovastatin or ultraviolet irradiation. Mol Biol Cell 6: 1197–1213.
Porter PL, Malone KE, Heagerty PJ, Alexander GM, Gatti LA, Firpo EJ et al. (1997). Expression of cell-cycle regulators p27Kip1 and cyclin E, alone and in combination, correlate with survival in young breast cancer patients. Nat Med 3: 222–225.
Prall OW, Sarcevic B, Musgrove EA, Watts CK, Sutherland RL . (1997). Estrogen-induced activation of Cdk4 and Cdk2 during G1-S phase progression is accompanied by increased cyclin D1 expression and decreased cyclin-dependent kinase inhibitor association with cyclin E-Cdk2. J Biol Chem 272: 10882–10894.
Rizzi F, Belloni L, Crafa P, Lazzaretti M, Remondini D, Ferretti S et al. (2008). A novel gene signature for molecular diagnosis of human prostate cancer by RT-qPCR. PLoS One 3: e3617.
Robertson JF . (1996). Oestrogen receptor: a stable phenotype in breast cancer. Br J Cancer 73: 5–12.
Sage J, Mulligan GJ, Attardi LD, Miller A, Chen S, Williams B et al. (2000). Targeted disruption of the three Rb-related genes leads to loss of G(1) control and immortalization. Genes Dev 14: 3037–3050.
Santamaria D, Ortega S . (2006). Cyclins and CDKS in development and cancer: lessons from genetically modified mice. Front Biosci 11: 1164–1188.
Shapiro GI . (2006). Cyclin-dependent kinase pathways as targets for cancer treatment. J Clin Oncol 24: 1770–1783.
Shapiro GI, Edwards CD, Kobzik L, Godleski J, Richards W, Sugarbaker DJ et al. (1995). Reciprocal Rb inactivation and p16INK4 expression in primary lung cancers and cell lines. Cancer Res 55: 505–509.
Sherr CJ, Roberts JM . (1999). CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev 13: 1501–1512.
Stengel KR, Thangavel C, Solomon DA, Angus SP, Zheng Y, Knudsen ES . (2009). Retinoblastoma/p107/p130 pocket proteins: protein dynamics and interactions with target gene promoters. J Biol Chem 284: 19265–19271.
Sutherland RL, Hall RE, Taylor IW . (1983). Cell proliferation kinetics of MCF-7 human mammary carcinoma cells in culture and effects of tamoxifen on exponentially growing and plateau-phase cells. Cancer Res 43: 3998–4006.
Swarbrick A, Lee CS, Sutherland RL, Musgrove EA . (2000). Cooperation of p27(Kip1) and p18(INK4c) in progestin-mediated cell cycle arrest in T-47D breast cancer cells. Mol Cell Biol 20: 2581–2591.
Takano Y, Takenaka H, Kato Y, Masuda M, Mikami T, Saegusa M et al. (1999). Cyclin D1 overexpression in invasive breast cancers: correlation with cyclin-dependent kinase 4 and oestrogen receptor overexpression, and lack of correlation with mitotic activity. J Cancer Res Clin Oncol 125: 505–512.
Toogood PL, Harvey PJ, Repine JT, Sheehan DJ, VanderWel SN, Zhou H et al. (2005). Discovery of a potent and selective inhibitor of cyclin-dependent kinase 4/6. J Med Chem 48: 2388–2406.
Vaughn DJ, Flaherty K, Lal P, Gallagher M, O′Dwyer P, Wilner K et al. (2009). Treatment of growing teratoma syndrome. N Engl J Med 360: 423–424.
Vivar OI, Lin CL, Firestone GL, Bjeldanes LF . (2009). 3,3′-Diindolylmethane induces a G(1) arrest in human prostate cancer cells irrespective of androgen receptor and p53 status. Biochem Pharmacol 78: 469–476.
Wakimoto N, Wolf I, Yin D, O'Kelly J, Akagi T, Abramovitz L et al. (2008). Nonsteroidal anti-inflammatory drugs suppress glioma via 15-hydroxyprostaglandin dehydrogenase. Cancer Res 68: 6978–6986.
Wang L, Wang J, Blaser BW, Duchemin AM, Kusewitt DF, Liu T et al. (2007). Pharmacologic inhibition of CDK4/6: mechanistic evidence for selective activity or acquired resistance in acute myeloid leukemia. Blood 110: 2075–2083.
Wang TC, Cardiff RD, Zukerberg L, Lees E, Arnold A, Schmidt EV . (1994). Mammary hyperplasia and carcinoma in MMTV-cyclin D1 transgenic mice. Nature 369: 669–671.
Watts CK, Brady A, Sarcevic B, deFazio A, Musgrove EA, Sutherland RL . (1995). Antiestrogen inhibition of cell cycle progression in breast cancer cells in associated with inhibition of cyclin-dependent kinase activity and decreased retinoblastoma protein phosphorylation. Mol Endocrinol 9: 1804–1813.
Yeager T, Stadler W, Belair C, Puthenveettil J, Olopade O, Reznikoff C . (1995). Increased p16 levels correlate with pRb alterations in human urothelial cells. Cancer Res 55: 493–497.
Yu Q, Geng Y, Sicinski P . (2001). Specific protection against breast cancers by cyclin D1 ablation. Nature 411: 1017–1021.
Yu Q, Sicinska E, Geng Y, Ahnstrom M, Zagozdzon A, Kong Y et al. (2006). Requirement for CDK4 kinase function in breast cancer. Cancer Cell 9: 23–32.
Zagorski WA, Knudsen ES, Reed MF . (2007). Retinoblastoma deficiency increases chemosensitivity in lung cancer. Cancer Res 67: 8264–8273.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Dean, J., Thangavel, C., McClendon, A. et al. Therapeutic CDK4/6 inhibition in breast cancer: key mechanisms of response and failure. Oncogene 29, 4018–4032 (2010). https://doi.org/10.1038/onc.2010.154
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.154
Keywords
This article is cited by
-
The CDK4/6 inhibitor revolution — a game-changing era for breast cancer treatment
Nature Reviews Clinical Oncology (2024)
-
Discovery of IHMT-337 as a potent irreversible EZH2 inhibitor targeting CDK4 transcription for malignancies
Signal Transduction and Targeted Therapy (2023)
-
AKT/mTOR signaling modulates resistance to endocrine therapy and CDK4/6 inhibition in metastatic breast cancers
npj Precision Oncology (2023)
-
RB loss determines selective resistance and novel vulnerabilities in ER-positive breast cancer models
Oncogene (2022)
-
Targeting the Retinoblastoma/E2F repressive complex by CDK4/6 inhibitors amplifies oncolytic potency of an oncolytic adenovirus
Nature Communications (2022)