Abstract
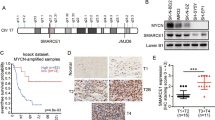
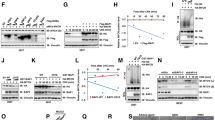
Recent advances in neuroblastoma (NB) research addressed that epigenetic alterations such as hypermethylation of promoter sequences, with consequent silencing of tumor-suppressor genes, can have significant roles in the tumorigenesis of NB. However, the exact role of epigenetic alterations, except for DNA hypermethylation, remains to be elucidated in NB research. In this paper, we clarified the direct binding of MYCN to Bmi1 promoter and upregulation of Bmi1 transcription by MYCN. Mutation introduction into an MYCN binding site in the Bmi1 promoter suggests that MYCN has more important roles in the transcription of Bmi1 than E2F-related Bmi1 regulation. A correlation between MYCN and polycomb protein Bmi1 expression was observed in primary NB tumors. Expression of Bmi1 resulted in the acceleration of proliferation and colony formation in NB cells. Bmi1-related inhibition of NB cell differentiation was confirmed by neurite extension assay and analysis of differentiation marker molecules. Intriguingly, the above-mentioned Bmi1-related regulation of the NB cell phenotype seems not to be mediated only by p14ARF/p16INK4a in NB cells. Expression profiling analysis using a tumor-specific cDNA microarray addressed the Bmi1-dependent repression of KIF1Bβ and TSLC1, which have important roles in predicting the prognosis of NB. Chromatin immunoprecipitation assay showed that KIF1Bβ and TSLC1 are direct targets of Bmi1 in NB cells. These findings suggest that MYCN induces Bmi1 expression, resulting in the repression of tumor suppressors through Polycomb group gene-mediated epigenetic chromosome modification. NB cell proliferation and differentiation seem to be partially dependent on the MYCN/Bmi1/tumor-suppressor pathways.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ando K, Ohira M, Ozaki T, Nakagawa A, Akazawa K, Suenaga Y et al. (2008). Expression of TSLC1, a candidate tumor suppressor gene mapped to chromosome 11q23, is downregulated in unfavorable neuroblastoma without promoter hypermethylation. Int J Cancer 123: 2087–2094.
Atsuta T, Fujimura S, Moriya H, Vidal M, Akasaka T, Koseki H . (2001). Production of monoclonal antibodies against mammalian Ring1B proteins. Hybridoma 20: 43–46.
Brodeur GM, Seeger RC, Schwab M, Varmus HE, Bishop JM . (1984). Amplification of N-myc in untreated human neuroblastomas correlates with advanced disease stage. Science 224: 1121–1124.
Bruggeman SW, Hulsman D, Tanger E, Buckle T, Blom M, Zevenhoven J et al. (2007). Bmi1 controls tumor development in an Ink4a/Arf-independent manner in a mouse model for glioma. Cancer Cell 12: 328–341.
Cao R, Tsukada Y, Zhang Y . (2005). Role of Bmi-1 and Ring1A in H2A ubiquitylation and Hox gene silencing. Mol Cell 20: 845–854.
Caron H . (1995). Allelic loss of chromosome 1 and additional chromosome 17 material are both unfavourable prognostic markers in neuroblastoma. Med Pediatr Oncol 24: 215–221.
Carén H, Ejeskär K, Fransson S, Hesson L, Latif F, Sjöberg RM et al. (2005). A cluster of genes located in 1p36 are down-regulated in neuroblastomas with poor prognosis, but not due to CpG island methylation. Mol Cancer 4: 10.
Cui H, Ma J, Ding J, Li T, Alam G, Ding HF . (2006). Bmi-1 regulates the differentiation and clonogenic self-renewal of I-type neuroblastoma cells in a concentration-dependent manner. J Biol Chem 281: 34696–34704.
Cui H, Hu B, Li T, Ma J, Alam G, Gunning WT et al. (2007). Bmi-1 is essential for the tumorigenicity of neuroblastoma cells. Am J Pathol 170: 1370–1378.
Easton J, Wei T, Lahti JM, Kidd VJ . (1998). Disruption of the cyclin D/cyclin-dependent kinase/INK4/retinoblastoma protein regulatory pathway in human neuroblastoma. Cancer Res 58: 2624–2632.
Esteller M . (2007). Cancer epigenomics: DNA methylomes and histone-modification maps. Nat Rev Genet 8: 286–298.
Fujimura Y, Isono K, Vidal M, Endoh M, Kajita H, Mizutani-Koseki Y et al. (2006). Distinct roles of Polycomb group gene products in transcriptionally repressed and active domains of Hoxb8. Development 133: 2371–2381.
Guney I, Wu S, Sedivy JM . (2006). Reduced c-Myc signaling triggers telomere-independent senescence by regulating Bmi-1 and p16(INK4a). Proc Natl Acad Sci USA 103: 3645–3650.
Hanahan D, Weinberg RA . (2000). The hallmarks of cancer. Cell 100: 57–70.
Iwama A, Oguro H, Negishi M, Kato Y, Morita Y, Tsukui H et al. (2004). Enhanced self-renewal of hematopoietic stem cells mediated by the polycomb gene product Bmi-1. Immunity 21: 843–851.
Jacobs JJ, Kieboom K, Marino S, DePinho RA, van Lohuizen M . (1999). The oncogene and Polycomb-group gene bmi-1 regulates cell proliferation and senescence through the ink4a locus. Nature 397: 164–168.
Jones PA, Baylin SB . (2002). The fundamental role of epigenetic events in cancer. Nat Rev Genet 3: 415–428.
Kamminga LM, Bystrykh LV, de Boer A, Houwer S, Douma J, Weersing E et al. (2005). The Polycomb group gene Ezh2 prevents hematopoietic stem cell exhaustion. Blood 107: 2170–2179.
Kamminga LM, de Haan G . (2006). Cellular memory and hematopoietic stem cell aging. Stem Cells 24: 1143–1149.
Kramps C, Strieder V, Sapetschnig A, Suske G, Lutz W . (2004). E2F and Sp1/Sp3 synergize but are not sufficient to activate the MYCN gene in neuroblastomas. J Biol Chem 279: 5110–5117.
Kurata K, Yanagisawa R, Ohira M, Kitagawa M, Nakagawara A, Kamijo T . (2008). Stress via p53 pathway causes apoptosis by mitochondrial Noxa upregulation in doxorubicin-treated neuroblastoma cells. Oncogene 27: 741–754.
Lessard J, Sauvageau G . (2003). Bmi-1 determines the proliferative capacity of normal and leukaemic stem cells. Nature 423: 255–260.
Leung C, Lingbeek M, Shakhova O, Liu J, Tanger E, Saremaslani P et al. (2004). Bmi1 is essential for cerebellar development and is overexpressed in human medulloblastomas. Nature 428: 337–341.
Lutz W, Stohr M, Schurmann J, Wenzel A, Lohr A, Schwab M . (1996). Conditional expression of N-myc in human neuroblastoma cells increases expression of alpha-prothymosin and ornithine decarboxylase and accelerates progression into S-phase early after mitogenic stimulation of quiescent cells. Oncogene 13: 803–812.
Molofsky AV, Pardal R, Iwashita T, Park IK, Clarke MF, Morrison SJ . (2003). Bmi-1 dependence distinguishes neural stem cell self-renewal from progenitor proliferation. Nature 425: 962–967.
Molofsky AV, Slutsky SG, Joseph NM, He S, Pardal R, Krishnamurthy J et al. (2006). Increasing p16INK4a expression decreases forebrain progenitors and neurogenesis during ageing. Nature 443: 448–452.
Munirajan AK, Ando K, Mukai A, Takahashi M, Suenaga Y, Ohira M et al. (2008). KIF1Bbeta functions as a haploinsufficient tumor suppressor gene mapped to chromosome 1p36.2 by inducing apoptotic cell death. J Biol Chem 283: 24426–24434.
Murakami Y . (2005). Involvement of a cell adhesion molecule, TSLC1/IGSF4, in human oncogenesis. Cancer Sci 96: 543–552.
Nowak K, Kerl K, Fehr D, Kramps C, Gessner C, Killmer K et al. (2006). BMI1 is a target gene of E2F-1 and is strongly expressed in primary neuroblastomas. Nucleic Acids Res 34: 1745–1754.
Ohira M, Oba S, Nakamura Y, Isogai E, Kaneko S, Nakagawa A et al. (2005). Expression profiling using a tumor-specific cDNA microarray predicts the prognosis of intermediate risk neuroblastomas. Cancer Cell 7: 337–350.
Orlando V, Strutt H, Paro R . (1997). Analysis of chromatin structure by in vivo formaldehyde cross-linking. Methods 11: 205–214.
Pietersen AM, van Lohuizen M . (2008). Stem cell regulation by polycomb repressors: postponing commitment. Curr Opin Cell Biol 20: 201–207.
Rajasekhar VK, Begemann M . (2007). Concise review: roles of polycomb group proteins in development and disease: a stem cell perspective. Stem Cells 25: 2498–2510.
Schwartz YB, Pirrotta V . (2008). Polycomb complexes and epigenetic states. Curr Opin Cell Biol 20: 266–273.
Sherr CJ . (2004). Principles of tumor suppression. Cell 116: 235–246.
Sparmann A, van Lohuizen M . (2006). Polycomb silencers control cell fate, development and cancer. Nat Rev Cancer 6: 846–856.
Strieder V, Lutz W . (2003). E2F proteins regulate MYCN expression in neuroblastomas. J Biol Chem 278: 2983–2989.
Sugino Y, Misawa A, Inoue J, Kitagawa M, Hosoi H, Sugimoto T et al. (2007). Epigenetic silencing of prostaglandin E receptor 2 (PTGER2) is associated with progression of neuroblastomas. Oncogene 26: 7401–7413.
Teitz T, Wei T, Valentine MB, Vanin EF, Grenet J, Valentine VA et al. (2000). Caspase 8 is deleted or silenced preferentially in childhood neuroblastomas with amplification of MYCN. Nat Med 6: 529–535.
Valk-Lingbeek ME, Bruggeman SW, van Lohuizen M . (2004). Stem cells and cancer; the polycomb connection. Cell 118: 409–418.
Viré E, Brenner C, Deplus R, Blanchon L, Fraga M, Didelot C et al. (2006). The Polycomb group protein EZH2 directly controls DNA methylation. Nature 439: 871–874.
Westemann F, Schwab M . (2002). Genetic parameters of neuroblastomas. Cancer Lett 184: 127–147.
Yan P, Mühlethaler A, Bourloud KB, Beck MN, Gross N . (2003). Hypermethylation-mediated regulation of CD44 gene expression in human neuroblastoma. Gene Chromosomes Cancer 36: 129–138.
Yang Q, Zage P, Kagan D, Tian Y, Seshadri R, Salwen HR et al. (2004). Association of epigenetic inactivation of RASSF1A with poor outcome in human neuroblastoma. Clin Cancer Res 10: 8493–8500.
Yang QW, Liu S, Tian Y, Salwen HR, Chlenski A, Weinstein J et al. (2003). Methylation-associated silencing of the thrombospondin-1 gene in human neuroblastoma. Cancer Res 63: 6299–6310.
Yang J, Chai L, Liu F, Fink LM, Lin P, Silberstein LE et al. (2007). Bmi-1 is a target gene for SALL4 in hematopoietic and leukemic cells. Proc Natl Acad Sci USA 104: 10494–10499.
Acknowledgements
We thank K Sakurai for technical assistance, and Daniel Mrozek, Medical English Service, for editorial assistance. This study was supported in part by a grant-in-aid from the Sankyo Foundation of Life Science, a grant-in-aid from the Ministry of Health, Labor, and Welfare for Third Term Comprehensive Control Research for Cancer, a grant-in-aid for Cancer Research (20–13) from the Ministry of Health, Labor, and Welfare of Japan, and a grant-in-aid from the Ministry of Education, Culture, Sports, Science and Technology, Japan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Oncogene website
Rights and permissions
About this article
Cite this article
Ochiai, H., Takenobu, H., Nakagawa, A. et al. Bmi1 is a MYCN target gene that regulates tumorigenesis through repression of KIF1B β and TSLC1 in neuroblastoma. Oncogene 29, 2681–2690 (2010). https://doi.org/10.1038/onc.2010.22
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2010.22
Keywords
This article is cited by
-
Discovery, characterization and potential roles of a novel NF-YAx splice variant in human neuroblastoma
Journal of Experimental & Clinical Cancer Research (2019)
-
MYCN and PRC1 cooperatively repress docosahexaenoic acid synthesis in neuroblastoma via ELOVL2
Journal of Experimental & Clinical Cancer Research (2019)
-
EZH2 regulates neuroblastoma cell differentiation via NTRK1 promoter epigenetic modifications
Oncogene (2018)
-
Epigenetic regulation of neuroblastoma development
Cell and Tissue Research (2018)
-
Combined Treatments with a Retinoid Receptor Agonist and Epigenetic Modulators in Human Neuroblastoma Cells
Molecular Neurobiology (2017)