Abstract
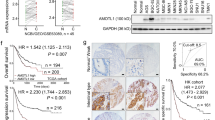
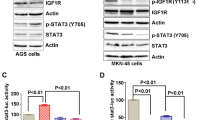
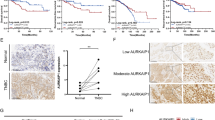
Aurora kinase A (AURKA) is located at 20q13, a region that is frequently amplified in gastric cancer. In this study, we have investigated the role of AURKA in regulating glycogen synthase kinase (GSK)-3β and β-catenin/TCF complex in gastric cancer cells. Our results demonstrate a significant increase in the phosphorylation of GSK-3β at Ser 9 following the overexpression of AURKA in AGS cells. The immunoprecipitation with antibodies specific for AURKA and GSK-3β indicated that the two proteins coexist in the same protein complex. The recombinant human AURKA protein phosphorylated the GSK-3β protein at Ser 9 in a concentration-dependent manner, in vitro. The phosphorylation of β-catenin (Ser33/37/Thr41) by GSK-3β is known to target β-catenin towards degradation. In line with our findings, the increase in phospho-GSK-3β level was accompanied by a significant decrease in β-catenin phosphorylation (Ser33/37/Thr41) and accumulation of β-catenin protein. The knockdown of AURKA reversed the phosphorylation of GSK-3β and the β-catenin protein levels. The immunofluorescence analysis demonstrated colocalization of AURKA and GSK-3β proteins and a significant increase in the nuclear β-catenin levels in cells overexpressing AURKA. The β-catenin/TCF transcription activity was measured using the pTopFlash and its mutant pFopFlash luciferase reporter vectors. Indeed, AURKA overexpression led to a significant increase in the pTopFlash reporter activity, whereas kinase dead AURKA mutant (D274A) had no effect. Consistent with these findings, we detected a significant mRNA up-regulation of several direct targets of the β-catenin/TCF transcription complex (cyclin D1, c-MYC, c-MYC-binding protein, CLDN1, FGF18 and vascular endothelial growth factor), and a two-fold increase in the proliferation rate in AURKA overexpressing cells. We conclude that the AURKA/GSK-3β interaction is important in regulating β-catenin, underscoring a novel oncogenic potential for AURKA in gastric tumorigenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Aberle H, Bauer A, Stappert J, Kispert A, Kemler R . (1997). Beta-catenin is a target for the ubiquitin-proteasome pathway. EMBO J 16: 3797–3804.
Behrens J, von Kries JP, Kuhl M, Bruhn L, Wedlich D, Grosschedl R et al. (1996). Functional interaction of beta-catenin with the transcription factor LEF-1. Nature 382: 638–642.
Bischoff JR, Anderson L, Zhu Y, Mossie K, Ng L, Souza B et al. (1998). A homologue of Drosophila aurora kinase is oncogenic and amplified in human colorectal cancers. EMBO J 17: 3052–3065.
Clements WM, Wang J, Sarnaik A, Kim OJ, MacDonald J, Fenoglio-Preiser C et al. (2002). Beta-catenin mutation is a frequent cause of Wnt pathway activation in gastric cancer. Cancer Res 62: 3503–3506.
Crawford HC, Fingleton BM, Rudolph-Owen LA, Goss KJ, Rubinfeld B, Polakis P et al. (1999). The metalloproteinase matrilysin is a target of beta-catenin transactivation in intestinal tumors. Oncogene 18: 2883–2891.
Damalas A, Ben-Ze'ev A, Simcha I, Shtutman M, Leal JF, Zhurinsky J et al. (1999a). Excess beta-catenin promotes accumulation of transcriptionally active p53. EMBO J 18: 3054–3063.
Damalas A, Ben-Ze'ev A, Simcha I, Shtutman M, Leal JF, Zhurinsky J et al. (1999b). Excess beta-catenin promotes accumulation of transcriptionally active p53. EMBO J 18: 3054–3063.
Dar AA, Zaika A, Piazuelo MB, Correa P, Koyama T, Belkhiri A et al. (2008). Frequent overexpression of Aurora kinase A in upper gastrointestinal adenocarcinomas correlates with potent antiapoptotic functions. Cancer 112: 1688–1698.
Earle CC, Maroun JA . (1999). Adjuvant chemotherapy after curative resection for gastric cancer in non-Asian patients: revisiting a meta-analysis of randomised trials. Eur J Cancer 35: 1059–1064.
El-Rifai W, Frierson HJ, Moskaluk C, Harper J, Petroni G, Bissonette E et al. (2001). Genetic differences between adenocarcinomas arising in Barrett's esophagus and gastric mucosa. Gastroenterology 121: 592–598.
El-Rifai W, Moskaluk CA, Abdrabbo MK, Harper J, Yoshida C, Riggins GJ et al. (2002). Gastric cancers overexpress S100A calcium-binding proteins. Cancer Res 62: 6823–6826.
Gamallo C, Palacios J, Moreno G, Calvo de Mora J, Suarez A, Armas A . (1999). beta-catenin expression pattern in stage I and II ovarian carcinomas: relationship with beta-catenin gene mutations, clinicopathological features, and clinical outcome. Am J Pathol 155: 527–536.
Giet R, Petretti C, Prigent C . (2005). Aurora kinases, aneuploidy and cancer, a coincidence or a real link? Trends Cell Biol 15: 241–250.
Giet R, Prigent C . (1999). Aurora/Ipl1p-related kinases, a new oncogenic family of mitotic serine-threonine kinases. J Cell Sci 112 (Part 21): 3591–3601.
Giles FJ, Cortes J, Jones D, Bergstrom D, Kantarjian H, Freedman SJ . (2007). MK-0457, a novel kinase inhibitor, is active in patients with chronic myeloid leukemia or acute lymphocytic leukemia with the T315I BCR-ABL mutation. Blood 109: 500–502.
Giles RH, van Es JH, Clevers H . (2003). Caught up in a Wnt storm: Wnt signaling in cancer. Biochim Biophys Acta 1653: 1–24.
He TC, Sparks AB, Rago C, Hermeking H, Zawel L, da Costa LT et al. (1998). Identification of c-MYC as a target of the APC pathway. Science 281: 1509–1512.
Hojilla CV, Kim I, Kassiri Z, Fata JE, Fang H, Khokha R . (2007). Metalloproteinase axes increase beta-catenin signaling in primary mouse mammary epithelial cells lacking TIMP3. J Cell Sci 120: 1050–1060.
Hu W, Kavanagh JJ, Deaver M, Johnston DA, Freedman RS, Verschraegen CF et al. (2005). Frequent overexpression of STK15/Aurora-A/BTAK and chromosomal instability in tumorigenic cell cultures derived from human ovarian cancer. Oncol Res 15: 49–57.
Jemal A, Siegel R, Ward E, Murray T, Xu J, Smigal C et al. (2006). Cancer statistics, 2006. CA Cancer J Clin 56: 106–130.
Kamada K, Yamada Y, Hirao T, Fujimoto H, Takahama Y, Ueno M et al. (2004). Amplification/overexpression of Aurora-A in human gastric carcinoma: potential role in differentiated type gastric carcinogenesis. Oncol Rep 12: 593–599.
Katayama H, Sasai K, Kawai H, Yuan ZM, Bondaruk J, Suzuki F et al. (2004). Phosphorylation by aurora kinase A induces Mdm2-mediated destabilization and inhibition of p53. Nat Genet 36: 55–62.
Kikuchi A, Kishida S, Yamamoto H . (2006). Regulation of Wnt signaling by protein–protein interaction and post-translational modifications. Exp Mol Med 38: 1–10.
Koh TJ, Bulitta CJ, Fleming JV, Dockray GJ, Varro A, Wang TC . (2000). Gastrin is a target of the beta-catenin/TCF-4 growth-signaling pathway in a model of intestinal polyposis. J Clin Invest 106: 533–539.
Miller JR, Hocking AM, Brown JD, Moon RT . (1999). Mechanism and function of signal transduction by the Wnt/beta-catenin and Wnt/Ca2+ pathways. Oncogene 18: 7860–7872.
Mitsiades CS, Mitsiades N, Koutsilieris M . (2004). The Akt pathway: molecular targets for anti-cancer drug development. Curr Cancer Drug Targets 4: 235–256.
Morin PJ, Sparks AB, Korinek V, Barker N, Clevers H, Vogelstein B et al. (1997). Activation of beta-catenin-Tcf signaling in colon cancer by mutations in beta-catenin or APC. Science 275: 1787–1790.
Nakatsuru S, Yanagisawa A, Ichii S, Tahara E, Kato Y, Nakamura Y et al. (1992). Somatic mutation of the APC gene in gastric cancer: frequent mutations in very well differentiated adenocarcinoma and signet-ring cell carcinoma. Hum Mol Genet 1: 559–563.
Ogawa E, Takenaka K, Katakura H, Adachi M, Otake Y, Toda Y et al. (2008). Perimembrane Aurora-A expression is a significant prognostic factor in correlation with proliferative activity in non-small-cell lung cancer (NSCLC). Ann Surg Oncol 15: 547–554.
Orford K, Crockett C, Jensen JP, Weissman AM, Byers SW . (1997). Serine phosphorylation-regulated ubiquitination and degradation of beta-catenin. J Biol Chem 272: 24735–24738.
Ouchi M, Fujiuchi N, Sasai K, Katayama H, Minamishima YA, Ongusaha PP et al. (2004). BRCA1 phosphorylation by Aurora-A in the regulation of G2 to M transition. J Biol Chem 279: 19643–19648.
Pap M, Cooper GM . (1998). Role of glycogen synthase kinase-3 in the phosphatidylinositol 3-Kinase/Akt cell survival pathway. J Biol Chem 273: 19929–19932.
Pennica D, Swanson TA, Welsh JW, Roy MA, Lawrence DA, Lee J et al. (1998). WISP genes are members of the connective tissue growth factor family that are up-regulated in wnt-1-transformed cells and aberrantly expressed in human colon tumors. Proc Natl Acad Sci USA 95: 14717–14722.
Polakis P . (1999). The oncogenic activation of beta-catenin. Curr Opin Genet Dev 9: 15–21.
Sherr CJ, Roberts JM . (1999). CDK inhibitors: positive and negative regulators of G1-phase progression. Genes Dev 13: 1501–1512.
Shtutman M, Zhurinsky J, Simcha I, Albanese C, D'Amico M, Pestell R et al. (1999). The cyclin D1 gene is a target of the beta-catenin/LEF-1 pathway. Proc Natl Acad Sci USA 96: 5522–5527.
Sourbier C, Lindner V, Lang H, Agouni A, Schordan E, Danilin S et al. (2006). The phosphoinositide 3-kinase/Akt pathway: a new target in human renal cell carcinoma therapy. Cancer Res 66: 5130–5142.
Sparks AB, Morin PJ, Vogelstein B, Kinzler KW . (1998). Mutational analysis of the APC/beta-catenin/Tcf pathway in colorectal cancer. Cancer Res 58: 1130–1134.
Tanaka T, Kimura M, Matsunaga K, Fukada D, Mori H, Okano Y . (1999). Centrosomal kinase AIK1 is overexpressed in invasive ductal carcinoma of the breast. Cancer Res 59: 2041–2044.
Terris B, Pineau P, Bregeaud L, Valla D, Belghiti J, Tiollais P et al. (1999). Close correlation between beta-catenin gene alterations and nuclear accumulation of the protein in human hepatocellular carcinomas. Oncogene 18: 6583–6588.
Tetsu O, McCormick F . (1999). Beta-catenin regulates expression of cyclin D1 in colon carcinoma cells. Nature 398: 422–426.
Tong T, Zhong Y, Kong J, Dong L, Song Y, Fu M et al. (2004). Overexpression of Aurora-A contributes to malignant development of human esophageal squamous cell carcinoma. Clin Cancer Res 10: 7304–7310.
Varis A, Puolakkainen P, Savolainen H, Kokkola A, Salo J, Nieminen O et al. (2001). DNA copy number profiling in esophageal Barrett adenocarcinoma: comparison with gastric adenocarcinoma and esophageal squamous cell carcinoma. Cancer Genet Cytogenet 127: 53–58.
Voeller HJ, Truica CI, Gelmann EP . (1998). Beta-catenin mutations in human prostate cancer. Cancer Res 58: 2520–2523.
Wang C, Li Z, Fu M, Bouras T, Pestell RG . (2004). Signal transduction mediated by cyclin D1: from mitogens to cell proliferation: a molecular target with therapeutic potential. Cancer Treat Res 119: 217–237.
Washington K, Chiappori A, Hamilton K, Shyr Y, Blanke C, Johnson D et al. (1998). Expression of beta-catenin, alpha-catenin, and E-cadherin in Barrett's esophagus and esophageal adenocarcinomas (In Process Citation). Mod Pathol 11: 805–813.
Yamada T, Takaoka AS, Naishiro Y, Hayashi R, Maruyama K, Maesawa C et al. (2000). Transactivation of the multidrug resistance 1 gene by T-cell factor 4/beta-catenin complex in early colorectal carcinogenesis. Cancer Res 60: 4761–4766.
Yang H, He L, Kruk P, Nicosia SV, Cheng JQ . (2006). Aurora-A induces cell survival and chemoresistance by activation of Akt through a p53-dependent manner in ovarian cancer cells. Int J Cancer 119: 2304–2312.
Zhang YJ, Jiang W, Chen CJ, Lee CS, Kahn SM, Santella RM et al. (1993). Amplification and overexpression of cyclin D1 in human hepatocellular carcinoma. Biochem Biophys Res Commun 196: 1010–1016.
Zhou H, Kuang J, Zhong L, Kuo WL, Gray JW, Sahin A et al. (1998). Tumour amplified kinase STK15/BTAK induces centrosome amplification, aneuploidy and transformation. Nat Genet 20: 189–193.
Acknowledgements
This study was supported by a pilot fund from the National Cancer Institute GI SPORE grant CA95103 and the Vanderbilt-Ingram Cancer Center support funds. The contents of this work are solely the responsibility of the authors and do not necessarily represent the official views of the National Cancer Institute or Vanderbilt University Medical Center.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Dar, A., Belkhiri, A. & El-Rifai, W. The aurora kinase A regulates GSK-3β in gastric cancer cells. Oncogene 28, 866–875 (2009). https://doi.org/10.1038/onc.2008.434
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2008.434
Keywords
This article is cited by
-
Chromosomal copy number amplification-driven Linc01711 contributes to gastric cancer progression through histone modification-mediated reprogramming of cholesterol metabolism
Gastric Cancer (2024)
-
The promotion action of AURKA on post-ischemic angiogenesis in diabetes-related limb ischemia
Molecular Medicine (2023)
-
Targeting AURKA in Cancer: molecular mechanisms and opportunities for Cancer therapy
Molecular Cancer (2021)
-
Therapeutically actionable signaling node to rescue AURKA driven loss of primary cilia in VHL-deficient cells
Scientific Reports (2021)
-
Aurora kinase A inhibition reverses the Warburg effect and elicits unique metabolic vulnerabilities in glioblastoma
Nature Communications (2021)