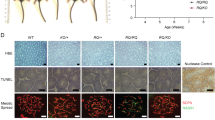
Abstract
Evidence exists that BRCA2 carriers may have an elevated risk of breast, ovarian, colon, prostate, and pancreatic cancer. In general, carriers are defined as individuals with protein truncating mutations within the BRCA2 gene. Many Brca2 knockout lines have been produced and characterized in the mouse. We previously produced a rat Brca2 knockout strain in which there is a nonsense mutation in exon 11 between BRC repeats 2 and 3, and a truncated protein is produced. Interestingly, while such a mutation in homozygous mice would lead to limited survival of approximately 3 months, the Brca2−/− rats are 100% viable and the vast majority live to over 1 year of age. Brca2−/− rats show a phenotype of growth inhibition and sterility in both sexes. Aspermatogenesis in the Brca2−/− rats is due to a failure of homologous chromosome synapsis. Long-term phenotypes include underdeveloped mammary glands, cataract formation and lifespan shortening due to the development of tumors and cancers in multiple organs. The establishment of the rat Brca2 knockout model provides a means to study the role of Brca2 in increasing cancer susceptibility and inducing a novel ocular phenotype not previously associated with this gene.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Allers T, Lichten M . (2001). Differential timing and control of noncrossover and crossover recombination during meiosis. Cell 106: 47–57.
Barlow C, Liyanage M, Moens PB, Tarsounas M, Nagashima K, Brown K et al. (1998). Atm deficiency results in severe meiotic disruption as early as leptonema of prophase 1. Development 125: 4007–4017.
Bignell G, Micklem G, Stratton MR, Ashworth A, Wooster R . (1997). The BRC repeats are conserved in mammalian BRCA2 proteins. Hum Mol Genet 6: 53–58.
Bigsby RM, Cardenas H, Caperell-Grant A, Grubbs CJ . (1999). Protective effects of estrogen in a rat model of age-related cataracts. Proc Natl Acad Sci USA 96: 9328–9332.
Bork P, Blomberg N, Nilges M . (1996). Internal repeats in the BRCA2 protein sequence. Nat Genet 13: 22–23.
Brenner S, Pepper D, Berns MW, Tan E, Brinkley BR . (1981). Kinetochore structure, duplication, and distribution in mammalian cells: analysis by human autoantibodies from scleroderma patients. J Cell Biol 91: 95–102.
Brown NM, Lamartiniere CA . (1995). Xenoestrogens alter mammary gland differentiation and cell proliferation in the rat. Environ Health Perspect 103: 708–713.
Connor F, Bertwistle D, Mee PJ, Ross GM, Swift S, Grigorieva E et al. (1997). Tumorigenesis and a DNA repair defect in mice with a truncating Brca2 mutation. Nat Genet 17: 423–430.
Cucinotta FA, Manuel FK, Jones J, Iszard G, Murrey J, Djojonegro B et al. (2001). Space radiation and cataracts in astronauts. Radiat Res 156: 460–466.
Davies AA, Masson JY, McIlwraith MJ, Stasiak AZ, Stasiak A, Venkitaraman AR et al. (2001). Role of BRCA2 in control of the RAD51 recombination and DNA repair protein. Mol Cell 7: 273–282.
Dobson MJ, Pearlman RE, Karaiskakis A, Spyropoulos B, Moens PB . (1994). Synaptonemal complex proteins: occurrence, epitope mapping and chromosome disjunction. J Cell Sci 107: 2749–2760.
Dynlacht JR, Tyree C, Valluri S, DesRosiers C, Caperell-Grant A, Mendonca MS et al. (2006). Effect of estrogen on radiation-induced cataractogenesis. Radiat Res 165: 9–15.
Eijpe M, Offenberg H, Goedecke W, Heyting C . (2000). Localisation of RAD50 and MRE11 in spermatocyte nuclei of mouse and rat. Chromosoma 109: 123–132.
Elliott B, Jasin M . (2002). Double-strand breaks and translocations in cancer. Cell Mol Life Sci 59: 373–385.
Friedman LS, Thistlethwaite FC, Patel KJ, Yu VP, Lee H, Venkitaraman AR et al. (1998). Thymic lymphomas in mice with a truncating mutation in Brca2. Cancer Res 58: 1338–1343.
Houghtaling S, Timmers C, Noll M, Finegold MJ, Jones SN, Meyn MS et al. (2003). Epithelial cancer in Fanconi anemia complementation group D2 (Fancd2) knockout mice. Genes Dev 17: 2021–2035.
Hunter N, Kleckner N . (2001). The single-end invasion: an asymmetric intermediate at the double-strand break to double-holliday junction transition of meiotic recombination. Cell 106: 59–70.
Keeney S . (2001). Mechanism and control of meiotic recombination initiation. Curr Top Dev Biol 52: 1–53.
Klein BE, Klein R, Lee KE . (1998). Incidence of age-related cataract: the Beaver Dam Eye Study. Arch Ophthalmol 116: 219–225.
Liu JG, Yuan L, Brundell E, Bjorkroth B, Daneholt B, Hoog C . (1996). Localization of the N-terminus of SCP1 to the central element of the synaptonemal complex and evidence for direct interactions between the N-termini of SCP1 molecules organized head-to-head. Exp Cell Res 226: 11–19.
Mahadevaiah SK, Turner JM, Baudat F, Rogakou EP, de Boer P, Blanco-Rodriguez J et al. (2001). Recombinational DNA double-strand breaks in mice precede synapsis. Nat Genet 27: 271–276.
McAllister KA, Bennett LM, Houle CD, Ward T, Malphurs J, Collins NK et al. (2002). Cancer susceptibility of mice with a homozygous deletion in the COOH-terminal domain of the Brca2 gene. Cancer Res 62: 990–994.
McAllister KA, Houle CD, Malphurs J, Ward T, Collins NK, Gersch W et al. (2006). Spontaneous and irradiation-induced tumor susceptibility in BRCA2 germline mutant mice and cooperative effects with a p53 germline mutation. Toxicol Pathol 34: 187–198.
Moens PB, Pearlman RE, Heng HH, Traut W . (1998). Chromosome cores and chromatin at meiotic prophase. Curr Top Dev Biol 37: 241–262.
Mohr U, Dungworth DL, Capen CC (eds). (1994). Pathobiology of the Aging Rat, Vol. 2. ILSI Press: Washington, DC, 93pp.
Moynahan ME . (2002). The cancer connection: BRCA1 and BRCA2 tumor suppression in mice and humans. Oncogene 21: 8994–9007.
Nakazawa M, Tawaratani T, Uchimoto H, Kawaminami A, Ueda M, Ueda A et al. (2001). Spontaneous neoplastic lesions in aged Sprague-Dawley rats. Exp Anim 50: 99–103.
Pellegrini L, Venkitaraman A . (2004). Emerging functions of BRCA2 in DNA recombination. Trends Biochem Sci 29: 310–316.
Roeder GS . (1997). Meiotic chromosomes: it takes two to tango. Genes Dev 11: 2600–2621.
Samuelson DJ, Haag JD, Lan H, Monson DM, Shultz MA, Kolman BD et al. (2003). Physical evidence of Mcs5, a QTL controlling mammary carcinoma susceptibility, in congenic rats. Carcinogenesis 24: 1445–1460.
Sarkisian CJ, Master SR, Huber LJ, Ha SI, Chodosh LA . (2001). Analysis of murine Brca2 reveals conservation of protein-protein interactions but differences in nuclear localization signals. J Biol Chem 276: 37640–37648.
Schulze W, Thoms F, Knuth UA . (1999). Testicular sperm extraction: comprehensive analysis with simultaneously performed histology in 1418 biopsies from 766 subfertile men. Hum Reprod Suppl 1: 82–96.
Sharan SK, Pyle A, Coppola V, Babus J, Swaminathan S, Benedict J et al. (2004). BRCA2 deficiency in mice leads to meiotic impairment and infertility. Development 131: 131–142.
Shivji MK, Venkitaraman AR . (2004). DNA recombination, chromosomal stability and carcinogenesis: insights into the role of BRCA2. DNA Repair (Amsterdam) 3: 835–843.
Siaud N, Dray E, Gy I, Gerard E, Takvorian N, Doutriaux MP . (2004). Brca2 is involved in meiosis in Arabidopsis thaliana as suggested by its interaction with Dmc1. Embo J 23: 1392–1401.
Villeneuve AM, Hillers KJ . (2001). Whence meiosis? Cell 106: 647–650.
Worgul BV, David J, Odrich S, Merriam Jr GR, Medvedovsky C, Merriam JC et al. (1991). Evidence of genotoxic damage in human cataractous lenses. Mutagenesis 6: 495–499.
Worgul BV, Smilenov L, Brenner DJ, Junk A, Zhou W, Hall EJ . (2002). Atm heterozygous mice are more sensitive to radiation-induced cataracts than are their wild-type counterparts. Proc Natl Acad Sci USA 99: 9836–9839.
Yoshida K, Miki Y . (2004). Role of BRCA1 and BRCA2 as regulators of DNA repair, transcription, and cell cycle in response to DNA damage. Cancer Sci 95: 866–871.
Zan Y, Haag JD, Chen KS, Shepel LA, Wigington D, Wang YR et al. (2003). Production of knockout rats using ENU mutagenesis and a yeast-based screening assay. Nat Biotechnol 21: 645–651.
Zhoucun A, Zhang S, Yang Y, Ma Y, Zhang W, Lin L . (2006). The common variant N372 H in BRCA2 gene may be associated with idiopathic male infertility with azoospermia or severe oligozoospermia. Eur J Obstet Gynecol Reprod Biol 124: 61–64.
Zickler D, Kleckner N . (1999). Meiotic chromosomes: integrating structure and function. Annu Rev Genet 33: 603–754.
Zierhut D, Lohr F, Schraube P, Huber P, Wenz F, Haas R et al. (2000). Cataract incidence after total-body irradiation. Int J Radiat Oncol Biol Phys 46: 131–135.
Acknowledgements
The authors thank Dr Ruth Sullivan (UW Research Animals Resource Center) for assistance in analyzing histopathology samples. This work was supported by the following grants, NIH CA106216 and DOD W81XWH-04-1-0436. Peti Thuwajit was supported by TRF-UW Postdoctoral Research Scholar #IPD4680004.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cotroneo, M., Haag, J., Zan, Y. et al. Characterizing a rat Brca2 knockout model. Oncogene 26, 1626–1635 (2007). https://doi.org/10.1038/sj.onc.1209960
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1209960
Keywords
This article is cited by
-
Rat models of human diseases and related phenotypes: a systematic inventory of the causative genes
Journal of Biomedical Science (2020)
-
Genetic variations of BRCA1 and BRCA2 genes in dogs with mammary tumours
Veterinary Research Communications (2014)
-
Systematic generation of in vivo G protein-coupled receptor mutants in the rat
The Pharmacogenomics Journal (2011)
-
Rat traps: filling the toolbox for manipulating the rat genome
Genome Biology (2010)
-
Comparative disease pattern of a patient with a novel BRCA2 truncation and knockout models for BRCA2
Breast Cancer Research and Treatment (2010)