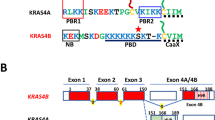
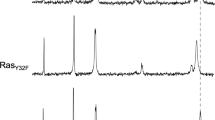
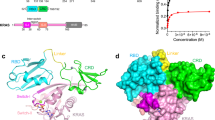
Abstract
We have investigated the productive interaction between the four mammalian Ras proteins (H-, N-, KA- and KB-Ras) and their activators, the mammalian exchange factors mSos1, GRF1 and GRP, by using a modified Saccharomyces cerevisiae whose growth is dependent on activation of a mammalian Ras protein by its activator. All four mammalian Ras proteins were activated with similar efficiencies by the individual exchange factors. The H-Ras mutant V103E, which is competent for membrane localization, nucleotide binding, intrinsic and stimulated GTPase activity as well as intrinsic exchange, was defective for activation by all factors tested, suggesting that the integrity of this residue is necessary for catalyzed exchange. However, when other H-Ras mutants were studied, some distinct sensitivities to the exchange factors were observed. GRP-mediated, but not mSos1-mediated, exchange was blocked in additional mutants, suggesting different structural requirements for GRP. Analysis of Ras-mediated gene activation in murine fibroblasts confirmed these results.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Anborgh PH, Qian X, Papageorge AG, Vass WC, DeClue JE, Lowy DR . 1999 Mol. Cell. Biol. 19: 4611–4622
Ballester R, Michaeli T, Ferguson K, Xu H-P, McCormick F, Wigler M . 1989 Cell 59: 681–686
Boriack-Sjodin PA, Margarit SM, Bar-Sagi D, Kuriyan J . 1998 Nature 394: 337–343
Bowtell D, Fu P, Simon M, Senior P . 1992 Proc. Natl. Acad. Sci. USA 89: 6511–6515
Broach JR . 1991 Trends Genetics 7: 28–33
Broach JR, Deschenes RJ . 1990 Adv. Cancer Res. 54: 79–139
Cen H, Papageorge AG, Zippel R, Lowy DR, Zhang K . 1992 EMBO J. 11: 4007–4015
Chardin P, Camonis JH, Gale NW, Van Aelst L, Schlessinger J, Wigler MH, Bar-Sagi D . 1993 Science 260: 1338–1343
Cox AD, Brtva TR, Lowe DG, Der CJ . 1994 Oncogene 9: 3281–3288
de Rooji J, Bos JL . 1997 Oncogene 14: 623–625
De Vendittis E, Vitelli A, Zahn R, Fasano O . 1986 EMBO J. 5: 3657–3663
Dhar R, Ellis RW, Shih TY, Oroszlan S, Shapiro B, Maizel J, Lowy D, Scolnick E . 1982 Science 217: 934–937
Ebinu JO, Bottorff DA, Chan EY, Stang SL, Dunn RJ, Stone JC . 1998 Science 280: 1082–1086
Fam NP, Fan WT, Wang Z, Zhang LJ, Chen H, Moran MF . 1997 Mol. Cell. Biol. 17: 1396–1406
Field J, Nikawa J, Broek D, MacDonald B, Rodgers L, Wilson IA, Lerner RA, Wigler M . 1988 Mol. Cell. Biol. 8: 2159–2165
Furth ME, Davis LJ, Fleurdelys B, Scolnick EM . 1982 J. Virol. 43: 294–304
Gibbs JB, Marshall MS, Scolnick EM, Dixon RAF, Vogel US . 1990 J. Biol. Chem. 265: 20437–20442
Gietz RD, Schiestl RH, Willems AR, Woods RA . 1995 Yeast 11: 355–360
Gotoh T, Niino Y, Tokuda M, Hatase O, Nakamura S, Matsuda M, Hattori S . 1997 J. Biol. Chem. 272: 18602–18607
Hall A, Brown R . 1985 Nucleic Acids Res. 13: 5255–5268
Hancock JF, Magee AI, Childs JE, Marshall CC . 1989 Cell 57: 1167–1177
Hancock JF, Paterson H, Marshall CJ . 1990 Cell 63: 133–139
Howe LR, Marshall CJ . 1993 Oncogene 8: 2583–2590
Huff SY, Quilliam LA, Cox AD, Der CJ . 1997 Oncogene 14: 133–143
Johnson L, Greenbaum D, Cichowski K, Mercer K, Murphy E, Schmitt E, Bronson RT, Umanoff H, Edelmann W, Kucherlapati R, Jacks T . 1997 Genes Dev. 11: 2468–2481
Jones MK, Jackson JH . 1998 J. Biol. Chem. 273: 1782–1787
Kaibuchi K, Mizuno T, Fujioka H, Yamamoto T, Kishi K, Fukumoto Y, Hori Y, Takai Y . 1991 Mol. Cell. Biol. 11: 2873–2880
Lamarche N, Tapon N, Stowers L, Burbelo PD, Aspenstrom P, Bridges T, Chant J, Hall A . 1996 Cell 87: 519–529
Leonardsen L, DeClue J, Lybæk H, Lowy D, Willumsen B . 1996 Oncogene 13: 2177–2187
Lowy DR, Willumsen BM . 1993 Ann. Rev. Biochem. 62: 851–891
Marais R, Light Y, Mason C, Paterson H, Olson MF, Marshall CJ . 1998 Science 280: 109–112
Martegani E, Vanoni M, Zippel R, Coccetti P, Brambilla R, Ferrari C, Sturani E, Alberghina L . 1992 EMBO J. 11: 2151–2157
Moreira JM, Holmberg S . 1998 EMBO J. 17: 6028–6038
Mosteller RD, Han JW, Broek D . 1994 Mol. Cell. Biol. 14: 1104–1112
Nielsen KH, Papageorge AG, Vass WC, Willumsen BM, Lowy DR . 1997 Mol. Cell. Biol. 17: 7132–7138
Orita S, Kaibuchi K, Kuroda S, Shimizu K, Nakanishi H, Takai Y . 1993 J. Biol. Chem. 268: 25542–25546
Pai EF, Krengel U, Petsko GA, Goody RS, Kabsch W, Wittinghofer A . 1990 EMBO J. 9: 2351–2359
Pilz RB, Willis RC, Boss GR . 1984 J. Biol. Chem. 259: 2927–2935
Quilliam L, Kato K, Rabun KM, Hisaka MM, Huff SY, Campbell-Burk S, Der CJ . 1994 Mol. Cell. Biol. 14: 1113–1121
Ridley AJ, Hall A . 1992 Cell 70: 389–399
Ridley AJ, Paterson HF, Johnston CL, Diekmann D, Hall A . 1992 Cell 70: 401–410
Rose MD, Winston F, Hieter H . 1990 Methods in Yeast Genetics A Laboratory Course Manual Cold Spring Harbor Laboratory Press
Segal M, Marbach I, Engelberg D, Simchen G, Levitzki A . 1992 J. Biol. Chem. 267: 22747–22751
Segal M, Marbach I, Willumsen BM, Levitzki A . 1995 Eur. J. Biochem. 228: 96–101
Segal M, Willumsen BM, Levitzki A . 1993 Proc. Natl. Acad. Sci. USA 90: 5564–5568
Shou C, Farnsworth CL, Neel BG, Feig LA . 1992 Nature 358: 351–354
Sigal IS, Gibbs JB, D'Alonzo JS, Scolnick EM . 1986 Proc. Natl. Acad. Sci. USA 83: 4725–4729
Tanaka K, Nakafuku M, Satoh T, Marshall MS, Gibbs JB, Matsumoto K, Kaziro Y, Tohe A . 1990 Cell 60: 803–807
Tatchell K . 1986 J. Bacteriol. 166: 364–367
Taylor SJ, Shalloway D . 1996 Curr. Biol. 6: 1621–1627
Thevelein JM . 1994 Yeast 10: 1753–1790
Toda T, Uno I, Ishikawa T, Powers S, Kataoka T, Broek D, Cameron S, Broach J, Matsumoto K, Wigler M . 1985 Cell 40: 27–36
Trahey M, Wong G, Halenbeck R, Rubinsfield B, Martin G, Ladner M, Long CM, Crosier WJ, Watt K, Koths K, McCormick F . 1988 Science 242: 1697–1700
Tsuchida N, Ryder T, Ohtsubo E . 1982 Science 217: 937–939
Velu TJ, Vass WC, Lowy DR, Tambourin PE . 1989 J. Virol. 63: 1384–1392
Wach A, Brachat A, Pohlmann R, Philippsen P . 1994 Yeast 10: 1793–1808
Willumsen BM . 1995 Methods Enzymol: Lipid Modifications of Proteins, Vol. 250. Casey PJ and Buss JE. (eds) Academic Press: Orlando, Fl pp. 269–284
Willumsen BM, Christensen A, Hubbert NL, Papageorge AG, Lowy DR . 1984a Nature 310: 583–586
Willumsen BM, Norris K, Papageorge AG, Hubbert NL, Lowy DR . 1984b EMBO J. 3: 2581–2585
Willumsen BM, Papageorge AG, Kung H-F, Bekesi E, Robins T, Johnsen M, Vass WC, Lowy DR . 1986 Mol. Cell. Biol. 6: 2646–2654
Willumsen BM, Vass WC, Velu TJ, Papageorge AG, Schiller J, Lowy DR . 1991 Mol. Cell. Biol. 11: 6026–6033
Acknowledgements
We thank Marianne Knudsen and Rikke Ingvorsen for technical assistance and Jeffrey DeClue for constructing the KB gene. The introduction to S. cerevisiae manipulations by Steen Holmberg is gratefully acknowledged, and we thank James Stone, Adrienne Cox, Alan Hall, Channing Der, and Douglas Lowy for providing materials; Douglas Lowy and Malene Hansen for reading the manuscript, and Kaare Teilum for help with Figure 6. This work was supported by NIH Grant CA41086 to JRB, and grants 97 100 13 from the Danish Cancer Society and 9600821 from the Danish Medical Research council to BM Willumsen.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nielsen, K., Gredsted, L., Broach, J. et al. Sensitivity of wild type and mutant ras alleles to Ras specific exchange factors: Identification of factor specific requirements. Oncogene 20, 2091–2100 (2001). https://doi.org/10.1038/sj.onc.1204306
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1204306