Abstract
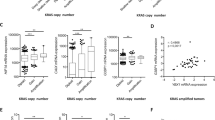
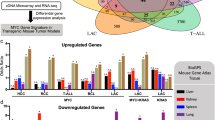
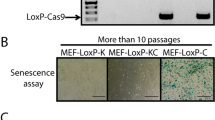
The adenovirus type 5 (Ad5) E4orf6 protein promotes focus formation of primary baby rat kidney (BRK) cells in cooperation with Ad5 E1 proteins. This activity is most likely related to the ability of the E4orf6 protein to bind to p53 and modulate its tumor suppressor functions. In this study we report that transformed BRK cells that stably express E4orf6 in addition to E1A and E1B (ABS cells) displayed multiple additional properties commonly associated with a high grade of oncogenic transformation compared to cells expressing only E1A and E1B (AB cells). These properties included morphological alterations, markedly enhanced growth rates and growth to much higher saturation densities. Following injection into nude mice ABS-derived tumors exhibited accelerated growth and, based on histopathological criteria, proofed to be much more malignant compared to tumors generated by AB cells. Interestingly, these highly transformed properties of ABS cells correlated with a dramatic reduction of p53 steady-state levels which inversely correlated with E4orf6 expression. From these results we conclude that expression of the Ad5 E4orf6 protein (i) confers additional transformed in vitro properties to primary rat cells expressing the Ad5 E1 proteins, and (ii) increases the tumorigenic and malignant potential of these cells in vivo. Our data suggest that the Ad5 E4orf6 protein enhances the intrinsic ability of E1-transformed rat cells to grow in a neoplastic state by completely inactivating p53 tumor suppressor function in combination with the E1A and E1B proteins.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Barker DD and Berk AJ. . 1987 Virology 156: 107–121.
Bernards R, de Leeuw MG, Houweling A and van der Eb AJ. . 1986 Virology 150: 126–139.
Bischoff JR, Kirn DH, Williams A, Heise C, Horn S, Muna M, Ng L, Nye JA, Sampson Johannes A, Fattaey A and McCormick F. . 1996 Science 274: 373–376.
Blair-Zajdel ME and Blair GE. . 1988 Oncogene 2: 579–584.
Chen PL, Chen Y, Bookstein R and Lee WH. . 1990 Science 250: 1576–1580.
Chiou S-K and White E. . 1997 J. Virol. 71: 3515–3525.
Debbas M and White E. . 1993 Genes & Dev. 7: 546–554.
Dittmer D, Pati S, Zambetti G, Chu S, Teresky AK, Moore M, Finlay C and Levine AJ. . 1993 Nature Genet. 4: 42–45.
Dobner T, Horikoshi N, Rubenwolf S and Shenk T. . 1996 Science 272: 1470–1473.
El-Deiry WS, Tokino T, Velculescu VE, Levy DB, Parsons R, Trent JM, Lin D, Mercer WE, Kinzler KW and Vogelstein B. . 1993 Cell 75: 817–825.
Goodrum FD, Shenk T and Ornelles DA. . 1996 J. Virol. 70: 6323–6335.
Graham FL. . 1984 The adenoviruses. Ginsberg HS. (ed.).. Plenum Press: New York pp.339–398.
Graham FL and van der Eb AJ. . 1973 Virology 52: 456–467.
Grand RJ, Grant ML and Gallimore PH. . 1994 Virology 203: 229–240.
Harlow E, Pim DC and Crawford LV. . 1981 J. Virol. 37: 564–573.
Harlow E, Franza BR Jr and Schley C. . 1985 J. Virol. 55: 533–546.
Harper JW, Adami GR, Wei N, Keyomarsi K and Elledge SJ. . 1993 Cell 75: 805–816.
Harvey M, Sands AT, Weiss RS, Hegi ME, Wiseman RW, Pantazis P, Giovanella BC, Tainsky MA, Bradley A and Donehower LA. . 1993 Oncogene 8: 2457–2467.
Heise C, Sampson Johannes A, Williams A, McCormick F, Von Hoff DD and Kirn DH. . 1997 Nat. Med. 3: 639–645.
Houweling A, van den Elsen PJ and van der Eb AJ. . 1980 Virology 105: 537–550.
Hundley JE, Koester SK, Troyer DA, Hilsenbeck SG, Subler MA and Windle JJ. . 1997 Mol. Cell. Biol. 17: 723–731.
Kemp CJ, Donehower LA, Bradley A and Balmain A. . 1993 Cell 74: 813–822.
Lin J, Chen J, Elenbaas B and Levine AJ. . 1994 Genes & Dev. 8: 1235–1246.
Logan J, Pilder S and Shenk T. . 1984 Cancer Cells 2: 527–532.
Lowe SW and Ruley HE. . 1993 Genes & Dev. 7: 535–545.
Marton MJ, Baim SB, Ornelles DA and Shenk T. . 1990 J. Virol. 64: 2345–2359.
McLorie W, McGlade CJ, Takayesu D and Branton PE. . 1991 J. Gen. Virol. 72: 1467–1471.
Moore M, Horikoshi N and Shenk T. . 1996 Proc. Natl. Acad. Sci. USA 93: 11295–11301.
Moran E. . 1994 Sem. Virol. 5: 327–340.
Neill SD, Hemstrom C, Virtanen A and Nevins JR. . 1990 Proc. Natl. Acad. Sci. USA 87: 2008–2012.
Nevels M, Rubenwolf S, Spruss T, Wolf H and Dobner T. . 1997 Proc. Natl. Acad. Sci. USA 94: 1206–1211.
Nevins JR and Vogt PK. . 1996 Virology, Vol.1. Fields BN, Knipe DM and Howley PM (eds).. Lippincott-Raven: New York pp.301–343.
Pietenpol JA, Tokino T, Thiagalingam S, El-Deiry WS, Kinzler KW and Vogelstein BS. . 1994 Proc. Natl. Acad. Sci. USA 91: 1998–2002.
Querido E, Marcellus R, Lai A, Rachel C, Teodoro JG, Ketner G and Branton PE. . 1997a J. Virol. 71: 3788–3798.
Querido E, Teodoro JG and Branton PE. . 1997b J. Virol. 71: 3526–3533.
Rubenwolf S, Schütt H, Nevels M, Wolf H and Dobner T. . 1997 J. Virol. 71: 1115–1123.
Sabbatini P, Lin J, Levine AJ and White E. . 1995 Genes & Dev. 9: 2184–2192.
Sarnow P, Ho YS, Williams J and Levine AJ. . 1982a Cell 28: 387–394.
Sarnow P, Sullivan CA and Levine AJ. . 1982b Virology 120: 510–517.
Sarnow P, Hearing P, Anderson CW, Halbert DN, Shenk T and Levine AJ. . 1984 J. Virol. 49: 692–700.
Shenk T. . 1996 Virology, Vol.2. Fields BN, Knipe DM and Howley PM. (eds).. Lippincott-Raven: New York pp.2111–2148.
Shiroki K, Maruyama K, Saito I, Fukui Y and Shimojo H. . 1981 J. Virol. 38: 1048–1054.
Spruß T, Schlemmer R, Bernhardt G, Wiesenmeyer F and Baumann O. . 1996 Immunodeficient Animals: Model for Cancer Research. Contrib. Oncol., Vol.51. Arnold W, Köpf-Maier P and Micheel B. (eds).. Karger: Basel pp.12–18.
Symonds H, Krall L, Remington L, Saenz-Robles M, Lowe S, Jacks R and Van Dyke T. . 1994 Cell 78: 703–711.
Tooze J. . 1981 Cold Spring Harbor, Vol. 2nd ed. Cold Spring Harbor Laboratory: New York pp.943–1054.
Ullrich SJ, Anderson CW, Mercer WE and Appella E. . 1992 J. Biol. Chem. 267: 15259–15262.
van den Heuvel SJL, van Laar T, Kast WM, Melief CJM, Zantema A and van der Eb AJ. . 1990 EMBO J. 9: 2621–2629.
White E and Cipriani R. . 1990 Mol. Cell. Biol. 10: 120–130.
White E. . 1994 Sem. Virol. 5: 341–348.
White E. . 1996 Genes & Dev. 10: 1–15.
Wolf D, Harris N and Rotter V. . 1984 Cell 38: 119–126.
Xiong Y, Hannon G, Zhang H, Casso D, Kobayashi R and Beach D. . 1993 Nature 366: 701–704.
Yew PR and Berk AJ. . 1992 Nature 357: 82–85.
Yew PR, Liu X and Berk AJ. . 1994 Genes & Dev. 8: 190–202.
Zantema A, Fransen JA, Davis OA, Ramaekers FC, Vooijs GP, DeLeys B and van der Eb AJ. . 1985 Virology 142: 44–58.
Acknowledgements
We thank D Lane (University of Dundee) for antibody CM-1; F Wiesenmeyer and O Baumann (Universität Regensburg) for animal work. This work was supported by the Deutsche Forschungsgemeinschaft (Do 343/4-1).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Nevels, M., Spruss, T., Wolf, H. et al. The adenovirus E4orf6 protein contributes to malignant transformation by antagonizing E1A-induced accumulation of the tumor suppressor protein p53. Oncogene 18, 9–17 (1999). https://doi.org/10.1038/sj.onc.1202284
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1202284
Keywords
This article is cited by
-
Sp100A is a tumor suppressor that activates p53-dependent transcription and counteracts E1A/E1B-55K-mediated transformation
Oncogene (2016)
-
Cell polarity proteins: common targets for tumorigenic human viruses
Oncogene (2008)
-
Blockage of CRM1-dependent nuclear export of the adenovirus type 5 early region 1B 55-kDa protein augments oncogenic transformation of primary rat cells
Oncogene (2005)
-
Nuclear export of adenovirus E4orf6 protein is necessary for its ability to antagonize apoptotic activity of BH3-only proteins
Oncogene (2003)
-
Adenovirus early E4 genes in viral oncogenesis
Oncogene (2001)