Abstract
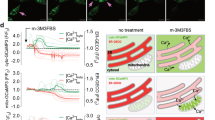
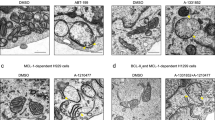
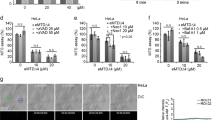
In several different cell lines, Bcl-2 prevents the induction of apoptosis (DNA fragmentation, PARP cleavage, phosphatidylserine exposure) by the pro-oxidant ter-butylhydroperoxide (t-BHP) but has no cytoprotective effect when apoptosis is induced by the thiol crosslinking agent diazenedicarboxylic acid bis 5N,N-dimethylamide (diamide). Both t-BHP and diamide cause a disruption of the mitochondrial transmembrane potential ΔΨm that is not inhibited by the broad spectrum caspase inhibitor Z-VAD.fmk, although Z-VAD.fmk does prevent nuclear DNA fragmentation and poly(ADP-ribose) polymerase cleavage in these models. Bcl-2 stabilizes the ΔΨm of t-BHP-treated cells but has no inhibitory effect on the ΔΨm collapse induced by diamide. As compared to normal controls, isolated mitochondria from Bcl-2 overexpressing cells are relatively resistant to the induction of ΔΨm disruption by t-BHP in vitro. Such Bcl-2 overexpressing mitochondria also fail to release apoptosis-inducing factor (AIF) and cytochrome c from the intermembrane space, whereas control mitochondria not overexpressing Bcl-2 do liberate AIF and cytochrome c in response to t-BHP. In contrast, Bcl-2 does not confer protection against diamide-triggered ΔΨm collapse and the release of AIF and cytochrome c. This indicates that Bcl-2 suppresses the permeability transition (PT) and the associated release of intermembrane proteins induced by t-BHP but not by diamide. To further investigate the mode of action of Bcl-2, semi-purified PT pore complexes were reconstituted in liposomes in a cell-free, organelle-free system. Recombinant Bcl-2 or Bcl-XL proteins augment the resistance of reconstituted PT pore complexes to pore opening induced by t-BHP. In contrast, mutated Bcl-2 proteins which have lost their cytoprotective potential also lose their PT-modulatory capacity. Again, Bcl-2 fails to confer protection against diamide in this experimental system. The reconstituted PT pore complex itself cannot release cytochrome c encapsulated into liposomes. Altogether these data suggest that pro-oxidants, thiol-reactive agents, and Bcl-2 can regulate the PT pore complex in a direct fashion, independently from their effects on cytochrome c. Furthermore, our results suggest a strategy for inducing apoptosis in cells overexpressing apoptosis-inhibitory Bcl-2 analogs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Zamzami, N., Marzo, I., Susin, S. et al. The thiol crosslinking agent diamide overcomes the apoptosis-inhibitory effect of Bcl-2 by enforcing mitochondrial permeability transition. Oncogene 16, 1055–1063 (1998). https://doi.org/10.1038/sj.onc.1201864
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/sj.onc.1201864
Keywords
This article is cited by
-
α-Tocopheryl succinate promotes selective cell death induced by vitamin K3 in combination with ascorbate
British Journal of Cancer (2010)
-
Prevention of premature senescence requires JNK regulation of Bcl-2 and reactive oxygen species
Oncogene (2010)
-
Novel Strategy for Treatment of Pulmonary Arterial Hypertension: Enhancement of Apoptosis
Lung (2010)
-
Potassium channels in the regulation of pulmonary artery smooth muscle cell proliferation and apoptosis: pharmacotherapeutic implications
British Journal of Pharmacology (2008)
-
Mitochondrial Medicine: Pharmacological targeting of mitochondria in disease
British Journal of Pharmacology (2007)