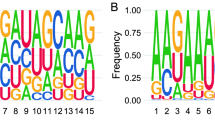
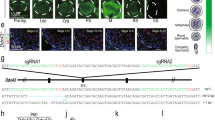
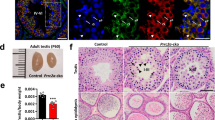
Abstract
Germ cells implement elaborate mechanisms to protect their genetic material and to regulate gene expression during differentiation. Piwi proteins bind Piwi-interacting RNAs (piRNAs), small germline RNAs whose biogenesis and functions are still largely elusive. We used high-throughput sequencing after cross-linking and immunoprecipitation (HITS-CLIP) coupled with RNA–sequencing (RNA-seq) to characterize the genome-wide target RNA repertoire of Mili (Piwil2) and Miwi (Piwil1), two Piwi proteins expressed in mouse postnatal testis. We report the in vivo pathway of primary piRNA biogenesis and implicate distinct nucleolytic activities that process Piwi-bound precursor transcripts. Our studies indicate that pachytene piRNAs are the end products of RNA processing. HITS-CLIP demonstrated that Miwi binds spermiogenic mRNAs directly, without using piRNAs as guides, and independent biochemical analyses of testis mRNA ribonucleoproteins (mRNPs) established that Miwi functions in the formation of mRNP complexes that stabilize mRNAs essential for spermiogenesis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Cox, D.N. et al. A novel class of evolutionarily conserved genes defined by piwi are essential for stem cell self-renewal. Genes Dev. 12, 3715–3727 (1998).
Deng, W. & Lin, H. miwi, a murine homolog of piwi, encodes a cytoplasmic protein essential for spermatogenesis. Dev. Cell 2, 819–830 (2002).
Kuramochi-Miyagawa, S. et al. Mili, a mammalian member of piwi family gene, is essential for spermatogenesis. Development 131, 839–849 (2004).
Boswell, R.E. & Mahowald, A.P. tudor, a gene required for assembly of the germ plasm in Drosophila melanogaster. Cell 43, 97–104 (1985).
Chen, C., Nott, T.J., Jin, J. & Pawson, T. Deciphering arginine methylation: Tudor tells the tale. Nat. Rev. Mol. Cell Biol. 12, 629–642 (2011).
Kirino, Y. et al. Arginine methylation of Piwi proteins catalysed by dPRMT5 is required for Ago3 and Aub stability. Nat. Cell Biol. 11, 652–658 (2009).
Nishida, K.M. et al. Functional involvement of Tudor and dPRMT5 in the piRNA processing pathway in Drosophila germlines. EMBO J. 28, 3820–3831 (2009).
Vagin, V.V. et al. Proteomic analysis of murine Piwi proteins reveals a role for arginine methylation in specifying interaction with Tudor family members. Genes Dev. 23, 1749–1762 (2009).
Kirino, Y. et al. Arginine methylation of Aubergine mediates Tudor binding and germ plasm localization. RNA 16, 70–78 (2010).
Yokota, S. Historical survey on chromatoid body research. Acta Histochem. Cytochem. 41, 65–82 (2008).
Lau, N.C. et al. Characterization of the piRNA complex from rat testes. Science 313, 363–367 (2006).
Aravin, A. et al. A novel class of small RNAs bind to MILI protein in mouse testes. Nature 442, 203–207 (2006).
Girard, A., Sachidanandam, R., Hannon, G.J. & Carmell, M.A. A germline-specific class of small RNAs binds mammalian Piwi proteins. Nature 442, 199–202 (2006).
Grivna, S.T., Beyret, E., Wang, Z. & Lin, H. A novel class of small RNAs in mouse spermatogenic cells. Genes Dev. 20, 1709–1714 (2006).
Siomi, M.C., Sato, K., Pezic, D. & Aravin, A.A. PIWI-interacting small RNAs: the vanguard of genome defence. Nat. Rev. Mol. Cell Biol. 12, 246–258 (2011).
Kawaoka, S., Izumi, N., Katsuma, S. & Tomari, Y. 3′ End formation of PIWI-interacting RNAs in vitro. Mol. Cell 43, 1015–1022 (2011).
Pillai, R.S. & Chuma, S. piRNAs and their involvement in male germline development in mice. Dev. Growth Differ. 54, 78–92 (2012).
Besse, F. & Ephrussi, A. Translational control of localized mRNAs: restricting protein synthesis in space and time. Nat. Rev. Mol. Cell Biol. 9, 971–980 (2008).
Heidaran, M.A., Showman, R.M. & Kistler, W.S. A cytochemical study of the transcriptional and translational regulation of nuclear transition protein 1 (TP1), a major chromosomal protein of mammalian spermatids. J. Cell Biol. 106, 1427–1433 (1988).
Kleene, K.C. Patterns of translational regulation in the mammalian testis. Mol. Reprod. Dev. 43, 268–281 (1996).
Carmell, M.A. et al. MIWI2 is essential for spermatogenesis and repression of transposons in the mouse male germline. Dev. Cell 12, 503–514 (2007).
Reuter, M. et al. Loss of the Mili-interacting Tudor domain-containing protein-1 activates transposons and alters the Mili-associated small RNA profile. Nat. Struct. Mol. Biol. 16, 639–646 (2009).
Shoji, M. et al. The TDRD9-MIWI2 complex is essential for piRNA-mediated retrotransposon silencing in the mouse male germline. Dev. Cell 17, 775–787 (2009).
Kuramochi-Miyagawa, S. et al. MVH in piRNA processing and gene silencing of retrotransposons. Genes Dev. 24, 887–892 (2010).
Frost, R.J. et al. MOV10L1 is necessary for protection of spermatocytes against retrotransposons by Piwi-interacting RNAs. Proc. Natl. Acad. Sci. USA 107, 11847–11852 (2010).
Zheng, K. et al. Mouse MOV10L1 associates with Piwi proteins and is an essential component of the Piwi-interacting RNA (piRNA) pathway. Proc. Natl. Acad. Sci. USA 107, 11841–11846 (2010).
De Fazio, S. et al. The endonuclease activity of Mili fuels piRNA amplification that silences LINE1 elements. Nature 480, 259–263 (2011).
Tanaka, T. et al. Tudor domain containing 7 (Tdrd7) is essential for dynamic ribonucleoprotein (RNP) remodeling of chromatoid bodies during spermatogenesis. Proc. Natl. Acad. Sci. USA 108, 10579–10584 (2011).
Vasileva, A., Tiedau, D., Firooznia, A., Muller-Reichert, T. & Jessberger, R. Tdrd6 is required for spermiogenesis, chromatoid body architecture, and regulation of miRNA expression. Curr. Biol. 19, 630–639 (2009).
Chi, S.W., Zang, J.B., Mele, A. & Darnell, R.B. Argonaute HITS-CLIP decodes microRNA-mRNA interaction maps. Nature 460, 479–486 (2009).
Robine, N. et al. A broadly conserved pathway generates 3′UTR-directed primary piRNAs. Curr. Biol. 19, 2066–2076 (2009).
Gunawardane, L.S. et al. A slicer-mediated mechanism for repeat-associated siRNA 5′ end formation in Drosophila. Science 315, 1587–1590 (2007).
Horwich, M.D. et al. The Drosophila RNA methyltransferase, DmHen1, modifies germline piRNAs and single-stranded siRNAs in RISC. Curr. Biol. 17, 1265–1272 (2007).
Kirino, Y. & Mourelatos, Z. The mouse homolog of HEN1 is a potential methylase for Piwi-interacting RNAs. RNA 13, 1397–1401 (2007).
Simon, B. et al. Recognition of 2′-O-methylated 3′-end of piRNA by the PAZ domain of a Piwi protein. Structure 19, 172–180 (2011).
Tian, Y., Simanshu, D.K., Ma, J.B. & Patel, D.J. Structural basis for piRNA 2′-O-methylated 3′-end recognition by Piwi PAZ (Piwi/Argonaute/Zwille) domains. Proc. Natl. Acad. Sci. USA 108, 903–910 (2011).
Kirino, Y. & Mourelatos, Z. Site-specific crosslinking of human microRNPs to RNA targets. RNA 14, 2254–2259 (2008).
Reuter, M. et al. Miwi catalysis is required for piRNA amplification-independent LINE1 transposon silencing. Nature 480, 264–267 (2011).
Zhang, C. & Darnell, R.B. Mapping in vivo protein-RNA interactions at single-nucleotide resolution from HITS-CLIP data. Nat. Biotechnol. 29, 607–614 (2011).
Herbert, T.P. & Hecht, N.B. The mouse Y-box protein, MSY2, is associated with a kinase on non-polysomal mouse testicular mRNAs. Nucleic Acids Res. 27, 1747–1753 (1999).
Bagarova, J., Chowdhury, T.A., Kimura, M. & Kleene, K.C. Identification of elements in the Smcp 5′ and 3′ UTR that repress translation and promote the formation of heavy inactive mRNPs in spermatids by analysis of mutations in transgenic mice. Reproduction 140, 853–864 (2010).
Kleene, K.C., Bagarova, J., Hawthorne, S.K. & Catado, L.M. Quantitative analysis of mRNA translation in mammalian spermatogenic cells with sucrose and Nycodenz gradients. Reproductive biology and endocrinology 8, 155 (2010).
Giorgini, F., Davies, H.G. & Braun, R.E. Translational repression by MSY4 inhibits spermatid differentiation in mice. Development 129, 3669–3679 (2002).
Kimura, M., Ishida, K., Kashiwabara, S. & Baba, T. Characterization of two cytoplasmic poly(A)-binding proteins, PABPC1 and PABPC2, in mouse spermatogenic cells. Biol. Reprod. 80, 545–554 (2009).
Yang, J., Medvedev, S., Reddi, P.P., Schultz, R.M. & Hecht, N.B. The DNA/RNA-binding protein MSY2 marks specific transcripts for cytoplasmic storage in mouse male germ cells. Proc. Natl. Acad. Sci. USA 102, 1513–1518 (2005).
Yang, J. et al. Absence of the DNA-/RNA-binding protein MSY2 results in male and female infertility. Proc. Natl. Acad. Sci. USA 102, 5755–5760 (2005).
Huang, H. et al. piRNA-associated germline nuage formation and spermatogenesis require MitoPLD profusogenic mitochondrial-surface lipid signaling. Dev. Cell 20, 376–387 (2011).
Watanabe, T. et al. MITOPLD is a mitochondrial protein essential for nuage formation and piRNA biogenesis in the mouse germline. Dev. Cell 20, 364–375 (2011).
Hermo, L., Pelletier, R.M., Cyr, D.G. & Smith, C.E. Surfing the wave, cycle, life history, and genes/proteins expressed by testicular germ cells. Part 2: changes in spermatid organelles associated with development of spermatozoa. Microsc. Res. Tech. 73, 279–319 (2010).
Haraguchi, C.M. et al. Chromatoid bodies: aggresome-like characteristics and degradation sites for organelles of spermiogenic cells. J. Histochem. Cytochem. 53, 455–465 (2005).
Austin, C.R. & Sapsford, C.S. The development of the rat spermatid. J. R. Microsc. Soc. 71, 397–406 (1951).
Dostie, J. & Dreyfuss, G. Translation is required to remove Y14 from mRNAs in the cytoplasm. Curr. Biol. 12, 1060–1067 (2002).
Faehnle, C.R. & Joshua-Tor, L. Argonaute MID domain takes centre stage. EMBO Rep. 11, 564–565 (2010).
Liu, X., Jin, D.Y., McManus, M.T. & Mourelatos, Z. Precursor microRNA-programmed silencing complex assembly pathways in mammals. Mol. Cell 46, 507–517 (2012).
Oko, R.J., Jando, V., Wagner, C.L., Kistler, W.S. & Hermo, L.S. Chromatin reorganization in rat spermatids during the disappearance of testis-specific histone, H1t, and the appearance of transition proteins TP1 and TP2. Biol. Reprod. 54, 1141–1157 (1996).
Alfonso, P.J. & Kistler, W.S. Immunohistochemical localization of spermatid nuclear transition protein 2 in the testes of rats and mice. Biol. Reprod. 48, 522–529 (1993).
Acknowledgements
We are grateful to J. Schug for assistance with Illumina analysis approaches in the early phase of this project and to S. Fayngerts, S. Rafail and members of the Mourelatos laboratory for technical help and lively discussions. The antibody to Tnp2 was a gift from W.S. Kistler (University of South Carolina, Columbia). This work was supported by the US National Institute of General Medical Sciences of the National Institutes of Health (R01GM0720777) and in part by Penn Institute for Regenerative Medicine and Perelman School of Medicine, University of Pennsylvania grants to Z.M. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health. Work in the Gregory lab was supported in part by grant #IRG-78-002-30 from the American Cancer Society as well as the Penn Genome Frontiers Institute and a grant with the Pennsylvania Department of Health. The Department of Health specifically disclaims responsibility for any analyses, interpretations or conclusions.
Author information
Authors and Affiliations
Contributions
A.V. and Z.M. conceived and directed experiments. A.V. performed HITS-CLIP, RNA-seq and Nycodenz experiments; Y.K. performed overexpression of Mili and Miwi and in vitro cross-linking experiments. Q.Z., M.M., P.A., A.V., B.D.G., Z.M. analyzed data and A.V. wrote the manuscript with input and editing from Q.Z., B.D.G. and Z.M.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7, Supplementary Note (PDF 5290 kb)
Supplementary Table 1
Number of raw and mapped deep sequencing reads of HITS-CLIP libraries. (XLSX 31 kb)
Supplementary Table 2
Comparison of CLIP and standard IP piRNA populations. (XLSX 28 kb)
Supplementary Table 3
Number of raw and mapped deep sequencing reads of RNA-seq libraries. (XLSX 34 kb)
Supplementary Table 4
Genomic locations of Intergenic piRNA hotspots. (XLSX 114 kb)
Supplementary Table 5
CLIP tag abundance and fold difference (glog-odds) of Miwi covered genes. (XLSX 64 kb)
Supplementary Table 6
Gene Ontology terms enriched in Miwi-covered genes. (XLSX 20 kb)
Supplementary Table 7
Mass spectrometry analysis of eluates from oligo(dT) pull-down experiments. (XLSX 10 kb)
Rights and permissions
About this article
Cite this article
Vourekas, A., Zheng, Q., Alexiou, P. et al. Mili and Miwi target RNA repertoire reveals piRNA biogenesis and function of Miwi in spermiogenesis. Nat Struct Mol Biol 19, 773–781 (2012). https://doi.org/10.1038/nsmb.2347
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2347
This article is cited by
-
Regulation of Miwi-mediated mRNA stabilization by Ck137956/Tssa is essential for male fertility
BMC Biology (2023)
-
Small RNAs, spermatogenesis, and male infertility: a decade of retrospect
Reproductive Biology and Endocrinology (2023)
-
Emerging roles and functional mechanisms of PIWI-interacting RNAs
Nature Reviews Molecular Cell Biology (2023)
-
GTSF1 accelerates target RNA cleavage by PIWI-clade Argonaute proteins
Nature (2022)
-
Unraveling mitochondrial piRNAs in mouse embryonic gonadal cells
Scientific Reports (2022)