Abstract
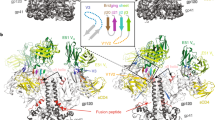
Broadly neutralizing antibodies such as 2F5 are directed against the membrane-proximal external region (MPER) of HIV-1 GP41 and recognize well-defined linear core sequences. These epitopes can be engrafted onto protein scaffolds to serve as immunogens with high structural fidelity. Although antibodies that bind to this core GP41 epitope can be elicited, they lack neutralizing activity. To understand this paradox, we used biophysical methods to investigate the binding of human 2F5 to the MPER in a membrane environment, where it resides in vivo. Recognition is stepwise, through a paratope more extensive than core binding site contacts alone, and dynamic rearrangement through an apparent scoop-like movement of heavy chain complementarity-determining region 3 (CDRH3) is essential for MPER extraction from the viral membrane. Core-epitope recognition on the virus requires the induction of conformational changes in both the MPER and the paratope. Hence, target neutralization through this lipid-embedded viral segment places stringent requirements on the plasticity of the antibody combining site.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
McElrath, M.J. & Haynes, B.F. Induction of immunity to human immunodeficiency virus type-1 by vaccination. Immunity 33, 542–554 (2010).
Han, Y., Wind-Rotolo, M., Yang, H.C., Siliciano, J.D. & Siliciano, R.F. Experimental approaches to the study of HIV-1 latency. Nat. Rev. Microbiol. 5, 95–106 (2007).
Feng, Y., Broder, C.C., Kennedy, P.E. & Berger, E.A. HIV-1 entry cofactor: functional cDNA cloning of a seven-transmembrane, G protein-coupled receptor. Science 272, 872–877 (1996).
Chan, D.C., Fass, D., Berger, J.M. & Kim, P.S. Core structure of gp41 from the HIV envelope glycoprotein. Cell 89, 263–273 (1997).
Harrison, S.C. Viral membrane fusion. Nat. Struct. Mol. Biol. 15, 690–698 (2008).
Richman, D.D., Wrin, T., Little, S.J. & Petropoulos, C.J. Rapid evolution of the neutralizing antibody response to HIV type 1 infection. Proc. Natl. Acad. Sci. USA 100, 4144–4149 (2003).
Kwong, P.D. et al. HIV-1 evades antibody-mediated neutralization through conformational masking of receptor-binding sites. Nature 420, 678–682 (2002).
Wei, X. et al. Antibody neutralization and escape by HIV-1. Nature 422, 307–312 (2003).
Simek, M.D. et al. Human immunodeficiency virus type 1 elite neutralizers: individuals with broad and potent neutralizing activity identified by using a high-throughput neutralization assay together with an analytical selection algorithm. J. Virol. 83, 7337–7348 (2009).
Cardoso, R.M. et al. Broadly neutralizing anti-HIV antibody 4E10 recognizes a helical conformation of a highly conserved fusion-associated motif in gp41. Immunity 22, 163–173 (2005).
Muster, T. et al. Cross-neutralizing activity against divergent human immunodeficiency virus type 1 isolates induced by the gp41 sequence ELDKWAS. J. Virol. 68, 4031–4034 (1994).
Nelson, J.D. et al. An affinity-enhanced neutralizing antibody against the membrane-proximal external region of human immunodeficiency virus type 1 gp41 recognizes an epitope between those of 2F5 and 4E10. J. Virol. 81, 4033–4043 (2007).
Trkola, A. et al. Human monoclonal antibody 2G12 defines a distinctive neutralization epitope on the gp120 glycoprotein of human immunodeficiency virus type 1. J. Virol. 70, 1100–1108 (1996).
Walker, L.M. et al. Broad and potent neutralizing antibodies from an African donor reveal a new HIV-1 vaccine target. Science 326, 285–289 (2009).
Zhou, T. et al. Structural basis for broad and potent neutralization of HIV-1 by antibody VRC01. Science 329, 811–817 (2010).
Zhou, T. et al. Structural definition of a conserved neutralization epitope on HIV-1 gp120. Nature 445, 732–737 (2007).
Salzwedel, K., West, J.T. & Hunter, E. A conserved tryptophan-rich motif in the membrane-proximal region of the human immunodeficiency virus type 1 gp41 ectodomain is important for Env-mediated fusion and virus infectivity. J. Virol. 73, 2469–2480 (1999).
Sun, Z.Y. et al. HIV-1 broadly neutralizing antibody extracts its epitope from a kinked gp41 ectodomain region on the viral membrane. Immunity 28, 52–63 (2008).
Ofek, G. et al. Structure and mechanistic analysis of the anti-human immunodeficiency virus type 1 antibody 2F5 in complex with its gp41 epitope. J. Virol. 78, 10724–10737 (2004).
Julien, J.P., Bryson, S., Nieva, J.L. & Pai, E.F. Structural details of HIV-1 recognition by the broadly neutralizing monoclonal antibody 2F5: epitope conformation, antigen-recognition loop mobility, and anion-binding site. J. Mol. Biol. 384, 377–392 (2008).
Alam, S.M. et al. The role of antibody polyspecificity and lipid reactivity in binding of broadly neutralizing anti-HIV-1 envelope human monoclonal antibodies 2F5 and 4E10 to glycoprotein 41 membrane proximal envelope epitopes. J. Immunol. 178, 4424–4435 (2007).
Sánchez-Martínez, S. et al. Specific phospholipid recognition by human immunodeficiency virus type-1 neutralizing anti-gp41 2F5 antibody. FEBS Lett. 580, 2395–2399 (2006).
Scherer, E.M., Leaman, D.P., Zwick, M.B., McMichael, A.J. & Burton, D.R. Aromatic residues at the edge of the antibody combining site facilitate viral glycoprotein recognition through membrane interactions. Proc. Natl. Acad. Sci. USA 107, 1529–1534 (2010).
Julien, J.P. et al. Ablation of the complementarity-determining region H3 apex of the anti-HIV-1 broadly neutralizing antibody 2F5 abrogates neutralizing capacity without affecting core epitope binding. J. Virol. 84, 4136–4147 (2010).
Xu, H. et al. Interactions between lipids and human anti-HIV antibody 4E10 can be reduced without ablating neutralizing activity. J. Virol. 84, 1076–1088 (2010).
Ofek, G. et al. Relationship between antibody 2F5 neutralization of HIV-1 and hydrophobicity of its heavy chain third complementarity-determining region. J. Virol. 84, 2955–2962 (2010).
Zwick, M.B. et al. The long third complementarity-determining region of the heavy chain is important in the activity of the broadly neutralizing anti-human immunodeficiency virus type 1 antibody 2F5. J. Virol. 78, 3155–3161 (2004).
Alam, S.M. et al. Role of HIV membrane in neutralization by two broadly neutralizing antibodies. Proc. Natl. Acad. Sci. USA 106, 20234–20239 (2009).
Guenaga, J. et al. Heterologous epitope-scaffold prime:boosting immuno-focuses B cell responses to the HIV-1 gp41 2F5 neutralization determinant. PLoS ONE 6, e16074 (2011).
Ofek, G. et al. Elicitation of structure-specific antibodies by epitope scaffolds. Proc. Natl. Acad. Sci. USA 107, 17880–17887 (2010).
Wales, T.E. & Engen, J.R. Hydrogen exchange mass spectrometry for the analysis of protein dynamics. Mass Spectrom. Rev. 25, 158–170 (2006).
Wales, T.E., Fadgen, K.E., Gerhardt, G.C. & Engen, J.R. High-speed and high-resolution UPLC separation at zero degrees celsius. Anal. Chem. 80, 6815–6820 (2008).
Miranker, A., Robinson, C.V., Radford, S.E., Aplin, R.T. & Dobson, C.M. Detection of transient protein folding populations by mass spectrometry. Science 262, 896–900 (1993).
Weis, D.D., Wales, T.E., Engen, J.R., Hotchko, M. & Ten Eyck, L.F. Identification and characterization of EX1 kinetics in H/D exchange mass spectrometry by peak width analysis. J. Am. Soc. Mass Spectrom. 17, 1498–1509 (2006).
Song, L. et al. Broadly neutralizing anti-HIV-1 antibodies disrupt a hinge-related function of gp41 at the membrane interface. Proc. Natl. Acad. Sci. USA 106, 9057–9062 (2009).
Takahashi, H., Nakanishi, T., Kami, K., Arata, Y. & Shimada, I. A novel NMR method for determining the interfaces of large protein-protein complexes. Nat. Struct. Biol. 7, 220–223 (2000).
Bryson, S., Julien, J.P., Hynes, R.C. & Pai, E.F. Crystallographic definition of the epitope promiscuity of the broadly neutralizing anti-human immunodeficiency virus type 1 antibody 2F5: vaccine design implications. J. Virol. 83, 11862–11875 (2009).
Tian, Y. et al. Structure-affinity relationships in the gp41 ELDKWA epitope for the HIV-1 neutralizing monoclonal antibody 2F5: effects of side-chain and backbone modifications and conformational constraints. J. Pept. Res. 59, 264–276 (2002).
Huang, C.C. et al. Structures of the CCR5 N terminus and of a tyrosine-sulfated antibody with HIV-1 gp120 and CD4. Science 317, 1930–1934 (2007).
Andreotti, A.H. Native state proline isomerization: an intrinsic molecular switch. Biochemistry 42, 9515–9524 (2003).
Lummis, S.C.R. et al. Cis–trans isomerization at a proline opens the pore of a neurotransmitter-gated ion channel. Nature 438, 248–252 (2005).
Padlan, E.A. Anatomy of the antibody molecule. Mol. Immunol. 31, 169–217 (1994).
Klein, J.S. et al. Examination of the contributions of size and avidity to the neutralization mechanisms of the anti-HIV antibodies b12 and 4E10. Proc. Natl. Acad. Sci. USA 106, 7385–7390 (2009).
Collis, A.V., Brouwer, A.P. & Martin, A.C. Analysis of the antigen combining site: correlations between length and sequence composition of the hypervariable loops and the nature of the antigen. J. Mol. Biol. 325, 337–354 (2003).
Johnson, G. & Wu, T.T. Preferred CDRH3 lengths for antibodies with defined specificities. Int. Immunol. 10, 1801–1805 (1998).
Kunert, R., Ruker, F. & Katinger, H. Molecular characterization of five neutralizing anti-HIV type 1 antibodies: identification of nonconventional D segments in the human monoclonal antibodies 2G12 and 2F5. AIDS Res. Hum. Retroviruses 14, 1115–1128 (1998).
Xiao, X. et al. Germline-like predecessors of broadly neutralizing antibodies lack measurable binding to HIV-1 envelope glycoproteins: implications for evasion of immune responses and design of vaccine immunogens. Biochem. Biophys. Res. Commun. 390, 404–409 (2009).
Xiao, X., Chen, W., Feng, Y. & Dimitrov, D.S. Maturation pathways of cross-reactive HIV-1 neutralizing antibodies. Viruses 1, 802–817 (2009).
Yin, J., Beuscher, A.E.t., Andryski, S.E., Stevens, R.C. & Schultz, P.G. Structural plasticity and the evolution of antibody affinity and specificity. J. Mol. Biol. 330, 651–656 (2003).
Mikell, I. et al. Characteristics of the earliest cross-neutralizing antibody response to HIV-1. PLoS Pathog. 7, e1001251 (2011).
Kim, S.T. et al. The αβ T cell receptor is an anisotropic mechanosensor. J. Biol. Chem. 284, 31028–31037 (2009).
Schmitz, K.R. & Ferguson, K.M. Interaction of antibodies with ErbB receptor extracellular regions. Exp. Cell Res. 315, 659–670 (2009).
Janssen, B.J. et al. Structural basis of semaphorin-plexin signalling. Nature 467, 1118–1122 (2010).
Pejchal, R. et al. A conformational switch in human immunodeficiency virus gp41 revealed by the structures of overlapping epitopes recognized by neutralizing antibodies. J. Virol. 83, 8451–8462 (2009).
Wales, T.E., Fadgen, K.E., Gerhardt, G.C. & Engen, J.R. High-speed and high-resolution UPLC separation at zero degrees Celsius. Anal. Chem. 80, 6815–6820 (2008).
Wang, L., Pan, H. & Smith, D.L. Hydrogen exchange-mass spectrometry: optimization of digestion conditions. Mol. Cell. Proteomics 1, 132–138 (2002).
Weis, D.D., Engen, J.R. & Kass, I.J. Semi-automated data processing of hydrogen exchange mass spectra using HX-Express. J. Am. Soc. Mass Spectrom. 17, 1700–1703 (2006).
Houde, D., Berkowitz, S.A. & Engen, J.R. The utility of hydrogen/deuterium exchange mass spectrometry in biopharmaceutical comparability studies. J. Pharm. Sci. 100, 2071–2086 (2011).
Shimada, I. NMR techniques for identifying the interface of a larger protein-protein complex: cross-saturation and transferred cross-saturation experiments. Methods Enzymol. 394, 483–506 (2005).
Acknowledgements
This work was supported by US National Institutes of Health (NIH) grants RO1AI84785 and U19AI91693 to E.L.R. and G.W., and a grant from the Gates Foundation, The Collaboration for AIDS Vaccine Discovery (CAVD) Program to E.L.R., G.W. and J.R.E. J.R.E. was also supported by NIH grant RO1-GM086507 and funding through a cooperative research agreement with the Waters Corporation. K.D.R. was supported by The Danish Council for Independent Research in Natural Sciences (FNU grant 09-063876). J.R.E. would like to thank T.E. Wales for expert technical assistance. L.S. was also supported by an US National High Magnetic Field Laboratory (NHMFL) User Collaboration Grants Program award. The NHMFL is funded by the US National Science Foundation through the Cooperative Agreement No. DMR-0654118, the State of Florida, and the US Department of Energy.
Author information
Authors and Affiliations
Contributions
Z.-Y.J.S. designed and conducted NMR experiments, analyzed the data and wrote the paper. G.W. directed the NMR effort and analyzed the NMR structure. A.F.F. contributed to the analysis of the CDRH3 loop. J.-H.W. contributed to the analysis of complex structure. X.S. and K.D.R. designed and carried out HX-MS experiments and contributed to the data analysis. J.R.E. contributed to the design and data analysis of HX-MS experiments and wrote the paper. L.S. conducted EPR experiments. S.M. prepared 2F5 and its variant Fab regions and carried out the labeling of MPER. Y.C. carried out cell transfection and FACS analysis. G.O., Y.Y. and P.D.K. provided 2F5 and mutant antibodies. M.K. designed and carried out SPR experiments, analyzed the data and wrote the paper. E.L.R. directed the project and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–7 and Supplementary Methods (PDF 3597 kb)
Rights and permissions
About this article
Cite this article
Kim, M., Sun, ZY., Rand, K. et al. Antibody mechanics on a membrane-bound HIV segment essential for GP41-targeted viral neutralization. Nat Struct Mol Biol 18, 1235–1243 (2011). https://doi.org/10.1038/nsmb.2154
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nsmb.2154
This article is cited by
-
An Engineered Biomimetic MPER Peptide Vaccine Induces Weakly HIV Neutralizing Antibodies in Mice
Annals of Biomedical Engineering (2020)
-
Development of prophylactic vaccines against HIV-1
Retrovirology (2013)