Abstract
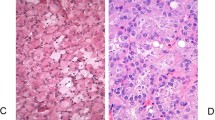
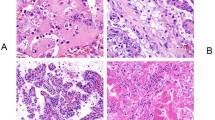
Clear cell renal cell carcinoma (RCC) represents the most common histological subtype of malignant kidney tumors. Based on symptoms alone, clear cell RCC is indistinguishable from other histological classes of RCC unless the tumor is present in the context of an RCC syndrome. Histopathological examination is, therefore, important to accurately identify clear cell RCC. Clear cell RCCs have characteristic morphological criteria; these tumors can be easily identified upon typical presentation, but diagnosis can be challenging when tumor cell pattern is unusual or when availability of tissue samples is limited. In this Review, the clinical, radiological and pathological characteristics of clear cell RCCs are described, as well as the potential tumors that can be confused with clear cell RCC and need to be considered in the differential diagnoses. Finally, the importance of an accurate diagnosis is highlighted in the context of the increasing use of preoperative tissue sampling and the prevalence of clear cell tumors associated with hereditary syndromes, which could have different therapeutic and prognostic implications for patients and their families.
Key Points
-
Renal cell carcinoma (RCC) is a type of renal tumor that encompasses a group of different morphological and genetic diseases
-
RCC can occur as sporadic cases or as part of hereditary syndromes
-
Morphological differences between subtypes are supported by underlying genetic changes
-
Clinically and radiologically, histotypes can be difficult to differentiate, especially for clear cell RCC
-
Misdiagnosis or misclassification of RCC can be minimized by extensive sampling of tumors
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Jemal, A., Siegel, R., Xu, J. & Ward, E. Cancer Statistics, 2010. CA Cancer J. Clin. 60, 277–300 (2010).
Ferlay, J. et al. GLOBOCAN 2008: Cancer incidence and mortality worldwide. International Agency for Research on Cancer [online], (2010).
Kümmerlin, I. P. E. D., Laguna, M. P., de la Rosette, J. J. M. C. H. & Bossuyt, P. M. M. in Renal Cell Cancer (eds de la Rosette, J. J. M. C. H., Sternberg, C. N. & van Poppel, H. P. A.) 1–8 (Springer, London, 2008).
Hunt, J. D., van der Hel, O. L., McMillan, G. P., Boffetta, P. & Brennan, P. Renal cell carcinoma in relation to cigarette smoking: meta-analysis of 24 studies. Int. J. Cancer 114, 101–108 (2005).
Chow, W. H., Dong, L. M. & Devesa, S. S. Epidemiology and risk factors for kidney cancer. Nat. Rev. Urol. 7, 245–257 (2010).
Dhote, R., Thiounn, N., Debré, B. & Vidal-Trecan, G. Risk factors for adult renal cell carcinoma. Urol. Clin. North Am. 31, 237–247 (2004).
Pascual, D. & Borque, A. Epidemiology of kidney cancer. Adv. Urol. 782381 (2008).
Linehan, W. M. et al. Hereditary kidney cancer: unique opportunity for disease-based therapy. Cancer 115, 2252–2261 (2009).
Schmidt, L. S. Birt-Hogg-Dubé syndrome, a genodermatosis that increases risk for renal carcinoma. Curr. Mol. Med. 4, 877–885 (2004).
Refae, M. A., Wong, N., Patenaude, F., Bégin, L. R. & Foulkes, W. D. Hereditary leiomyomatosis and renal cell cancer: an unusual and aggressive form of hereditary renal carcinoma. Nat. Clin. Pract. Oncol. 4, 256–261 (2007).
Schreiner, A. et al. Distinctive morphology of renal cell carcinomas in tuberous sclerosis. Int. J. Surg. Pathol. 18, 409–418 (2009).
Ricketts, C. et al. Germline SDHB mutations and familial renal cell carcinoma. J. Natl Cancer Inst. 100, 1260–1262 (2008).
Nishimura, H. et al. Renal cell carcinoma in autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 54, 165–168 (2009).
Hajj, P. et al. Prevalence of renal cell carcinoma in patients with autosomal dominant polycystic kidney disease and chronic renal failure. Urology 74, 631–634 (2009).
Sunela, K. L., Kataja, M. J. & Kellokumpu-Lehtinen, P. L. Changes in symptoms of renal cell carcinoma over four decades. BJU Int. 106, 649–653 (2010).
Jacques, A. E. T. & Reznek, R. H. in Imaging in Oncological Urology (eds de la Rosettem J. J. M. C. H., Manyak, M. J., Haringhani, M. G. & Wijkstra) 61–83 (Springer, London, 2009).
Pantuck, A. J., Zisman, A., Rauch, M. K. & Belldegrun, A. Incidental renal tumors. Urology 56, 190–196 (2000).
Motzer, R. J., Russo, P., Nanus, D. M. & Berg, W. J. Renal cell carcinoma. Curr. Probl. Cancer 21, 185–232 (1997).
Nguyen, M. M., Gill, I. S. & Ellison, L. M. The evolving presentation of renal carcinoma in the United States: trends from the Surveillance, Epidemiology, and End Results program. J. Urol. 176, 2397–2400 (2006).
Chawla, S. N. et al. The natural history of observed enhancing renal masses: meta-analysis and review of the world literature. J. Urol. 175, 425–431 (2006).
Martignoni, G. et al. in Clinical Pathology of Urologic Tumors (ed. Mikuz, G.) 253 (Informa Healthcare, Boca Raton, FL, 2007).
Decastro, G. J. & McKiernan, J. M. Epidemiology, clinical staging, and presentation of renal cell carcinoma. Urol. Clin. North Am. 35, 581–592 (2008).
Gold, P. J., Fefer, A. & Thompson, J. A. Paraneoplastic manifestations of renal cell carcinoma. Semin. Urol. Oncol. 14, 216–222 (1996).
Sheth, S. & Fishman, E. In Imaging of Kidney Cancer (eds Guermazi, A. & Abo-Kamil, R.) 29–47 (Springer, Berlin, Heidelberg, New York, 2006).
Tello, R. et al. MR imaging of renal masses interpreted on CT to be suspicious. AJR Am. J. Roentgenol. 174, 1017–1022 (2000).
Valera, V. A. et al. Protein expression profiling in the spectrum of renal cell carcinomas. J. Cancer 1, 184–196 (2010).
International Agency for Research on Cancer. Pathology and genetics of tumours of the urinary system and male genital organs. International Agency for Research on Cancer [online], (2004).
Grignon, D. J. & Che, M. Clear cell renal cell carcinoma. Clin. Lab. Med. 25, 305–316 (2005).
Lohse, C. M. & Cheville, J. C. A review of prognostic pathologic features and algorithms for patients treated surgically for renal cell carcinoma. Clin. Lab. Med. 25, 433–464 (2005).
Jinzaki, M. et al. Double-phase helical CT of small renal parenchymal neoplasms: correlation with pathologic findings and tumor angiogenesis. J. Comput. Assist. Tomogr. 24, 835–842 (2000).
Herts, B. R. et al. Enhancement characteristics of papillary renal neoplasms revealed on triphasic helical CT of the kidneys. AJR Am. J. Roentgenol. 178, 367–372 (2002).
Pedrosa, I. et al. MR classification of renal masses with pathologic correlation. Eur. Radiol. 18, 365–375 (2008).
Outwater, E. K., Bhatia, M., Siegelman, E. S., Burke, M. A. & Mitchell, D. G. Lipid in renal clear cell carcinoma: detection on opposed-phase gradient-echo MR images. Radiology 205, 103–107 (1997).
Yoshimitsu, K. et al. MR detection of cytoplasmic fat in clear cell renal cell carcinoma utilizing chemical shift gradient-echo imaging. J. Magn. Reson. Imaging 9, 579–585 (1999).
Raj, G. V. et al. Predicting the histology of renal masses using preoperative Doppler ultrasonography. J. Urol. 177, 53–58 (2007).
Murphy, W. M., Grignon, D. J., Perlman, E. J. & American Registry of Pathology. in Tumors of the Kidney, Bladder, and Related Urinary Structures (eds Murphy, W. M., Grignon, D. J., Perlman, E. J. & American Registry of Pathology) 109–121 (American Registry of Pathology, Washington, DC, 2004).
Frank, I. et al. Solid renal tumors: an analysis of pathological features related to tumor size. J. Urol. 170, 2217–2220 (2003).
Fuhrman, S. A., Lasky, L. C. & Limas, C. Prognostic significance of morphologic parameters in renal cell carcinoma. Am. J. Surg. Pathol. 6, 655–663 (1982).
Ficarra, V. et al. Original and reviewed nuclear grading according to the Fuhrman system: a multivariate analysis of 388 patients with conventional renal cell carcinoma. Cancer 103, 68–75 (2005).
Rioux-Leclercq, N. et al. Prognostic ability of simplified nuclear grading of renal cell carcinoma. Cancer 109, 868–874 (2007).
Reuter, V., Russo, P. & Motzer, R. J. in Comprehensive Textbook of Genitourinary Oncology (eds Vogelzang, N., Scardino, P. & Shipley, W. U.) 680–710 (Lippincott Williams & Wilkins, Philadelphia, 2006).
Reuter, V. E. & Tickoo, S. K. in Sternberg's Diagnostic Surgical Pathology 4th edn (eds Carter, D., Greenson, J. K., Oberman, H. A., Reuter, V. E. & Stohler, M. H.) 1955 (Lippincott Williams & Wilkins, Philadelphia, 2004).
Hammerich, K. H., Ayala, G. E. & Wheeler, T. M. Application of immunohistochemistry to the genitourinary system (prostate, urinary bladder, testis, and kidney). Arch. Pathol. Lab. Med. 132, 432–440 (2008).
Mackay, B., Ordonez, N. G., Khoursand, J. & Bennington, J. L. The ultrastructure and immunocytochemistry of renal cell carcinoma. Ultrastruct. Pathol. 11, 483–502 (1987).
Erlandson, R. A., Shek, T. W. & Reuter, V. E. Diagnostic significance of mitochondria in four types of renal epithelial neoplasms: an ultrastructural study of 60 tumors. Ultrastruct. Pathol. 21, 409–417 (1997).
Linehan, W. M., Rubin, J. S. & Bottaro, D. P. VHL loss of function and its impact on oncogenic signaling networks in clear cell renal cell carcinoma. Int. J. Biochem. Cell Biol. 41, 753–756 (2009).
McRonald, F. E. et al. CpG methylation profiling in VHL related and VHL unrelated renal cell carcinoma. Mol. Cancer 8, 31 (2009).
Cohen, H. T. & McGovern, F. J. Renal-cell carcinoma. N. Engl. J. Med. 353, 2477–2490 (2005).
Wiesener, M. S. et al. Constitutive activation of hypoxia-inducible genes related to overexpression of hypoxia-inducible factor-1α in clear cell renal carcinomas. Cancer Res. 61, 5215–5222 (2001).
Varela, I. et al. Exome sequencing identifies frequent mutation of the SWI/SNF complex gene PBRM1 in renal carcinoma. Nature 469, 539–542 (2011).
Schuetz, A. N. et al. Molecular classification of renal tumors by gene expression profiling. J. Mol. Diagn. 7, 206–218 (2005).
Young, A. N., Master, V. A., Paner, G. P., Wang, M. D. & Amin, M. B. Renal epithelial neoplasms: diagnostic applications of gene expression profiling. Adv. Anat. Pathol. 15, 28–38 (2008).
Zhuang, Z. et al. From tissue phenotype to proteotype: sensitive protein identification in microdissected tumor tissue. Int. J. Oncol. 28, 103–110 (2006).
Fridman, E. et al. Accurate molecular classification of renal tumors using microRNA expression. J. Mol. Diagn. 12, 687–696 (2010).
Hafron, J. et al. Imaging characteristics of minimal fat renal angiomyolipoma with histologic correlations. Urology 66, 1155–1159 (2005).
Oliva, M. R. et al. Renal cell carcinoma: T1 and T2 signal intensity characteristics of papillary and clear cell types correlated with pathology. AJR Am. J. Roentgenol. 192, 1524–1530 (2009).
Quaia, E., Bussani, R., Cova, M. & Mucelli, R. P. Radiologic-pathologic correlations of intratumoral tissue components in the most common solid and cystic renal tumors. Pictorial review. Eur. Radiol. 15, 1734–1744 (2005).
Shinmoto, H. et al. Small renal cell carcinoma: MRI with pathologic correlation. J. Magn. Reson. Imaging 8, 690–694 (1998).
Somani, B. K. et al. Image-guided biopsy-diagnosed renal cell carcinoma: critical appraisal of technique and long-term follow-up. Eur. Urol. 51, 1289–1295 (2007).
Samplaski, M. K., Zhou, M., Lane, B. R., Herts, B. & Campbell, S. C. Renal mass sampling: an enlightened perspective. Int. J. Urol. 18, 5–19 (2011).
Kümmerlin, I. et al. Diagnostic problems in the subtyping of renal tumors encountered by five pathologists. Pathol. Res. Pract. 205, 27–34 (2009).
Truong, L. D. & Shen, S. S. Immunohistochemical diagnosis of renal neoplasms. Arch. Pathol. Lab. Med. 135, 92–109 (2011).
Paraf, F. et al. Clear cell carcinoma of the thyroid; diagnostic difficulties [French]. Ann. Pathol. 9, 369–372 (1989).
Cameron, R. I., Ashe, P., O'Rourke, D. M., Foster, H. & McCluggage, W. G. A panel of immunohistochemical stains assists in the distinction between ovarian and renal clear cell carcinoma. Int. J. Gynecol. Pathol. 22, 272–276 (2003).
Cajulis, R. S., Katz, R. L., Dekmezian, R., el-Naggar, A. & Ro, J. Y. Fine needle aspiration biopsy of renal cell carcinoma. Cytologic parameters and their concordance with histology and flow cytometric data. Acta Cytol. 37, 367–372 (1993).
Renshaw, A. A., Lee, K. R., Madge, R. & Granter, S. R. Accuracy of fine needle aspiration in distinguishing subtypes of renal cell carcinoma. Acta Cytol. 41, 987–994 (1997).
Eshed, I., Elias, S. & Sidi, A. A. Diagnostic value of CT-guided biopsy of indeterminate renal masses. Clin. Radiol. 59, 262–267 (2004).
Richter, F., Kasabian, N. G., Irwin, R. J. Jr, Watson, R. A. & Lang, E. K. Accuracy of diagnosis by guided biopsy of renal mass lesions classified indeterminate by imaging studies. Urology 55, 348–352 (2000).
Wood, B. J. et al. Imaging guided biopsy of renal masses: indications, accuracy and impact on clinical management. J. Urol. 161, 1470–1474 (1999).
Lechevallier, E. et al. Fine-needle percutaneous biopsy of renal masses with helical CT guidance. Radiology 216, 506–510 (2000).
Wang, R., Li, A. Y. & Wood, D. P. Jr. The role of percutaneous renal biopsy in the management of small renal masses. Curr. Urol. Rep. 12, 18–23 (2011).
Rybicki, F. J. et al. Percutaneous biopsy of renal masses: sensitivity and negative predictive value stratified by clinical setting and size of masses. AJR Am. J. Roentgenol. 180, 1281–1287 (2003).
Silverman, S. G., Gan, Y. U., Mortele, K. J., Tuncali, K. & Cibas, E. S. Renal masses in the adult patient: the role of percutaneous biopsy. Radiology 240, 6–22 (2006).
Schmidt, L. S. et al. Birt-Hogg-Dube syndrome, a genodermatosis associated with spontaneous pneumothorax and kidney neoplasia, maps to chromosome 17p11.2. Am. J. Hum. Genet. 69, 876–882 (2001).
Pavlovich, C. P. et al. Renal tumors in the Birt-Hogg-Dubé syndrome. Am. J. Surg. Pathol. 26, 1542–1552 (2002).
Kiuru, M. & Launonen, V. Hereditary leiomyomatosis and renal cell cancer (HLRCC). Curr. Mol. Med. 4, 869–875 (2004).
Linehan, W. M. et al. Molecular diagnosis and therapy of kidney cancer. Annu. Rev. Med. 61, 329–343 (2010).
Lehtonen, H. J. et al. Conventional renal cancer in a patient with fumarate hydratase mutation. Hum. Pathol. 38, 793–796 (2007).
Petersen, R. O., Sesterhenn, I. & Davis, C. J. in Urologic Pathology 45–65 (Lippincott Williams & Wilkins, Philadelphia, 2009).
Bernstein, J., Robbins, T. O. & Kissane, J. M. The renal lesions of tuberous sclerosis. Semin. Diagn. Pathol. 3, 97–105 (1986).
Astuti, D. et al. Gene mutations in the succinate dehydrogenase subunit SDHB cause susceptibility to familial pheochromocytoma and to familial paraganglioma. Am. J. Hum. Genet. 69, 49–54 (2001).
Stedman, T. L. Stedman's Medical Dictionary (Lippincott Williams & Wilkins, Philadelphia, 2006).
Merriam–Webster Inc. Merriam–Webster dictionary (Merriam-Webster, Springfield, 2005).
Acknowledgements
This research was supported in part by the Intramural Research Program of the National Cancer Institute, NIH. The content of this publication does not necessarily reflect the views or policies of the Department of Health and Human Services, nor does mention of trade names, commercial products, or organizations imply endorsement by the US Government. C. P. Vega, University of California, Irvine, CA, is the author of and is solely responsible for the content of the learning objectives, questions and answers of the Medscape, LLC-accredited continuing medical education activity associated with this article.
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to researching, discussing, writing, editing and reviewing this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Valera, V., Merino, M. Misdiagnosis of clear cell renal cell carcinoma. Nat Rev Urol 8, 321–333 (2011). https://doi.org/10.1038/nrurol.2011.64
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrurol.2011.64
This article is cited by
-
Glycogen metabolism has a key role in the cancer microenvironment and provides new targets for cancer therapy
Journal of Molecular Medicine (2016)
-
Primary clear cell renal carcinoma cells display minimal mitochondrial respiratory capacity resulting in pronounced sensitivity to glycolytic inhibition by 3-Bromopyruvate
Cell Death & Disease (2015)
-
Fructose-1,6-bisphosphatase opposes renal carcinoma progression
Nature (2014)
-
Expression parameters of the metabolic pathway genes pyruvate dehydrogenase kinase-1 (PDK-1) and DJ-1/PARK7 in renal cell carcinoma (RCC)
World Journal of Urology (2013)
-
Renal cancer biomarkers: the promise of personalized care
BMC Medicine (2012)