Key Points
-
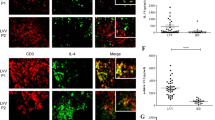
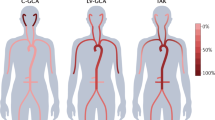
Giant cell arteritis (GCA), the most frequent form of large-vessel vasculitis, occurs in a strictly defined tissue context and requires corruption of the immune-privileged tissue niche of the arterial wall
-
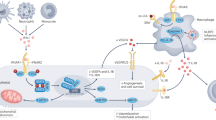
Receptors and ligands from the Notch family facilitate information exchange between vascular stromal cells and immune cells, and are critically involved in the development of vasculitis
-
The therapeutic potential of targeting the stromal compartment in vasculitis is currently unexplored
-
Granulomatous inflammation in GCA is characterized by a cytokine cascade, in which the initiating signals are poorly defined, but the many effectors match those encountered in protective immune responses
-
A cytokine cluster involving the IL-6–IL-17 axis is highly active in early and untreated disease, is rapidly suppressed by corticosteroids and is redundant for vasculitis
-
A cytokine cluster centring on the IL-12–IFN-γ axis is more resistant to immunosuppression and reveals pathogenic similarities between allograft arteriosclerosis and GCA
Abstract
Vasculitis of the medium and large arteries, most often presenting as giant cell arteritis (GCA), is an infrequent, but potentially fatal, type of immune-mediated vascular disease. The site of the aberrant immune reaction, the mural layers of the artery, is strictly defined by vascular dendritic cells, endothelial cells, vascular smooth muscle cells and fibroblasts, which engage in an interaction with T cells and macrophages to, ultimately, cause luminal stenosis or aneurysmal wall damage of the vessel. A multitude of effector cytokines, all known as critical mediators in host-protective immunity, have been identified in vasculitic lesions. Two dominant cytokine clusters—the IL-6–IL-17 axis and the IL-12–IFN-γ axis—have been linked to disease activity. These two clusters seem to serve different roles in the vasculitic process. The IL-6–IL-17 cluster is highly responsive to standard corticosteroid therapy, whereas the IL-12–IFN-γ cluster is resistant to steroid-mediated immunosuppression. The information exchange between vascular and immune cells and stabilization of the vasculitic process involves members of the Notch receptor and ligand family. Focusing on elements in the tissue context of GCA, instead of broadly suppressing host immunity, might enable a more tailored therapeutic approach that avoids unwanted adverse effects of aggressive immunosuppression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gabriel, S. E. & Michaud, K. Epidemiological studies in incidence, prevalence, mortality, and comorbidity of the rheumatic diseases. Arthritis Res. Ther. 11, 229 (2009).
Phillip, R. & Luqmani, R. Mortality in systemic vasculitis: a systematic review. Clin. Exp. Rheumatol. 26, S94–S104 (2008).
Richards, B. L., March, L. & Gabriel, S. E. Epidemiology of large-vessel vasculidities. Best Pract. Res. Clin. Rheumatol. 24, 871–883 (2010).
Weiss, P. F. Pediatric vasculitis. Pediatr. Clin. North Am. 59, 407–423 (2012).
Legein, B., Temmerman, L., Biessen, E. A. & Lutgens, E. Inflammation and immune system interactions in atherosclerosis. Cell. Mol. Life Sci. 70, 3847–3869 (2013).
Libby, P. Inflammation in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 32, 2045–2051 (2012).
Mackie, S. L., Hensor, E. M., Morgan, A. W. & Pease, C. T. Should I send my patient with previous giant cell arteritis for imaging of the thoracic aorta? A systematic literature review and meta-analysis. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2012-202145.
Kermani, T. A. et al. Large-vessel involvement in giant cell arteritis: a population-based cohort study of the incidence-trends and prognosis. Ann. Rheum. Dis. http://dx.doi.org/10.1136/annrheumdis-2012-202408.
Luqmani, R. Large vessel vasculitides: update for the cardiologist. Curr. Opin. Cardiol. 27, 578–584 (2012).
Weyand, C. M. & Goronzy, J. J. Medium- and large-vessel vasculitis. N. Engl. J. Med. 349, 160–169 (2003).
Jennette, J. C. et al. 2012 revised International Chapel Hill Consensus Conference Nomenclature of Vasculitides. Arthritis Rheum. 65, 1–11 (2013).
Shiran, H., Haddad, F., Miller, D. C. & Liang, D. Comparison of aortic root diameter to left ventricular outflow diameter versus body surface area in patients with marfan syndrome. Am. J. Cardiol. 110, 1518–1522 (2012).
Roman, M. J., Devereux, R. B., Kramer-Fox, R. & O'Loughlin, J. Two-dimensional echocardiographic aortic root dimensions in normal children and adults. Am. J. Cardiol. 64, 507–512 (1989).
Heistad, D. D. & Marcus, M. L. Role of vasa vasorum in nourishment of the aorta. Blood Vessels 16, 225–238 (1979).
Kassab, G. S. Biomechanics of the cardiovascular system: the aorta as an illustratory example. J. R. Soc. Interface 3, 719–740 (2006).
Seok, J. et al. Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc. Natl Acad. Sci. USA 110, 3507–3512 (2013).
Lacolley, P., Regnault, V., Nicoletti, A., Li, Z. & Michel, J. B. The vascular smooth muscle cell in arterial pathology: a cell that can take on multiple roles. Cardiovasc. Res. 95, 194–204 (2012).
McGettrick, H. M., Butler, L. M., Buckley, C. D., Rainger, G. E. & Nash, G. B. Tissue stroma as a regulator of leukocyte recruitment in inflammation. J. Leukoc. Biol. 91, 385–400 (2012).
Weyand, C. M. et al. Vascular dendritic cells in giant cell arteritis. Ann. NY Acad. Sci. 1062, 195–208 (2005).
Pryshchep, O., Ma-Krupa, W., Younge, B. R., Goronzy, J. J. & Weyand, C. M. Vessel-specific Toll-like receptor profiles in human medium and large arteries. Circulation 118, 1276–1284 (2008).
Stenmark, K. R. et al. The adventitia: essential regulator of vascular wall structure and function. Annu. Rev. Physiol. 75, 23–47 (2013).
Ma-Krupa, W. et al. Activation of arterial wall dendritic cells and breakdown of self-tolerance in giant cell arteritis. J. Exp. Med. 199, 173–183 (2004).
Krupa, W. M. et al. Trapping of misdirected dendritic cells in the granulomatous lesions of giant cell arteritis. Am. J. Pathol. 161, 1815–1823 (2002).
Wagner, A. D., Bjornsson, J., Bartley, G. B., Goronzy, J. J. & Weyand, C. M. Interferon-γ-producing T cells in giant cell vasculitis represent a minority of tissue-infiltrating cells and are located distant from the site of pathology. Am. J. Pathol. 148, 1925–1933 (1996).
Martinez-Taboada, V., Brack, A., Hunder, G. G., Goronzy, J. J. & Weyand, C. M. The inflammatory infiltrate in giant cell arteritis selects against B lymphocytes. J. Rheumatol. 23, 1011–1014 (1996).
Ma-Krupa, W., Kwan, M., Goronzy, J. J. & Weyand, C. M. Toll-like receptors in giant cell arteritis. Clin. Immunol. 115, 38–46 (2005).
Deng, J. et al. Toll-like receptors 4 and 5 induce distinct types of vasculitis. Circ. Res. 104, 488–495 (2009).
Koenig, C. L. et al. Identification of a Burkholderia-like strain from temporal arteries of subjects with giant cell arteritis. Arthritis Rheum. 64, S373 (2012).
Rodriguez-Pla, A. & Stone, J. H. Vasculitis and systemic infections. Curr. Opin. Rheumatol. 18, 39–47 (2006).
Koren, O. et al. Human oral, gut, and plaque microbiota in patients with atherosclerosis. Proc. Natl Acad. Sci. USA 108 (Suppl. 1), 4592–4598 (2011).
Dejaco, C. et al. NKG2D stimulated T-cell autoreactivity in giant cell arteritis and polymyalgia rheumatica. Ann. Rheum. Dis. 72, 1852–1859 (2013).
Dasgupta, B. & Panayi, G. S. Interleukin-6 in serum of patients with polymyalgia rheumatica and giant cell arteritis. Br. J. Rheumatol. 29, 456–458 (1990).
Roche, N. E. et al. Correlation of interleukin-6 production and disease activity in polymyalgia rheumatica and giant cell arteritis. Arthritis Rheum. 36, 1286–1294 (1993).
Hernandez-Rodriguez, J. et al. Tissue production of pro-inflammatory cytokines (IL-1β, TNF-α and IL-6) correlates with the intensity of the systemic inflammatory response and with corticosteroid requirements in giant-cell arteritis. Rheumatology (Oxford) 43, 294–301 (2004).
Weyand, C. M. & Goronzy, J. J. Giant-cell arteritis and polymyalgia rheumatica. Ann. Intern. Med. 139, 505–515 (2003).
Bode, J. G., Albrecht, U., Haussinger, D., Heinrich, P. C. & Schaper, F. Hepatic acute phase proteins--regulation by IL-6- and IL-1-type cytokines involving STAT3 and its crosstalk with NF-κB-dependent signaling. Eur. J. Cell Biol. 91, 496–505 (2012).
Weyand, C. M., Fulbright, J. W., Hunder, G. G., Evans, J. M. & Goronzy, J. J. Treatment of giant cell arteritis: interleukin-6 as a biologic marker of disease activity. Arthritis Rheum. 43, 1041–1048 (2000).
Kishimoto, T. IL-6: from its discovery to clinical applications. Int. Immunol. 22, 347–352 (2010).
Weaver, C. T., Harrington, L. E., Mangan, P. R., Gavrieli, M. & Murphy, K. M. Th17: an effector CD4 T cell lineage with regulatory T cell ties. Immunity 24, 677–688 (2006).
Bettelli, E., Korn, T. & Kuchroo, V. K. Th17: the third member of the effector T cell trilogy. Curr. Opin. Immunol. 19, 652–657 (2007).
Chen, Z., Laurence, A. & O'Shea, J. J. Signal transduction pathways and transcriptional regulation in the control of Th17 differentiation. Semin. Immunol. 19, 400–408 (2007).
Harrington, L. E., Mangan, P. R. & Weaver, C. T. Expanding the effector CD4 T-cell repertoire: the Th17 lineage. Curr. Opin. Immunol. 18, 349–356 (2006).
Maddur, M. S., Miossec, P., Kaveri, S. V. & Bayry, J. Th17 cells: biology, pathogenesis of autoimmune and inflammatory diseases, and therapeutic strategies. Am. J. Pathol. 181, 8–18 (2012).
Lee, W. W. et al. Regulating human Th17 cells via differential expression of IL-1 receptor. Blood 115, 530–540 (2010).
Torchinsky, M. B. & Blander, J. M. T helper 17 cells: discovery, function, and physiological trigger. Cell. Mol. Life Sci. 67, 1407–1421 (2010).
Donnelly, R. P. et al. Interleukin-26: an IL-10-related cytokine produced by Th17 cells. Cytokine Growth Factor Rev. 21, 393–401 (2010).
Rutz, S., Eidenschenk, C. & Ouyang, W. IL-22, not simply a Th17 cytokine. Immunol. Rev. 252, 116–132 (2013).
Sutherland, A. P. et al. IL-21 promotes CD8+ CTL activity via the transcription factor T-bet. J. Immunol. 190, 3977–3984 (2013).
Camporeale, A. & Poli, V. IL-6, IL-17 and STAT3: a holy trinity in auto-immunity? Front. Biosci. 17, 2306–2326 (2012).
Schutyser, E., Struyf, S. & Van Damme, J. The CC chemokine CCL20 and its receptor CCR6. Cytokine Growth Factor Rev. 14, 409–426 (2003).
Deng, J., Younge, B. R., Olshen, R. A., Goronzy, J. J. & Weyand, C. M. Th17 and Th1 T-cell responses in giant cell arteritis. Circulation 121, 906–915 (2010).
Samson, M. et al. Th1 and Th17 lymphocytes expressing CD161 are implicated in giant cell arteritis and polymyalgia rheumatica pathogenesis. Arthritis Rheum. 64, 3788–3798 (2012).
Terrier, B. et al. Interleukin-21 modulates Th1 and Th17 responses in giant cell arteritis. Arthritis Rheum. 64, 2001–2011 (2012).
Wagner, A. D., Goronzy, J. J. & Weyand, C. M. Functional profile of tissue-infiltrating and circulating CD68+ cells in giant cell arteritis. Evidence for two components of the disease. J. Clin. Invest. 94, 1134–1140 (1994).
Weyand, C. M., Wagner, A. D., Bjornsson, J. & Goronzy, J. J. Correlation of the topographical arrangement and the functional pattern of tissue-infiltrating macrophages in giant cell arteritis. J. Clin. Invest. 98, 1642–1649 (1996).
Kimura, A. & Kishimoto, T. IL-6: regulator of Treg/Th17 balance. Eur. J. Immunol. 40, 1830–1835 (2010).
Barbi, J., Pardoll, D. & Pan, F. Metabolic control of the Treg/Th17 axis. Immunol. Rev. 252, 52–77 (2013).
Espigol-Frigole, G. et al. Increased IL-17A expression in temporal artery lesions is a predictor of sustained response to glucocorticoid treatment in patients with giant-cell arteritis. Ann. Rheum. Dis. 72, 1481–1487 (2013).
Fogal, B. et al. Neutralizing IL-6 reduces human arterial allograft rejection by allowing emergence of CD161+ CD4+ regulatory T cells. J. Immunol. 187, 6268–6280 (2011).
Issa, F., Chandrasekharan, D. & Wood, K. J. Regulatory T cells as modulators of chronic allograft dysfunction. Curr. Opin. Immunol. 23, 648–654 (2011).
Mohan, S. V., Liao, Y. J., Kim, J. W., Goronzy, J. J. & Weyand, C. M. Giant cell arteritis: immune and vascular aging as disease risk factors. Arthritis Res. Ther. 13, 231 (2011).
Weyand, C. M., Younge, B. R. & Goronzy, J. J. IFN-γ and IL-17: the two faces of T-cell pathology in giant cell arteritis. Curr. Opin. Rheumatol. 23, 43–49 (2011).
Loock, J. et al. Treatment of refractory giant cell arteritis with cyclophosphamide:a retrospective analysis of 35 patients from three centres. Clin. Exp. Rheumatol. 30, S70–S76 (2012).
Unizony, S. et al. Tocilizumab for the treatment of large-vessel vasculitis (giant cell arteritis, Takayasu arteritis) and polymyalgia rheumatica. Arthritis Care Res. (Hoboken) 64, 1720–1729 (2012).
Xenitidis, T., Horger, M., Zeh, G., Kanz, L. & Henes, J. C. Sustained inflammation of the aortic wall despite tocilizumab treatment in two cases of Takayasu arteritis. Rheumatology (Oxford) 52, 1729–1731 (2013).
Evans, J. M., O'Fallon, W. M. & Hunder, G. G. Increased incidence of aortic aneurysm and dissection in giant cell (temporal) arteritis. A population-based study. Ann. Intern. Med. 122, 502–507 (1995).
Weyand, C. M., Liao, Y. J. & Goronzy, J. J. The immunopathology of giant cell arteritis: diagnostic and therapeutic implications. J. Neuroophthalmol. 32, 259–265 (2012).
Brack, A. et al. Glucocorticoid-mediated repression of cytokine gene transcription in human arteritis-SCID chimeras. J. Clin. Invest. 99, 2842–2850 (1997).
Pober, J. S. & Tellides, G. Participation of blood vessel cells in human adaptive immune responses. Trends Immunol. 33, 49–57 (2012).
Min, W. & Pober, J. S. AIP1 in graft arteriosclerosis. Trends Cardiovasc. Med. 21, 229–233 (2011).
Choi, J., Enis, D. R., Koh, K. P., Shiao, S. L. & Pober, J. S. T lymphocyte-endothelial cell interactions. Annu. Rev. Immunol. 22, 683–709 (2004).
Tellides, G. & Pober, J. S. Interferon-gamma axis in graft arteriosclerosis. Circ. Res. 100, 622–632 (2007).
MacMicking, J. D. Interferon-inducible effector mechanisms in cell-autonomous immunity. Nat. Rev. Immunol. 12, 367–382 (2012).
Zhang, S. Y. et al. Inborn errors of interferon (IFN)-mediated immunity in humans: insights into the respective roles of IFN-α/β, IFN-γ, and IFN-λ in host defense. Immunol. Rev. 226, 29–40 (2008).
van Boxel-Dezaire, A. H. & Stark, G. R. Cell type-specific signaling in response to interferon-γ. Curr. Top. Microbiol. Immunol. 316, 119–154 (2007).
Lee, Y. K., Mukasa, R., Hatton, R. D. & Weaver, C. T. Developmental plasticity of Th17 and Treg cells. Curr. Opin. Immunol. 21, 274–280 (2009).
Yu, L. et al. AIP1 prevents graft arteriosclerosis by inhibiting interferon-γ-dependent smooth muscle cell proliferation and intimal expansion. Circ. Res. 109, 418–427 (2011).
Kaiser, M., Weyand, C. M., Bjornsson, J. & Goronzy, J. J. Platelet-derived growth factor, intimal hyperplasia, and ischemic complications in giant cell arteritis. Arthritis Rheum. 41, 623–633 (1998).
Kaiser, M., Younge, B., Bjornsson, J., Goronzy, J. J. & Weyand, C. M. Formation of new vasa vasorum in vasculitis. Production of angiogenic cytokines by multinucleated giant cells. Am. J. Pathol. 155, 765–774 (1999).
Ahmad, U. et al. IFN-gamma primes intact human coronary arteries and cultured coronary smooth muscle cells to double-stranded RNA- and self-RNA-induced inflammatory responses by upregulating TLR3 and melanoma differentiation-associated gene 5. J. Immunol. 185, 1283–1294 (2010).
Eid, R. E. et al. Interleukin-17 and interferon-γ are produced concomitantly by human coronary artery-infiltrating T cells and act synergistically on vascular smooth muscle cells. Circulation 119, 1424–1432 (2009).
Petursdottir, V., Nordborg, E. & Nordborg, C. Atrophy of the aortic media in giant cell arteritis. APMIS 104, 191–198 (1996).
Bai, Y. et al. Interferon-gamma induces X-linked inhibitor of apoptosis-associated factor-1 and Noxa expression and potentiates human vascular smooth muscle cell apoptosis by STAT3 activation. J. Biol. Chem. 283, 6832–6842 (2008).
Lacotte, S., Brun, S., Muller, S. & Dumortier, H. CXCR3, inflammation, and autoimmune diseases. Ann. NY Acad. Sci. 1173, 310–317 (2009).
Ciccia, F. et al. IL-33 is overexpressed in the inflamed arteries of patients with giant cell arteritis. Ann. Rheum. Dis. 72, 258–264 (2013).
Ciccia, F. et al. Expression of interleukin-32 in the inflamed arteries of patients with giant cell arteritis. Arthritis Rheum. 63, 2097–2104 (2011).
Nizzoli, G. et al. Human CD1c+ dendritic cells secrete high levels of IL-12 and potently prime cytotoxic T-cell responses. Blood 122, 932–942 (2013).
Makkuni, D. et al. Is intimal hyperplasia a marker of neuro-ophthalmic complications of giant cell arteritis? Rheumatology (Oxford) 47, 488–490 (2008).
Salvarani, C. et al. Risk factors for visual loss in an Italian population-based cohort of patients with giant cell arteritis. Arthritis Rheum. 53, 293–297 (2005).
Gonzalez-Gay, M. A. et al. Visual manifestations of giant cell arteritis. Trends and clinical spectrum in 161 patients. Medicine (Baltimore) 79, 283–292 (2000).
Singh, A. G. et al. Visual manifestations in giant cell arteritis: trend over five decades. Arthritis Rheum. 64, S993 (2012).
Khokha, R., Murthy, A. & Weiss, A. Metalloproteinases and their natural inhibitors in inflammation and immunity. Nat. Rev. Immunol. 13, 649–665 (2013).
Enzerink, A. & Vaheri, A. Fibroblast activation in vascular inflammation. J. Thromb. Haemost. 9, 619–626 (2011).
O'Shea, J. J., Ma, A. & Lipsky, P. Cytokines and autoimmunity. Nat. Rev. Immunol. 2, 37–45 (2002).
Hoffman, G. S. et al. Infliximab for maintenance of glucocorticosteroid-induced remission of giant cell arteritis: a randomized trial. Ann. Intern. Med. 146, 621–630 (2007).
Kotter, I., Henes, J. C., Wagner, A. D., Loock, J. & Gross, W. L. Does glucocorticosteroid-resistant large-vessel vasculitis (giant cell arteritis and Takayasu arteritis) exist and how can remission be achieved? A critical review of the literature. Clin. Exp. Rheumatol. 30, S114–129 (2012).
Adizie, T., Christidis, D., Dharmapaliah, C., Borg, F. & Dasgupta, B. Efficacy and tolerability of leflunomide in difficult-to-treat polymyalgia rheumatica and giant cell arteritis: a case series. Int. J. Clin. Pract. 66, 906–909 (2012).
Lie, J. T. Illustrated histopathologic classification criteria for selected vasculitis syndromes. American College of Rheumatology Subcommittee on Classification of Vasculitis. Arthritis Rheum. 33, 1074–1087 (1990).
Klein, R. G., Hunder, G. G., Stanson, A. W. & Sheps, S. G. Large artery involvement in giant cell (temporal) arteritis. Ann. Intern. Med. 83, 806–812 (1975).
Han, J. W. et al. Vessel wall-embedded dendritic cells induce T-cell autoreactivity and initiate vascular inflammation. Circ. Res. 102, 546–553 (2008).
Agrawal, A., Sridharan, A., Prakash, S. & Agrawal, H. Dendritic cells and aging: consequences for autoimmunity. Expert Rev. Clin. Immunol. 8, 73–80 (2012).
Shaw, A. C. et al. Dysregulation of human Toll-like receptor function in aging. Ageing Res. Rev. 10, 346–353 (2011).
Shaw, A. C., Joshi, S., Greenwood, H., Panda, A. & Lord, J. M. Aging of the innate immune system. Curr. Opin. Immunol. 22, 507–513 (2010).
Barone, F., Nayar, S. & Buckley, C. D. The role of non-hematopoietic stromal cells in the persistence of inflammation. Front. Immunol. 3, 416 (2012).
Roozendaal, R. & Mebius, R. E. Stromal cell-immune cell interactions. Annu. Rev. Immunol. 29, 23–43 (2011).
Piggott, K. et al. Blocking the NOTCH pathway inhibits vascular inflammation in large-vessel vasculitis. Circulation 123, 309–318 (2011).
Bray, S. J. Notch signalling: a simple pathway becomes complex. Nat. Rev. Mol. Cell Biol. 7, 678–689 (2006).
Tzoneva, G. & Ferrando, A. A. Recent advances on NOTCH signaling in T-ALL. Curr. Top. Microbiol. Immunol. 360, 163–182 (2012).
Boucher, J., Gridley, T. & Liaw, L. Molecular pathways of notch signaling in vascular smooth muscle cells. Front. Physiol. 3, 81 (2012).
Yang, K. & Proweller, A. Vascular smooth muscle Notch signals regulate endothelial cell sensitivity to angiogenic stimulation. J. Biol. Chem. 286, 13741–13753 (2011).
Koyanagi, A., Sekine, C. & Yagita, H. Expression of Notch receptors and ligands on immature and mature T cells. Biochem. Biophys. Res. Commun. 418, 799–805 (2012).
Van de Walle, I. et al. Specific Notch receptor-ligand interactions control human TCR-αβ/γδ development by inducing differential Notch signal strength. J. Exp. Med. 210, 683–697 (2013).
Ilarregui, J. M. et al. Tolerogenic signals delivered by dendritic cells to T cells through a galectin-1-driven immunoregulatory circuit involving interleukin 27 and interleukin 10. Nat. Immunol. 10, 981–991 (2009).
Svensson, M., Maroof, A., Ato, M. & Kaye, P. M. Stromal cells direct local differentiation of regulatory dendritic cells. Immunity. 21, 805–816 (2004).
Huang, Y. et al. Kidney-derived stromal cells modulate dendritic and T cell responses. J. Am. Soc. Nephrol. 20, 831–841 (2009).
Li, Q., Guo, Z., Xu, X., Xia, S. & Cao, X. Pulmonary stromal cells induce the generation of regulatory DC attenuating T-cell-mediated lung inflammation. Eur. J. Immunol. 38, 2751–2761 (2008).
Zhang, M. et al. Splenic stroma drives mature dendritic cells to differentiate into regulatory dendritic cells. Nat. Immunol. 5, 1124–1133 (2004).
Xiao, J. et al. Syndecan-1 displays a protective role in aortic aneurysm formation by modulating T cell-mediated responses. Arterioscler. Thromb. Vasc. Biol. 32, 386–396 (2012).
Pober, J. S. & Sessa, W. C. Evolving functions of endothelial cells in inflammation. Nat. Rev. Immunol. 7, 803–815 (2007).
Régent, A. et al. Identification of target antigens of anti-endothelial cell and anti-vascular smooth muscle cell antibodies in patients with giant cell arteritis: a proteomic approach. Arthritis Res. Ther. 13, R107 (2011).
Dimitrijevic, I., Andersson, C., Rissler, P. & Edvinsson, L. Increased tissue endothelin-1 and endothelin-B receptor expression in temporal arteries from patients with giant cell arteritis. Ophthalmology 117, 628–636 (2010).
Acknowledgements
The authors would like to acknowledge support from grants from the NIH (R01 EY011916, P01 HL058000, U19 AI057266 and U19 AI090019) and the Govenar Discovery Fund (C. M. Weyand).
Author information
Authors and Affiliations
Contributions
Both authors made equal contributions to all aspects of this manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Weyand, C., Goronzy, J. Immune mechanisms in medium and large-vessel vasculitis. Nat Rev Rheumatol 9, 731–740 (2013). https://doi.org/10.1038/nrrheum.2013.161
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2013.161
This article is cited by
-
Angiogenesis related genes in Takayasu Arteritis (TAK): robust association with Tag SNPs of IL-18 and FGF-2 in a South Asian Cohort
Journal of Human Genetics (2024)
-
[18F]FDG PET/CT for therapeutic assessment of Abatacept in early-onset polymyalgia rheumatica
European Journal of Nuclear Medicine and Molecular Imaging (2024)
-
Distinct gene signatures of monocytes and B cells in patients with giant cell arteritis: a longitudinal transcriptome analysis
Arthritis Research & Therapy (2023)
-
Diagnostic model based on bioinformatics and machine learning to distinguish Kawasaki disease using multiple datasets
BMC Pediatrics (2022)
-
UBA1 gene mutation in giant cell arteritis
Clinical Rheumatology (2022)