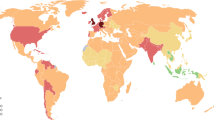
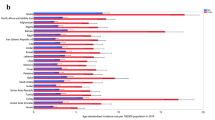
Abstract
The accumulative global burden of autoimmune and inflammatory rheumatic diseases is substantial. Studying the distribution of these conditions across various global regions and ethnic groups by means of geoepidemiology might readily provide epidemiological data and also advance our understanding of their genetic and environmental underpinnings. In order to depict the geoepidemiology of autoimmune and inflammatory rheumatic diseases, namely rheumatoid arthritis, juvenile rheumatoid arthritis, systemic lupus erythematosus, systemic sclerosis, ankylosing spondylitis and Sjögren's syndrome, we present a comprehensive collection of epidemiological reports from various world regions, including the prevalence of each of these conditions. The accumulated data show that the reviewed rheumatic diseases are global phenomena, and, with some variance, seem to be relatively evenly distributed. This finding is in contrast with the obviously uneven distribution of some major nonrheumatic autoimmune conditions. In addition, geoepidemiology demonstrates that ethnogenetic susceptibility interacts with lifestyle and environmental factors, which include socioeconomic status, infectious agents (triggering or protective agents), environmental pollutants, and vitamin D (dependent on sunlight exposure), in determining the risk of developing rheumatic autoimmunity.
Key Points
-
Geoepidemiology provides a comprehensible picture of the burden of autoimmune and inflammatory rheumatic diseases across various regions and ethnicities, and helps to unravel potential causative factors
-
The global distribution of rheumatic autoimmune diseases, with some variance, seems to be ubiquitous, although prominent gradients are present in the distribution of some major nonrheumatic autoimmune conditions
-
Our knowledge of the genetics of rheumatic autoimmunity is supported and advanced by geoepidemiology
-
The risk of developing these illnesses is also affected by environmental factors, such as socioeconomic status and exposure to infectious agents (protective or pathogenic), ultraviolet radiation and pollution
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Shapira, Y., Agmon-Levin, N. & Shoenfeld, Y. Defining and analyzing geoepidemiology and human autoimmunity. J. Autoimmun. 34, J168–J177 (2010).
Shoenfeld, Y., Selmi, C., Zimlichman, E. & Gershwin, M. E. The autoimmunologist: geoepidemiology, a new center of gravity, and prime time for autoimmunity. J. Autoimmun. 31, 325–330 (2008).
Cooper, G. S. & Stroehla, B. C. The epidemiology of autoimmune diseases. Autoimmun. Rev. 2, 119–125 (2003).
Eaton, W. W., Rose, N. R., Kalaydjian, A., Pedersen, M. G. & Mortensen, P. B. Epidemiology of autoimmune diseases in Denmark. J. Autoimmun. 29, 1–9 (2007).
NIH Autoimmune Diseases Coordinating Committee. Progress in Autoimmune Diseases Research (National Institutes of Health, Bethesda, 2005).
WHO. Atlas—Multiple Sclerosis Resources in the World (WHO, Geneva, 2008).
DIAMOND Project Group. Incidence and trends of childhood type 1 diabetes worldwide 1990–1999. Diabet. Med. 23, 857–866 (2006).
Symmons, D., Mathers, C. & Pfleger, B. Global Burden of Disease 2000: the global burden of rheumatoid arthritis in the year 2000 (WHO, Geneva, 2000).
Jacobson, D. L., Gange, S. J., Rose, N. R. & Graham, N. M. Epidemiology and estimated population burden of selected autoimmune diseases in the United States. Clin. Immunol. Immunopathol. 84, 223–243 (1997).
Gregersen, P. K. & Olsson, L. Recent advances in the genetics of autoimmune disease. Annu. Rev. Immunol. 27, 363–391 (2009).
Rønningen, K. S., Keiding, N. & Green, A. ; EURODIAB ACE Study Group. Correlations between the incidence of childhood-onset type I diabetes in Europe and HLA genotypes. Diabetologia 44, 51–59 (2001).
Mathieu, A. et al. The interplay between the geographic distribution of HLA-B27 alleles and their role in infectious and autoimmune disease: A unifying hypothesis. Autoimmun. Rev. 8, 420–425 (2009).
Kochi, Y., Suzuki, A., Yamada, R. & Yamamoto, K. Genetics of rheumatoid arthritis: underlying evidence of ethnic differences. J. Autoimmun. 32, 158–162 (2009).
Zhou, X. et al. Genome-wide association study for regions of systemic sclerosis susceptibility in a Choctaw Indian population with high disease prevalence. Arthritis Rheum. 48, 2585–2592 (2003).
Balluz, L. et al. Investigation of systemic lupus erythematosus in Nogales, Arizona. Am. J. Epidemiol. 154, 1029–1036 (2001).
Silman, A. J., Howard, Y., Hicklin, A. J. & Black, C. Geographical clustering of scleroderma in south and west London. Br. J. Rheumatol. 29, 93–96 (1990).
Shoenfeld, Y. et al. The mosaic of autoimmunity: genetic factors involved in autoimmune diseases. Isr. Med. Assoc. J. 10, 3–7 (2008).
Shoenfeld, Y. et al. The mosaic of autoimmunity: hormonal and environmental factors involved in autoimmune diseases. Isr. Med. Assoc. J. 10, 8–12 (2008).
Hewagama, A. & Richardson, B. The genetics and epigenetics of autoimmune diseases. J. Autoimmun. 33, 3–11 (2009).
Hopkinson, N. D., Doherty, M. & Powell, R. J. Clinical features and race specific incidence/prevalence rates of systemic lupus erythematosus in a geographically complete cohort of patients. Ann. Rheum. Dis. 53, 675–680 (1994).
Clark, I. A., Al-Yaman, F. M., Cowden, W. B. & Rockett, K. A. Does malarial tolerance, through nitric oxide, explain the low incidence of autoimmune disease in tropical Africa? Lancet 348, 1492–1494 (1996).
Manners, P. J. & Bower, C. Worldwide prevalence of juvenile arthritis—why does it vary so much? J. Rheumatol. 29, 1520–1530 (2002).
Fox, R. I. Epidemiology, pathogenesis, animal models, and treatment of Sjögren's syndrome. Curr. Opin. Rheumatol. 6, 501–508 (1994).
Vinagre, F., Santos, M. J., Prata, A., da Silva, J. C. & Santos, A. I. Assessment of salivary gland function in Sjögren's syndrome: the role of salivary gland scintigraphy. Autoimmun. Rev. 8, 672–676 (2009).
Minaur, N., Sawyers, S., Parker, J. & Darmawan, J. Rheumatic disease in an Australian Aboriginal community in North Queensland, Australia. A WHO-ILAR COPCORD survey. J. Rheumatol. 31, 965–972 (2004).
Silman, A. J. et al. Absence of rheumatoid arthritis in a Nigerian population. J. Rheumatol. 20, 618–622 (1993).
Darmawan, J., Muirden, K. D., Valkenburg, H. A. & Wigley, R. D. The epidemiology of rheumatoid arthritis in Indonesia. Br. J. Rheumatol. 32, 537–540 (1993).
Zeng, Q. Y. et al. Rheumatic diseases in China. Arthritis Res. Ther. 10, R17 (2008).
Kalla, A. A. Rheumatoid arthritis in the developing world. Best Pract. Res. Clin. Rheumatol. 17, 863–875 (2003).
Al Rawi, Z. S., Alazzawi, A. J., Alajili, F. M. & Alwakil, R. Rheumatoid arthritis in population samples in Iraq. Ann. Rheum. Dis. 37, 73–75 (1978).
Solomon, L., Robin, G. & Valkenburg, H. A. Rheumatoid arthritis in an urban South African Negro population. Ann. Rheum. Dis. 34, 128–135 (1975).
Bang, S. Y. et al. Smoking increases rheumatoid arthritis susceptibility in individuals carrying the HLA-DRB1 shared epitope, regardless of rheumatoid factor or anti-cyclic citrullinated peptide antibody status. Arthritis Rheum. 62, 369–377 (2010).
Doran, M. F., Pond, G. R., Crowson, C. S., O'Fallon, W. M. & Gabriel, S. E. Trends in incidence and mortality in rheumatoid arthritis in Rochester, Minnesota, over a forty-year period. Arthritis Rheum. 46, 625–631 (2002).
Kivity, S., Agmon-Levin, N., Blank, M. & Shoenfeld, Y. Infections and autoimmunity—friends or foes? Trends Immunol. 30, 409–414 (2009).
Lidar, M. et al. Infectious serologies and autoantibodies in inflammatory bowel disease: Insinuations at a true pathogenic role. Ann. NY Acad. Sci. 1173, 640–648 (2009).
Ram, M. et al. The putative protective role of hepatitis B virus (HBV) infection from autoimmune disorders. Autoimmun. Rev. 7, 621–625 (2008).
Agmon-Levin, N. et al. Prevalence of hepatitis C serum antibody in autoimmune diseases. J. Autoimmun. 32, 261–266 (2009).
Zandman-Goddard, G. et al. Neuropsychiatric lupus and infectious triggers. Lupus 17, 380–384 (2008).
Saurenmann, R. K. et al. Epidemiology of juvenile idiopathic arthritis in a multiethnic cohort: ethnicity as a risk factor. Arthritis Rheum. 56, 1974–1984 (2007).
McGill, P. E. & Oyoo, G. O. Rheumatic disorders in sub-Saharan Africa. East Afr. Med. J. 79, 214–216 (2002).
Oen, K., Fast, M. & Postl, B. Epidemiology of juvenile rheumatoid arthritis in Manitoba, Canada, 1975–92: cycles in incidence. J. Rheumatol. 22, 745–750 (1995).
Peterson, L. S., Mason, T., Nelson, A. M., O'Fallon, W. M. & Gabriel, S. E. Juvenile rheumatoid arthritis in Rochester, Minnesota 1960–1993. Is the epidemiology changing? Arthritis Rheum. 39, 1385–1390 (1996).
Enzer, I. et al. An epidemiologic study of trends in prevalence of rheumatoid factor seropositivity in Pima Indians: evidence of a decline due to both secular and birth-cohort influences. Arthritis Rheum. 46, 1729–1734 (2002).
Kaipiainen-Seppanen, O. & Savolainen, A. Changes in the incidence of juvenile rheumatoid arthritis in Finland. Rheumatology (Oxford) 40, 928–932 (2001).
Helmick, C. G. et al. Estimates of the prevalence of arthritis and other rheumatic conditions in the United States. Part I. Arthritis Rheum. 58, 15–25 (2008).
Samanta, A., Feehally, J., Roy, S. & Nichol, F. E. High prevalence of systemic disease and mortality in Asian subjects with systemic lupus erythematosus. Ann. Rheum. Dis. 50, 490–492 (1991).
Maskarinec, G. & Katz, A. R. Prevalence of systemic lupus erythematosus in Hawaii: is there a difference between ethnic groups? Hawaii Med. J. 54, 406–409 (1995).
Seagasothy, M. & Phillips, P. A. Systemic lupus erythematosus in Aborigines and Caucasians in central Australia: a comparative study. Lupus 10, 439–444 (2001).
Peschken, C. A. & Esdaile, J. M. Systemic lupus erythematosus in North American Indians: a population based study. J. Rheumatol. 27, 1884–1891 (2000).
Bernstein, L., Teal, C. R., Joslyn, S. & Wilson, J. Ethnicity-related variation in breast cancer risk factors. Cancer 97 (Suppl.), 222–229 (2003).
Molokhia, M., McKeigue, P. M., Cuadrado, M. & Hughes, G. Systemic lupus erythematosus in migrants from west Africa compared with Afro-Caribbean people in the UK. Lancet 357, 1414–1415 (2001).
Bae, S., Fraser, P. & Liang, M. H. The epidemiology of systemic lupus erythematosus in populations of African ancestry. Arthritis Rheum. 41, 2091–2099 (1998).
Westlake, S. L. & Edwards, C. J. Anti-malarials and lupus in West Africa use and lupus in Africans. Lupus 18, 193–195 (2009).
Shoenfeld, N., Amital, H. & Shoenfeld, Y. The effect of melanism and vitamin D synthesis on the incidence of autoimmune disease. Nat. Clin. Pract. Rheumatol. 5, 99–105 (2009).
Shapira, Y., Agmon-Levin, N. & Shoenfeld, Y. Mycobacterium tuberculosis, autoimmunity and vitamin D. Clin. Rev. Allergy Immunol. 38, 169–177 (2010).
Ponsonby, A. L., McMichael, A. & van der Mei, I. Ultraviolet radiation and autoimmune disease: insights from epidemiological research. Toxicology 181–182, 71–78 (2002).
Kiyohara, C. et al. Cigarette smoking, N-acetyltransferase 2 polymorphisms and systemic lupus erythematosus in a Japanese population. Lupus 18, 630–638 (2009).
Molokhia, M. & McKeigue, P. Systemic lupus erythematosus: genes versus environment in high risk populations. Lupus 15, 827–832 (2006).
Berkun, Y. et al. Infectious antibodies in systemic lupus erythematosus patients. Lupus 18, 1129–1135 (2009).
Kardestuncer, T. & Frumkin, H. Systemic lupus erythematosus in relation to environmental pollution: an investigation in an African-American community in North Georgia. Arch. Environ. Health 52, 85–90 (1997).
Parks, C. G. & Cooper, G. S. Occupational exposures and risk of systemic lupus erythematosus: a review of the evidence and exposure assessment methods in population- and clinic-based studies. Lupus 15, 728–736 (2006).
Chifflot, H., Fautrel, B., Sordet, C., Chatelus, E. & Sibilia, J. Incidence and prevalence of systemic sclerosis: a systematic literature review. Semin. Arthritis Rheum. 37, 223–235 (2008).
Shivananda, S. et al. Incidence of inflammatory bowel disease across Europe: is there a difference between north and south? Results of the European Collaborative Study on Inflammatory Bowel Disease (EC-IBD). Gut 39, 690–697 (1996).
Mayes, M. D. et al. Prevalence, incidence, survival, and disease characteristics of systemic sclerosis in a large US population. Arthritis Rheum. 48, 2246–2255 (2003).
Laing, T. J. et al. Racial differences in scleroderma among women in Michigan. Arthritis Rheum. 40, 734–742 (1997).
Tan, F. K. et al. HLA haplotypes and microsatellite polymorphisms in and around the major histocompatibility complex region in a Native American population with a high prevalence of scleroderma (systemic sclerosis). Tissue Antigens 53, 74–80 (1999).
Tan, F. K. et al. Association of fibrillin 1 single-nucleotide polymorphism haplotypes with systemic sclerosis in Choctaw and Japanese populations. Arthritis Rheum. 44, 893–901 (2001).
Valesini, G., Litta, A. & Bonavita, M. S. Geographical clustering of scleroderma in a rural area in the province of Rome. Clin. Exp. Rheumatol. 11, 41–47 (1993).
Van der Linden, S. M., Valkenburg, H. A., de Jongh, B. M. & Cats, A. The risk of developing ankylosing spondylitis in HLA-B27 positive individuals: a comparison of relatives of spondylitis patients with the general population. Arthritis Rheum. 27, 241–249 (1984).
Hukuda, S. et al. Spondyloarthropathies in Japan: nationwide questionnaire survey performed by the Japan Ankylosing Spondylitis Society. J. Rheumatol. 28, 554–559 (2001).
Khan, M. A. Spondyloarthropathies in non-Caucasian populations of the world. In Advances in Inflammation Research. Vol. 9: the Spondyloarthropathies (eds Ziff, M. & Cohen, S. B.) 91–99 (Raven Press, New York, (1985).
Gofton, J. P., Bennett, P. H., Smythe, H. A. & Decker, J. L. Sacroilitis and ankylosing spondylitis in North American Indians. Ann. Rheum. Dis. 31, 474–481 (1972).
Boyer, G. S. et al. Prevalence of spondyloarthropathies in Alaskan Eskimos. J. Rheumatol. 21, 2292–2297 (1994).
Braun, J., Bollow, M. & Remlinger, G. Prevalence of spondylarthropathies in HLA-B27 positive and negative blood donors. Arthritis Rheum. 41, 58–67 (1998).
Mijiyawa, M., Oniankitan, O. & Khan, M. A. Spondyloarthropathies in sub-Saharan Africa. Curr. Opin. Rheumatol. 12, 281–286 (2000).
Baum, J. & Ziff, M. The rarity of ankylosing spondylitis in the black race. Arthritis Rheum. 14, 12–18 (1971).
Hill, A. V. et al. HLA class I typing by PCR: HLA-B27 and an African B27 subtype. Lancet 337, 640–642 (1991).
Brown, M. A. et al. Ankylosing spondylitis in West Africans—evidence for a non-HLA-B27 protective effect. Ann. Rheum. Dis. 56, 68–70 (1997).
Khan, M. A. et al. HLA-B27 in ankylosing spondylitis: differences in frequency and relative risk in American blacks and Caucasians. J. Rheumatol. 3 (Suppl.), 39–43 (1977).
Silman, A. J. & Rooney, B. K. Epidemiology of Sjögren's syndrome. In The 100-year Anniversary of Henrik Sjögren (eds Eriksson, E. & Jonsson, R.) 53–57 (Hygiea, Jönköping, 1999).
Alamanosa, Y. & Drosos, A. A. Epidemiology of adult rheumatoid arthritis. Autoimmun. Rev. 4, 130–136 (2005).
Symmons, D. et al. The prevalence of rheumatoid arthritis in the United Kingdom; new estimates for a new century. Rheumatology (Oxford) 41, 793–800 (2002).
Stojanovic, R., Vlajinac, H., Palic-Obradovic, D., Janosevic, S. & Adanja, B. Prevalence of rheumatoid arthritis in Belgrade, Yugoslavia. Br. J. Rheumatol. 37, 729–732 (1998).
Shichikawa, K. et al. Changes in the incidence and prevalence of rheumatoid arthritis in Kamitonda, Wakayama, Japan, 1965–1996. Ann. Rheum. Dis. 58, 751–756 (1999).
Spindler, A. et al. Prevalence of rheumatoid arthritis in Tucuman, Argentina. J. Rheumatol. 29, 1166–1170 (2002).
Simonsson, M., Bergman, S., Jacobsson, L. T., Petersson, I. F. & Svensson, B. The prevalence of rheumatoid arthritis in Sweden. Scand. J. Rheumatol. 28, 340–343 (1999).
Riise, T., Jacobsen, B. K. & Gran, J. T. Incidence and prevalence of rheumatoid arthritis in the county of Troms, northern Norway. J. Rheumatol. 27, 1386–1389 (2000).
Drosos, A. A. et al. Epidemiology of adult rheumatoid arthritis in northwest Greece 1987–1995. J. Rheumatol. 24, 2129–2133 (1997).
Arguedas, O., Fasth, A., Andersson-Gäre, B. & Porras, O. Juvenile chronic arthritis in urban San José, Costa Rica: a 2 year prospective study. J. Rheumatol. 25, 1844–1850 (1998).
Pruunsild, C. et al. Prevalence and short-term outcome of juvenile idiopathic arthritis: a population-based study in Estonia. Clin. Exp. Rheumatol. 25, 649–653 (2007).
Martínez Mengual, L., Fernández Menéndez, J. M. & Solís Sánchez, G. Epidemiological study of juvenile idiopathic arthtitis in the last sixteen years in Asturias (Spain). An. Pediatr. 66, 24–30 (2007).
Hanova, P., Pavelka, K., Dostal, C., Holcatova, I. & Pikhart, H. Epidemiology of rheumatoid arthritis, juvenile idiopathic arthritis and gout in two regions of the Czech Republic in a descriptive population-based survey in 2002–2003. Clin. Exp. Rheumatol. 24, 499–507 (2006).
Danner, S. et al. Epidemiology of juvenile idiopathic arthritis in Alsace, France. J. Rheumatol. 33, 1377–1381 (2006).
von Koskull, S., Truckenbrodt, H., Holle, R. & Hörmann, A. Incidence and prevalence of juvenile arthritis in an urban population of southern Germany: a prospective study. Ann. Rheum. Dis. 60, 940–945 (2001).
Manners, P. J. & Diepeveen, D. A. Prevalence of juvenile chronic arthritis in a population of 12-year-old children in urban Australia. Pediatrics 98, 84–90 (1996).
Gare, B. A. & Fasth, A. Epidemiology of juvenile chronic arthritis in southwestern Sweden: a 5-year prospective population study. Pediatrics 90, 950–958 (1992).
Oen, K. G. & Cheang, M. Epidemiology of chronic arthritis in childhood. Semin. Arthritis Rheum. 26, 575–591 (1996).
Gudmundsson, S. & Steinsson, K. Systemic lupus erythematosus in Iceland 1975 through 1984. A nationwide epidemiological study in an unselected population. J. Rheumatol. 17, 1162–1167 (1990).
Voss, A., Green, A. & Junker, P. Systemic lupus erythematosus in Denmark: clinical and epidemiological characterization of a county-based cohort. Scand. J. Rheumatol. 27, 98–105 (1998).
Nossent, J. C. Systemic lupus erythematosus on the Caribbean island of Curaçao: an epidemiological investigation. Ann. Rheum. Dis. 51, 1197–1201 (1992).
Nossent, H. C. Systemic lupus erythematosus in the Arctic region of Norway. J. Rheumatol. 28, 539–546 (2001).
Vilar, M. J. & Sato, E. I. Estimating the incidence of systemic lupus erythematosus in a tropical region (Natal, Brazil). Lupus 11, 528–532 (2002).
Lopez, P., Mozo, L., Gutierrez, C. & Suarez, A. Epidemiology of systemic lupus erythematosus in a northern Spanish population: gender and age influence on immunological features. Lupus 12, 860–865 (2003).
Deligny, C. et al. Systemic lupus erythematosus in Martinique: an epidemiologic study. Rev. Med. Interne 23, 21–29 (2002).
Somers, E. C., Thomas, S. L., Smeeth, L., Schoonen, W. M. & Hall, A. J. Incidence of systemic lupus erythematosus in the United Kingdom, 1990–1999. Arthritis Rheum. 57, 612–618 (2007).
Michelle, P. Epidemiology of systemic lupus erythematosus. Best Pract. Res. Clin. Rheumatol. 16, 847–858 (2002).
Arias-Nuñez, M. C. et al. Systemic sclerosis in northwestern Spain: a 19-year epidemiologic study. Medicine 87, 272–280 (2008).
Roberts-Thomson, P. J. et al. Scleroderma in South Australia: epidemiological observations of possible pathogenic significance. Intern. Med. J. 31, 220–229 (2001).
Geirsson, A. J., Steinsson, K., Guthmundsson, S. & Sigurthsson, V. Systemic sclerosis in Iceland. A nationwide epidemiological study. Ann. Rheum. Dis. 53, 502–505 (1994).
Alamanos, Y. et al. Epidemiology of systemic sclerosis in northwest Greece 1981 to 2002. Semin. Arthritis Rheum. 34, 714–720 (2005).
Tamaki, T., Mori, S. & Takehara, K. Epidemiological study of patients with systemic sclerosis in Tokyo. Arch. Dermatol. Res. 283, 366–371 (1991).
Le Guern, V. et al. Prevalence of systemic sclerosis in a French multi-ethnic county. Rheumatology (Oxford) 43, 1129–1137 (2004).
Onen, F. et al. Prevalence of ankylosing spondylitis and related spondyloarthritides in an urban area of Izmir, Turkey. J. Rheumatol. 35, 305–309 (2008).
Kaipiainen-Seppanen, O., Aho, K. & Heliovaara, M. Incidence and prevalence of ankylosing spondylitis in Finland. J. Rheumatol. 24, 496–499 (1997).
Alamanos, Y. et al. Epidemiology of ankylosing spondylitis in Northwest Greece, 1983–2002. Rheumatology (Oxford) 43, 615–618 (2004).
Carbone, L. D. et al. Ankylosing spondylitis in Rochester, Minnesota, 1935–1989: is the epidemiology changing? Arthritis Rheum. 35, 1476–1482 (1992).
Bakland, G., Nossent, H. C. & Gran, J. T. Incidence and prevalence of ankylosing spondylitis in Northern Norway. Arthritis Rheum. 53, 850–855 (2005).
Bowman, S. J., Ibrahim, G. H., Holmes, G., Hamburger, J. & Ainsworth, J. R. Estimating the prevalence among Caucasian women of primary Sjögren's syndrome in two general practices in Birmingham, UK. Scand. J. Rheumatol. 33, 39–43 (2004).
Pillemer, S. R. et al. Incidence of physician-diagnosed primary Sjögren's syndrome in residents of Olmsted County, Minnesota. Mayo Clin. Proc. 76, 593–599 (2001).
Thomas, E., Hay, E. M., Hajeer, A. & Silman, A. J. Sjögren's syndrome: a community-based study of prevalence and impact. Br. J. Rheumatol. 37, 1069–1076 (1998).
Bjerrum, K. B. Keratoconjunctivitis sicca and primary Sjögren's syndrome in a Danish population aged 30–60 years. Acta Ophthalmol. Scand. 75, 281–286 (1997).
Wakai, K. et al. Estimated prevalence of Sjögren's syndrome in Japan: findings from a nationwide epidemiological survey. J. Epidemiol. 5, 125–129 (1995).
Alamanos, Y. et al. Epidemiology of primary Sjögren's syndrome in north-west Greece, 1982–2003. Rheumatology (Oxford) 45, 187–191 (2006).
Plesivcnik Novljan, M. et al. Incidence of primary Sjögren's syndrome in Slovenia. Ann. Rheum. Dis. 63, 874–876 (2004).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Shapira, Y., Agmon-Levin, N. & Shoenfeld, Y. Geoepidemiology of autoimmune rheumatic diseases. Nat Rev Rheumatol 6, 468–476 (2010). https://doi.org/10.1038/nrrheum.2010.86
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2010.86
This article is cited by
-
Anti-Toxoplasma gondii antibodies as a risk factor for the prevalence and severity of systemic lupus erythematosus
Parasites & Vectors (2024)
-
Genome-wide identification of RNA modification-related single nucleotide polymorphisms associated with rheumatoid arthritis
BMC Genomics (2023)
-
Vitamin B12 (cobalamin) supplementation for the management of autoimmune rheumatic diseases: potential indications and opportunity for future research
Rheumatology International (2023)
-
Missing Causality and Heritability of Autoimmune Hepatitis
Digestive Diseases and Sciences (2023)
-
Entzündlich-rheumatische Erkrankungen bei Migranten
Die Innere Medizin (2023)