Abstract
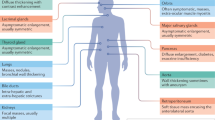
Patients with chronic inflammatory rheumatic diseases, such as rheumatoid arthritis (RA), systemic lupus erythematosus (SLE), and especially primary Sjögren's syndrome (SS), are at higher risk than the general population of developing B-cell non-Hodgkin lymphoma (NHL). Analyses of the association between various lymphoma subtypes and specific disease entities suggest that this association might be mediated by disease-specific mechanisms, as well as by mechanisms unique to lymphoma subtype. These specific associations can provide important information about abnormal B-cell stimulation in these conditions. Patients with primary SS, SLE and RA are at high risk of developing diffuse large B-cell lymphomas, a group of high-grade NHLs with remarkable heterogeneity. Patients with primary SS are at particularly high risk of developing marginal-zone B-cell lymphomas. The risk factors of lymphoma development in primary SS seem to be closely related to the underlying mechanisms of abnormal stimulation and/or impaired censoring mechanisms of B cells. In patients with RA and SLE, more intense disease activity and/or long-lasting disease might be indications of a higher risk of lymphoma development. This Review will focus on the risk of lymphoma, common and disease-specific mechanisms of B-cell lymphoma development, and on the clinical consequences of lymphoma in patients with inflammatory rheumatic diseases.
Key Points
-
Patients with primary Sjögren's syndrome (SS), systemic lupus erythematosus (SLE) and rheumatoid arthritis (RA) are at an increased risk of developing B-cell lymphomas, with the highest risk in patients with primary SS
-
Prevalence of lymphoma subtypes in patients with primary SS, SLE and RA might partly reflect underlying disease-specific mechanisms of abnormal B-cell activation
-
There are strong indications that many of these B-cell lymphomas depend on antigen binding and/or their local microenvironment, apart from the accumulation of molecular oncogenic lesions
-
In primary SS, clinicopathological risk factors have been identified that allow identification of patients who are at an increased risk of developing B-cell lymphoma and, therefore, warrant careful follow-up
-
Progress in B-cell targeted therapies, in particular by B-cell depletion with rituximab, markedly improves the therapeutic options for indolent and aggressive B-cell lymphomas
-
The clinical benefits resulting from B-cell depletion therapy further illustrate the pivotal role of B cells in the immunopathogenesis of inflammatory rheumatic diseases
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Delves PJ and Roitt IM (2000) The immune system. Second of two parts. N Engl J Med 343:108–117
Radbruch A et al. (2006) Competence and competition: the challenge of becoming a long-lived plasma cell. Nat Rev Immunol 6: 741–750
Youinou P et al. (2006) B lymphocytes in the front line of autoimmunity. Autoimmun Rev 5: 215–221
Lipsky PE (2001) Systemic lupus erythematosus: an autoimmune disease of B cell hyperactivity. Nat Immunol 2: 764–766
Dörner T and Lipsky PE (2002) Abnormalities of B cell phenotype, immunoglobulin gene expression and the emergence of autoimmunity in Sjögren's syndrome. Arthritis Res 4: 360–371
Fox RI (2005) Sjögren's syndrome. Lancet 366: 321–331
Weyand CM et al. (2005) B cells in rheumatoid synovitis. Arthritis Res Ther 7 (Suppl 3): S9–S12
Machold KP et al. (2006) Early rheumatoid arthritis. Curr Opin Rheumatol 18: 282–288
Zintsaras E et al. (2005) The risk of lymphoma development in autoimmune diseases: a meta-analysis. Arch Intern Med 165: 2337–2344
De Vita S and Quartuccio L (2006) Treatment of rheumatoid arthritis with rituximab: an update and possible indications. Autoimmun Rev 5: 443–448
Hennessy BT et al. (2004) Non-Hodgkin lymphoma: an update. Lancet Oncol 5: 341–353
Mackay IR and Rose NR (2001) Autoimmunity and lymphoma: tribulations of B cells. Nat Immunol 2: 793–795
Ruzickova S et al. (2002) Chronic lymphocytic leukemia preceded by cold-agglutinin disease: intraclonal immunoglobulin light-chain diversity in V(H) 4-34 expressing single leukemic B cells. Blood 100: 3419–3422 [Erratum in: Blood 2003; 101: 1676]
Geborek P et al. (2005) Tumor necrosis factor blockers do not increase overall risk in patients with rheumatoid arthritis, but may be associated with an increased risk of lymphomas. Ann Rheum Dis 64: 657–658
Setoguchi S et al. (2006) Tumor necrosis factor alpha antagonist use and cancer in patients with rheumatoid arthritis. Arthritis Rheum 54: 2757–2764
Bongartz T et al. (2006) Anti-TNF antibody therapy in rheumatoid arthritis and the risk of serious infections and malignancies: systemic review and meta-analysis of rare harmful effects in randomized controlled trials. JAMA 295: 2275–2285
Weller S et al. (2001) CD40-CD40L independent Ig gene hypermutation suggests a second B cell diversification pathway in humans. Proc Natl Acad Sci USA 98: 1166–1170
Voulgarelis M et al. (1999) Malignant lymphoma in primary Sjögren's syndrome. A multicenter, retrospective, clinical study by the European Concerted Action on Sjögren's syndrome. Arthritis Rheum 42: 1765–1772
Manganelli P et al. (2006) Hematologic manifestations of primary Sjögren's syndrome. Clin Exp Rheumatol 24: 438–448
Theander E et al. (2006) Lymphoma and other malignancies in primary Sjögren's syndrome: a cohort study on cancer incidence and lymphoma predictors. Ann Rheum Dis 65: 796–803
Smedby KE et al. (2006) Autoimmune and chronic inflammatory disorders and risk of non-Hodgkin lymphoma by subtype. J Natl Cancer Inst 98: 51–60
Monti S et al. (2005) Molecular profiling of diffuse large B-cell lymphoma identifies robust subtypes including one characterized by host inflammatory response. Blood 105: 1851–1861
Baecklund E et al. (2006) Characteristics of diffuse large B cell lymphomas in rheumatoid arthritis. Arthritis Rheum 54: 3774–3781
Landgren O et al. (2006) Autoimmunity and susceptibility to Hodgkin lymphoma: a population-based case-control study in Scandinavia. J Natl Cancer Inst 98: 1321–1330
Re D et al. (2005) Molecular pathogenesis of Hodgkin's lymphoma. J Clin Oncol 23: 6379–6386
Sánchez-Beato M et al. (2003) Cell cycle deregulation in B cell lymphomas. Blood 101: 1220–1235
Isaacson PG and Du MQ (2004) MALT lymphoma: from morphology to molecules. Nat Rev Cancer 4: 644–653
Carbone A and Gloghini A (2005) AIDS-related lymphomas: from pathogenesis to pathology. Br J Haematol 130: 662–670
Carreras J et al. (2006) High numbers of tumor-infiltrating FOXP3-positive regulatory T cell are associated with improved survival in follicular lymphoma. Blood 108: 2957–2964
Glas AM et al. (2007) Gene-expression and immunohistochemical study of specific T-cell subsets and accessory cell types in the transformation and prognosis of follicular lymphoma. J Clin Oncol 25: 390–398
Hasselblom S et al. (2007) The number of tumour-infiltrating TIA-1+ cytotoxic T cells but not FOXP3+ regulatory T cells predicts outcome in diffuse large B-cell lymphoma. Br J Haematol 137: 364–373
Jaffe ES (2002) Lymphoid lesions of the head and neck: a model of lymphocyte homing and lymphomagenesis. Mod Pathol 15: 255–263
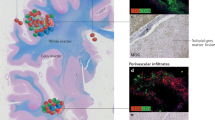
Salomonsson S et al. (2003) Cellular basis of ectopic germinal center formation and autoantibody production in the target organ of patients with Sjögren's syndrome. Arthritis Rheum 48: 3187–3201
Cupedo T and Mebius RE (2003) Role of chemokines in the development of secondary and tertiary lymphoid tissues. Semin Immunol 15: 243–248
Du M et al. (1996) Ongoing mutation in MALT lymphoma immunoglobulin gene suggests that antigen stimulation plays a role in clonal expansion. Leukemia 10: 1190–1197
Bahler S and Swerdlow SH (1998) Clonal salivary gland infiltrates associated with myoepithelial sialadenitis (Sjögren's syndrome) begin as nonmalignant antigen-selected expansions. Blood 91: 1864–1872
Martin T et al. (2000) Salivary gland lymphomas in patients with Sjögren's syndrome may frequently develop from rheumatoid factor B cells. Arthritis Rheum 43: 908–916
Hansen et al. (2006) Dissemination of a Sjögren's syndrome-associated extranodal marginal-zone B cell lymphoma: circulating lymphoma cells and invariant mutational pattern of nodal Ig heavy and light chain variable-region gene rearrangements. Arthritis Rheum 54: 127–137
Weller S et al. (2004) Human blood IgM “memory” B cells are circulating splenic marginal zone B cells harbouring a prediversified immunoglobulin repertoire. Blood 104: 3647–3654
Tsuiji M et al. (2006) A checkpoint for autoreactivity in human IgM+ memory B cell development. J Exp Med 203: 293–400
Sutherland AP et al. (2006) Targeting BAFF: immunomodulation for autoimmune diseases and lymphomas. Pharmacol Ther 112: 774–786
Ettinger R et al. (2007) IL21 & BAFF/BlyS synergize in stimulating plasma cell differentiation from a unique population of human splenic memory B cells. J Immunol 178: 2872–2882
Groom J et al. (2002) Association of BAFF/BLyS overexpression and altered B cell differentiation with Sjögren's syndrome. J Clin Invest 109: 59–68
Batten M et al. (2004) TNF deficiency fails to protect BAFF transgenic mice against autoimmunity and reveals a predisposition to B cell lymphoma. J Immunol 172: 812–822
Anaya JM et al. (1996) Clinicopathological factors relating malignant lymphoma with Sjögren's syndrome. Semin Arthritis Rheum 25: 337–346
Ramos-Casals M et al. (2005) Hepatitis C virus, Sjögren's syndrome and B-cell lymphoma: linking infection, autoimmunity and cancer. Autoimmun Rev 4: 8–15
Tedeschi A et al. (2007) Cryoglobulinemia. Blood Rev 21: 183–200
Fabris M et al. (2007) B-lymphocyte stimulator (BlyS) up-regulation in mixed cryoglobulinaemia syndrome and hepatitis-C virus infection. Rheumatology 46: 37–43
Tzioufas AG et al. (1996) Mixed monoclonal cryoglobulinemia and monoclonal rheumatoid factor cross-reactive idiotypes as predictive factors for the development of lymphoma in primary Sjögren's syndrome. Arthritis Rheum 39: 767–772
Ioannidis JP et al. (2002) Long-term risk of mortality and lymphoproliferative disease and predictive classification of primary Sjögren's syndrome. Arthritis Rheum 46: 741–747
De Vita S et al. (2001) Preliminary classification of nonmalignant B cell proliferation in Sjögren's syndrome: perspectives on pathobiology and treatment based on an integrated clinico-pathologic and molecular approach. Blood Cells Mol Dis 27: 757–766
Gottenberg JE et al. (2007) Serum immunoglobulin free light chain assessment in rheumatoid arthritis and Sjögren's syndrome. Ann Rheum Dis 66: 23–27
Bernatsky S et al. (2005) An international cohort study of cancer in systemic lupus erythematosus. Arthritis Rheum 52: 1481–1490
Raderer M et al. (2005) High relapse rate in patients with MALT lymphoma warrants lifelong follow-up. Clin Cancer Res 11: 3349–3352
Pijpe J et al. (2005) Rituximab treatment in patients with Sjögren's syndrome: an open-label phase II study. Arthritis Rheum 52: 2740–2750
Pijpe J et al. (2005) Changes in salivary gland immunohistology and function after rituximab monotherapy in a patient with Sjögren's syndrome and associated MALT lymphoma. Ann Rheum Dis 64: 958–960
Voulgarelis M et al. (2006) Long term remission of Sjögren's syndrome associated aggressive B cell non Hodgkin's lymphomas following combined B cell depletion therapy and CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone) Ann Rheum Dis 65: 1033–1037
Steinfeld SD et al. (2006) Epratuzumab (humanised anti-CD22 antibody) in primary Sjögren's syndrome: an open-label phase I/II study. Arthritis Res Ther 8: R129
Golenberg DM (2006) Epratuzumab in the therapy of oncological and immunological diseases. Expert Rev Anticancer Ther 6:1341–1353
Acknowledgements
This work was supported by Deutsche Forschungsgemeinschaft Grant Do 491/4-7.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Hansen, A., Lipsky, P. & Dörner, T. B-cell lymphoproliferation in chronic inflammatory rheumatic diseases. Nat Rev Rheumatol 3, 561–569 (2007). https://doi.org/10.1038/ncprheum0620
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncprheum0620
This article is cited by
-
Hematological malignancies in systemic lupus erythematosus: clinical characteristics, risk factors, and prognosis—a case-control study
Arthritis Research & Therapy (2022)
-
CP-25 alleviates antigen-induced experimental Sjögren's syndrome in mice by inhibiting JAK1-STAT1/2-CXCL13 signaling and interfering with B-cell migration
Laboratory Investigation (2021)
-
Primary Sjögren’s syndrome and B-non-Hodgkin lymphoma: role of CD4+ T lymphocytopenia
Rheumatology International (2013)
-
A case of IgG4-related disease associated with diffuse large B cell lymphoma
Clinical Journal of Gastroenterology (2013)
-
B-Cell Tolerance Defects in the B6.Aec1/2 Mouse Model of Sjögren’s Syndrome
Journal of Clinical Immunology (2012)