Abstract
Narcolepsy is a neurological disorder that is characterized by excessive daytime sleepiness and cataplexy—a loss of muscle tone generally triggered by certain strong emotions with sudden onset. The underlying cause of most cases of human narcolepsy is a loss of neurons that produce hypocretin (Hcrt, also known as orexin). These cells normally serve to drive and synchronize the activity of monoaminergic and cholinergic cells. Sleepiness results from the reduced activity of monoaminergic, cholinergic and other cells that are normally activated by Hcrt neurons, as well as from the loss of Hcrt itself. Cataplexy is caused by an episodic loss of activity in noradrenergic cells that support muscle tone, and a linked activation of a medial medullary cell population that suppresses muscle tone. Current treatments for narcolepsy include stimulants to combat sleepiness and antidepressants to reduce cataplexy. Sodium oxybate produces both reductions in cataplexy and improved waking alertness. Future treatments are likely to include Hcrt or Hcrt agonists to reverse the underlying neurochemical deficit.
Key Points
-
Narcolepsy is characterized by excessive daytime sleepiness, disrupted night-time sleep, cataplexy, sleep paralysis, hypnagogic hallucinations, and short latency from waking to the initiation of rapid eye movement sleep
-
Cataplexy most commonly occurs during behaviors accompanied by strong, positive emotions, with laughter being the most common trigger
-
In 2000, two simultaneously published papers determined that most human narcolepsy was linked to a loss of hypothalamic cells containing hypocretin (Hcrt, also known as orexin)
-
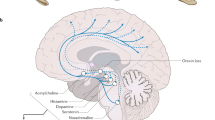
In the brain, Hcrt somata are found only in the hypothalamus; Hcrt cells project widely throughout the brain, and generally have excitatory effects on their postsynaptic cells
-
In brains from people with narcolepsy, only 10%, on average, of the normal numbers of Hcrt cells are seen; the loss of tonic Hcrt drive to histaminergic, dopaminergic, cholinergic and thalamic cells makes it difficult to sustain waking periods
-
The possibility that most cases of narcolepsy are autoimmune in etiology has led to several recent attempts to treat newly diagnosed narcolepsy by manipulating the immune system
-
There is evidence that Hcrt replacement therapy has the potential for reversing many of the symptoms of narcolepsy; several symptomatic treatments for narcolepsy are also available
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
American Academy of Sleep Medicine Nosology Committee (2005) International Classification of Sleep Disorders. Chicago: American Academy of Sleep Medicine
Westphal C (1877) Eigenthümliche mit Einschläfen verbundene Anfälle. Arch Psychiat 7: 631–635
Gelineau JBE (1881) De la narcolepsie. Surgères, Charente-Inferieure: Imprimerie de Surgères 64: 626–628
Peyron C et al. (2000) A mutation in a case of early onset narcolepsy and a generalized absence of hypocretin peptides in human narcoleptic brains. Nat Med 6: 991–997
Thannickal TC et al. (2000) Reduced number of hypocretin neurons in human narcolepsy. Neuron 27: 469–474
De Lecea L et al. (1998) The hypocretins: hypothalamus-specific peptides with neuroexcitatory activity. Proc Natl Acad Sci USA 95: 322–327
Sakurai T et al. (1998) Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 92: 573–585
Peyron C et al. (1998) Neurons containing hypocretin (orexin) project to multiple neuronal systems. J Neurosci 18: 9996–10015
Siegel JM (2004) Hypocretin (orexin): role in normal behavior and neuropathology. Annu Rev Psychol 55: 125–148
Rosin DL et al. (2003) Hypothalamic orexin (hypocretin) neurons express vesicular glutamate transporters VGLUT1 or VGLUT2. J Comp Neurol 465: 593–603
van den Pol AN et al. (1998) Presynaptic and postsynaptic actions and modulation of neuroendocrine neurons by a new hypothalamic peptide, hypocretin/orexin. J Neurosci 18: 7962–7971
Peever JH et al. (2003) Excitatory effects of hypocretin-1 (orexin-A) in the trigeminal motor nucleus are reversed by NMDA antagonism. J Neurophysiol 89: 2591–2600
John J et al. (2003) Intravenously administered hypocretin-1 alters brain amino acid release: an in vivo microdialysis study in rats. J Physiol (Lond) 548.2: 557–562
Li Y et al. (2002) Hypocretin/orexin excites hypocretin neurons via a local glutamate neuron—a potential mechanism for orchestrating the hypothalamic arousal system. Neuron 36: 1169–1181
Grivel J et al. (2005) The wake-promoting hypocretin/orexin neurons change their response to noradrenaline after sleep deprivation. J Neurosci 25: 4127–4130
Nishino S et al. (2000) Hypocretin (orexin) deficiency in human narcolepsy. Lancet 355: 39–41
Ripley B et al. (2001) CSF hypocretin/orexin levels in narcolepsy and other neurological conditions. Neurology 57: 2253–2258
Gerashchenko D et al. (2003) Relationship between CSF hypocretin levels and hypocretin neuronal loss. Exp Neurol 184: 1010–1016
Wu MF et al. (2002) Hypocretin release in normal and narcoleptic dogs after food and sleep deprivation, eating, and movement. Am J Physiol Regul Integr Comp Physiol 283: R1079–R1086
Thannickal TC et al. (2003) Pattern of hypocretin (orexin) soma and axon loss, and gliosis, in human narcolepsy. Brain Pathol 13: 340–351
Blouin AM et al. (2005) Narp immunostaining of human hypocretin (orexin) neurons: loss in narcolepsy. Neurology 65: 1189–1192
Crocker A et al. (2005) Concomitant loss of dynorphin, NARP, and orexin in narcolepsy. Neurology 65: 1184–1188
Montplaisir J and Poirier G (1988) HLA in narcolepsy in Canada. In HLA in Narcolepsy, 97–107 (Eds Honda Y and Juji T) Berlin: Springer-Verlag
Tachibana N et al. (2005) Hypersomnolence and increased REM sleep with low cerebrospinal fluid hypocretin level in a patient after removal of craniopharyngioma. Sleep Med 6: 567–569
Nishino S and Kanbayashi T (2005) Symptomatic narcolepsy, cataplexy and hypersomnia, and their implications in the hypothalamic hypocretin/orexin system. Sleep Med Rev 9: 269–310
Partinen M et al. (1994) Twin studies in narcolepsy. Sleep 17 (8 Suppl): S13–S16
Honda Y et al. (1984) Narcolepsy and HLA: positive DR2 as a prerequisite for the development of narcolepsy. Folia Psychiatr Neurol Jpn 38: 360
Mignot E et al. (2001) Complex HLA-DR and -DQ interactions confer risk of narcolepsy-cataplexy in three ethnic groups. Am J Hum Genet 68: 686–699
Sinha AA et al. (1990) Autoimmune diseases: the failure of self tolerance. Science 248: 1380–1388
Overeem S et al. (2006) Immunohistochemical screening for autoantibodies against lateral hypothalamic neurons in human narcolepsy. J Neuroimmunol 174: 187–191
Overeem S et al. (2003) Screening for anti-ganglioside antibodies in hypocretin-deficient human narcolepsy. Neurosci Lett 341: 13–16
Black JL III et al. (2002) Search for neuron-specific and nonneuron-specific antibodies in narcoleptic patients with and without HLA DQB1*0602. Sleep 25: 719–723
John J et al. (2004) Cataplexy-active neurons in the posterior hypothalamus: implications for the role of histamine in sleep and waking behavior. Neuron 42: 619–634
Siegel JM et al. (1991) Neuronal activity in narcolepsy: identification of cataplexy related cells in the medial medulla. Science 252: 1315–1318
Wu MF et al. (1999) Locus coeruleus neurons: cessation of activity during cataplexy. Neuroscience 91: 1389–1399
Mileykovskiy BY et al. (2005) Behavioral correlates of activity in identified hypocretin/orexin neurons. Neuron 46: 787–798
Beuckmann CT et al. (2004) Expression of a poly-glutamine-ataxin-3 transgene in orexin neurons induces narcolepsy-cataplexy in the rat. J Neurosci 24: 4469–4477
Guilleminault C (1976) Cataplexy. In Narcolepsy, 125–143 (Eds Guilleminault C et al.) New York: Spectrum
Lee MG et al. (2005) Discharge of identified orexin/hypocretin neurons across the sleep-waking cycle. J Neurosci 25: 6716–6720
Hecht M et al. (2003) Report of a case of immunosuppression with prednisone in an 8-year-old boy with an acute onset of hypocretin-deficiency narcolepsy. Sleep 26: 809–810
Dauvilliers Y et al. (2004) Successful management of cataplexy with intravenous immunoglobulins at narcolepsy onset. Ann Neurol 56: 905–908
Lecendreux M et al. (2003) Clinical efficacy of high-dose intravenous immunoglobulins near the onset of narcolepsy in a 10-year-old boy. J Sleep Res 12: 347–348
Boehmer LN et al. (2004) Treatment with immunosuppressive and anti-inflammatory agents delays onset of canine genetic narcolepsy and reduces symptom severity. Exp Neurol 188: 292–299
Kastin AJ and Akerstrom V (1999) Orexin A but not orexin B rapidly enters brain from blood by simple diffusion. J Pharmacol Exp Ther 289: 219–223
John J et al. (2000) Hypocretin-1 reduces cataplexy and normalizes sleep and waking durations in narcoleptic dogs. Sleep 23: A12
Siegel JM (2003) Hypocretin administration as a treatment for human narcolepsy. Sleep 26: 932–933
Fujiki N et al. (2003) Effects of IV and ICV hypocretin-1 (orexin A) in hypocretin receptor-2 gene mutated narcoleptic dogs and IV hypocretin-1 replacement therapy in a hypocretin-ligand-deficient narcoleptic dog. Sleep 26: 953–959
Barreiro ML et al. (2005) Pattern of orexin expression and direct biological actions of orexin-A in rat testis. Endocrinology 146: 5164–5175
Zhang S et al. (2005) Expression of orexin receptors in the brain and peripheral tissues of the male sheep. Regul Pept 124: 81–87
Ehrstrom M et al. (2005) Stimulatory effect of endogenous orexin A on gastric emptying and acid secretion independent of gastrin. Regul Pept 132: 9–16
Kirchgessner AL (2002) Orexins in the brain–gut axis. Endocr Rev 23: 1–15
Mieda M et al. (2004) Orexin peptides prevent cataplexy and improve wakefulness in an orexin neuron-ablated model of narcolepsy in mice. Proc Natl Acad Sci USA 101: 4649–4654
Hanson LR et al. (2004) Intranasal administration of hypocretin 1 (orexin A) bypasses the blood–brain barrier and targets the brain: a new strategy for the treatment of narcolepsy. Drug Delivery Technology 4: 66–71
Born J et al. (2002) Sniffing neuropeptides: a transnasal approach to the human brain. Nat Neurosci 5: 514–516
Wisor JP and Eriksson KS (2005) Dopaminergic-adrenergic interactions in the wake promoting mechanism of modafinil. Neuroscience 132: 1027–1034
Willie JT et al. (2005) Modafinil more effectively induces wakefulness in orexin-null mice than in wild-type littermates. Neuroscience 130: 983–995
Mignot E and Nishino S (2005) Emerging therapies in narcolepsy–cataplexy. Sleep 28: 754–763
Carter LP et al. (2006) Discriminative stimulus effects of GHB and GABAB agonists are differentially attenuated by CGP35348. Eur J Pharmacol 538: 85–93
Acknowledgements
Supported by US PHS grants NS14610, MH64109 and the Medical Research Service of the Department of Veterans Affairs. We thank Adam Siegel for drawing Figure 1.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Siegel, J., Boehmer, L. Narcolepsy and the hypocretin system—where motion meets emotion. Nat Rev Neurol 2, 548–556 (2006). https://doi.org/10.1038/ncpneuro0300
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpneuro0300
This article is cited by
-
Cataplexy—clinical aspects, pathophysiology and management strategy
Nature Reviews Neurology (2014)
-
Waking with the hypothalamus
Pflügers Archiv - European Journal of Physiology (2012)
-
Einsatz intravenöser Immunglobuline in der Neurologie
Der Nervenarzt (2011)