Key Points
-
In patients with autosomal dominant polycystic kidney disease (ADPKD), rates of patient and graft survival following kidney transplantation are excellent
-
Prophylactic native nephrectomy is restricted to patients with severe pain, early satiety, recurrent bleeding, infections or stones and those in whom space must be made to implant the graft
-
Patients with liver involvement require pretransplant imaging; combined liver–kidney transplantation should be considered in cases of symptomatic hepatomegaly or recurrent cholangitis if the glomerular filtration rate is ≤30 ml/min/1.73 m2
-
General recommendations for the screening of intracranial aneurysms in patients with ADPKD should be used to select patients for pretransplant screening
-
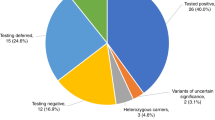
Genetic testing should be done in living related-donor candidates aged <30 years when imaging is normal or equivocal
-
No reason exists to preferentially use mammalian target of rapamycin inhibitors as immunosuppressive agents in kidney transplant recipients with ADPKD
Abstract
In patients with autosomal dominant polycystic kidney disease (ADPKD) evaluated for kidney transplantation, issues related to native nephrectomy, cystic liver involvement, screening for intracranial aneurysms and living-related kidney donation deserve special consideration. Prophylactic native nephrectomy is restricted to patients with a history of cyst infection or recurrent haemorrhage or to those in whom space must be made to implant the graft. Patients with liver involvement require pretransplant imaging. Selection of patients for pretransplant screening of intracranial aneurysms should follow the general recommendations for patients with ADPKD. In living related-donor candidates aged <30 years and at-risk of ADPKD, molecular genetic testing should be carried out when ultrasonography and MRI findings are normal or equivocal. After kidney transplantation, patient and graft survival rates are excellent and the volume of native kidneys decreases. However, liver cysts continue to grow and treatment with a somatostatin analogue should be considered in patients with massive cyst involvement. Cerebrovascular events have a marginal effect on post-transplant morbidity and mortality. An increased risk of new-onset diabetes mellitus and nonmelanoma skin cancers has been reported, but several studies have challenged these findings. Finally, no data currently support the preferential use of mammalian target of rapamycin inhibitors as immunosuppressive agents in transplant recipients with ADPKD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Torres, V. E., Harris, P. C. & Pirson, Y. Autosomal dominant polycystic kidney disease. Lancet 369, 1287–1301 (2007).
Harris, P. C. & Torres, V. E. Polycystic kidney disease. Annu. Rev. Med. 60, 321–337 (2009).
Ong, A. C. & Harris, P. C. Molecular pathogenesis of ADPKD: the polycystin complex gets complex. Kidney Int. 67, 1234–1247 (2005).
Iglesias, C. G. et al. Epidemiology of adult polycystic kidney disease, Olmsted County, Minnesota: 1935–1980 Am. J. Kidney Dis. 2, 630–639 (1983).
Davies, F. et al. Polycystic kidney disease re-evaluated: a population-based study. Q. J. Med. 79, 477–485 (1991).
Collins, A. J. et al. Excerpts from the United States Renal Data System 2004 annual data report: atlas of end-stage renal disease in the United States. Am. J. Kidney Dis. 45 (Suppl. 1), A5–A7, S1–S280 (2005).
Pirson, Y. Extrarenal manifestations of autosomal dominant polycystic kidney disease. Adv. Chronic. Kidney Dis. 17, 173–180 (2010).
Luciano, R. L. & Dahl, N. K. Extra-renal manifestations of autosomal dominant polycystic kidney disease (ADPKD): considerations for routine screening and management. Nephrol. Dial. Transplant. 29, 247–254 (2014).
Torra, R. et al. Prevalence of cysts in seminal tract and abnormal semen parameters in patients with autosomal dominant polycystic kidney disease. Clin. J. Am. Soc. Nephrol. 3, 790–793 (2008).
Kumar, S., Adeva, M., King, B. F., Kamath, P. S. & Torres, V. E. Duodenal diverticulosis in autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 21, 3576–3578 (2006).
Driscoll, J. A., Bhalla, S., Liapis, H., Ibricevic, A. & Brody, S. L. Autosomal dominant polycystic kidney disease is associated with an increased prevalence of radiographic bronchiectasis. Chest 133, 1181–1188 (2008).
Harris, P. C. et al. Cyst number but not the rate of cystic growth is associated with the mutated gene in autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 17, 3013–3019 (2006).
Dobin, A. et al. Segregation analysis of autosomal dominant polycystic kidney disease. Genet. Epidemiol. 10, 189–200 (1993).
The International Polycystic Kidney Disease Consortium. Polycystic kidney disease: the complete structure of the PKD1 gene and its protein. Cell 81, 289–298 (1995).
Hughes, J. et al. The polycystic kidney disease 1 (PKD1) gene encodes a novel protein with multiple cell recognition domains. Nat. Genet. 10, 151–160 (1995).
Mochizuki, T. et al. PKD2, a gene for polycystic kidney disease that encodes an integral membrane protein. Science 272, 1339–1342 (1996).
Yoder, B. K. Role of primary cilia in the pathogenesis of polycystic kidney disease. J. Am. Soc. Nephrol. 18, 1381–1388 (2007).
Hateboer, N. et al. Comparison of phenotypes of polycystic kidney disease types 1 and 2. European PKD1-PKD2 Study Group. Lancet 353, 103–107 (1999).
Cornec-Le, G. E. et al. Type of PKD1 mutation influences renal outcome in ADPKD. J. Am. Soc. Nephrol. 24, 1006–1013 (2013).
Paterson, A. D. et al. Progressive loss of renal function is an age-dependent heritable trait in type 1 autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 16, 755–762 (2005).
Fain, P. R. et al. Modifier genes play a significant role in the phenotypic expression of PKD1. Kidney Int. 67, 1256–1267 (2005).
Magistroni, R. et al. Genotype-renal function correlation in type 2 autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 14, 1164–1174 (2003).
Persu, A. et al. Comparison between siblings and twins supports a role for modifier genes in ADPKD. Kidney Int. 66, 2132–2136 (2004).
Alam, A. & Perrone, R. D. Management of ESRD in patients with autosomal dominant polycystic kidney disease. Adv. Chronic. Kidney Dis. 17, 164–172 (2010).
Haynes, R., Kheradmand, F. & Winearls, C. G. Survival after starting renal replacement treatment in patients with autosomal dominant polycystic kidney disease: a single-centre 40-year study. Nephron Clin. Pract. 120, c42–c47 (2012).
Mosconi, G. et al. Renal transplant in patients with polycystic disease: the Italian experience. Transplant. Proc. 45, 2635–2640 (2013).
Jacquet, A. et al. Outcomes of renal transplantation in patients with autosomal dominant polycystic kidney disease: a nationwide longitudinal study. Transpl. Int. 24, 582–587 (2011).
Orskov, B., Romming Sorensen, V., Feldt-Rasmussen, B. & Strandgaard, S. Improved prognosis in patients with autosomal dominant polycystic kidney disease in Denmark. Clin. J. Am. Soc. Nephrol. 5, 2034–2039 (2010).
Perrone, R. D., Ruthazer, R. & Terrin, N. C. Survival after end-stage renal disease in autosomal dominant polycystic kidney disease: contribution of extrarenal complications to mortality. Am. J. Kidney Dis. 38, 777–784 (2001).
Spithoven, E. M. et al. Renal replacement therapy for ADPKD in Europe: prevalence and survival. An analysis of data from the ERA-EDTA Registry. Nephrol. Dial. Transplant. (in press).
Kirkman, M. A. et al. Native nephrectomy for autosomal dominant polycystic kidney disease: before or after kidney transplantation? BJU Int. 108, 590–594 (2011).
Bennett, W. M. Peritransplant management of retained native kidneys in autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 28, 245–246 (2013).
Kasiske, B. L. et al. The evaluation of renal transplant candidates: clinical practice guidelines. Patient Care and Education Committee of the American Society of Transplant Physicians. J. Am. Soc. Nephrol. 6, 1–34 (1995).
European Renal Best Practice Transplantation Guideline Development Group. ERBP Guideline on the Management and Evaluation of the Kidney Donor and Recipient. Nephrol. Dial. Transplant. 28 (Suppl. 2), ii1–ii71 (2013).
Yarimizu, S. N. et al. Mortality and morbidity in pretransplant bilateral nephrectomy: analysis of 305 cases. Urology 12, 55–58 (1978).
Mendelssohn, D. C., Harding, M. E., Cardella, C. J., Cook, G. T. & Uldall, P. R. Management of end-stage autosomal dominant polycystic kidney disease with hemodialysis and transplantation. Clin. Nephrol. 30, 315–319 (1988).
Patel, P. et al. Native nephrectomy in transplant patients with autosomal dominant polycystic kidney disease. Ann. R. Coll. Surg. Engl. 93, 391–395 (2011).
Pirson, Y. in Oxford Textbook of Clinical Nephrology (Ed. Davison, A. M.) 2304–2323 (Liepman, 2005).
Neeff, H. P. et al. One hundred consecutive kidney transplantations with simultaneous ipsilateral nephrectomy in patients with autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 28, 466–471 (2013).
Cornelis, F. et al. Embolization of polycystic kidneys as an alternative to nephrectomy before renal transplantation: a pilot study. Am. J. Transplant. 10, 2363–2369 (2010).
Wagner, M. D., Prather, J. C. & Barry, J. M. Selective, concurrent bilateral nephrectomies at renal transplantation for autosomal dominant polycystic kidney disease. J. Urol. 177, 2250–2254 (2007).
Kramer, A. et al. Simultaneous bilateral native nephrectomy and living donor renal transplantation are successful for polycystic kidney disease: the University of Maryland experience. J. Urol. 181, 724–728 (2009).
Martin, A. D. et al. Laparoscopic bilateral native nephrectomies with simultaneous kidney transplantation. BJU Int. 110, E1003–E1007 (2012).
Verhoest, G. et al. Transperitoneal laparoscopic nephrectomy for autosomal dominant polycystic kidney disease. JSLS 16, 437–442 (2012).
Gevers, T. J. & Drenth, J. P. Diagnosis and management of polycystic liver disease. Nat. Rev. Gastroenterol. Hepatol. 10, 101–108 (2013).
Strazzabosco, M. & Somlo, S. Polycystic liver diseases: congenital disorders of cholangiocyte signaling. Gastroenterology 140, 1855–1859. e1 (2011).
Bae, K. T. et al. Magnetic resonance imaging evaluation of hepatic cysts in early autosomal-dominant polycystic kidney disease: the Consortium for Radiologic Imaging Studies of Polycystic Kidney Disease cohort. Clin. J. Am. Soc. Nephrol. 1, 64–69 (2006).
Alvaro, D. et al. Estrogens and insulin-like growth factor 1 modulate neoplastic cell growth in human cholangiocarcinoma. Am. J. Pathol. 169, 877–888 (2006).
Chapman, A. B. Cystic disease in women: clinical characteristics and medical management. Adv. Ren. Replace. Ther. 10, 24–30 (2003).
Sherstha, R. et al. Postmenopausal estrogen therapy selectively stimulates hepatic enlargement in women with autosomal dominant polycystic kidney disease. Hepatology 26, 1282–1286 (1997).
Caroli, A. et al. Reducing polycystic liver volume in ADPKD: effects of somatostatin analogue octreotide. Clin. J. Am. Soc. Nephrol. 5, 783–789 (2010).
Hogan, M. C. et al. Randomized clinical trial of long-acting somatostatin for autosomal dominant polycystic kidney and liver disease. J. Am. Soc. Nephrol. 21, 1052–1061 (2010).
van Keimpema, L. et al. Lanreotide reduces the volume of polycystic liver: a randomized, double-blind, placebo-controlled trial. Gastroenterology 137, 1661–1668 (2009).
Grunfeld, J. P. et al. Liver changes and complications in adult polycystic kidney disease. Adv. Nephrol. Necker Hosp. 14, 1–20 (1985).
Jouret, F. et al. Positron-emission computed tomography in cyst infection diagnosis in patients with autosomal dominant polycystic kidney disease. Clin. J. Am. Soc. Nephrol. 6, 1644–1650 (2011).
Bleeker-Rovers, C. P. et al. Diagnosis of renal and hepatic cyst infections by 18F-fluorodeoxyglucose positron emission tomography in autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 41, E18–E21 (2003).
Jouret, F. et al. Diagnosis of cyst infection in patients with autosomal dominant polycystic kidney disease: attributes and limitations of the current modalities. Nephrol. Dial. Transplant. 2, 3746–3751 (2012).
Sallee, M. et al. Cyst infections in patients with autosomal dominant polycystic kidney disease. Clin. J. Am. Soc. Nephrol. 4, 1183–1189 (2009).
Telenti, A. et al. Hepatic cyst infection in autosomal dominant polycystic kidney disease. Mayo Clin. Proc. 65, 933–942 (1990).
Chauveau, D., Fakhouri, F. & Grunfeld, J. P. Liver involvement in autosomal-dominant polycystic kidney disease: therapeutic dilemma. J. Am. Soc. Nephrol. 11, 1767–1775 (2000).
Kanaan, N., Goffin, E., Pirson, Y., Devuyst, O. & Hassoun, Z. Carbohydrate antigen 19-9 as a diagnostic marker for hepatic cyst infection in autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 55, 916–922 (2010).
Waanders, E. et al. Carbohydrate antigen 19-9 is extremely elevated in polycystic liver disease. Liver Int. 29, 1389–1395 (2009).
van, K. L. et al. Patients with isolated polycystic liver disease referred to liver centres: clinical characterization of 137 cases. Liver Int. 31, 92–98 (2011).
Torres, V. E. & Harris, P. C. Autosomal dominant polycystic kidney disease: the last 3 years. Kidney Int. 76, 149–168 (2009).
Hoevenaren, I. A. et al. Polycystic liver: clinical characteristics of patients with isolated polycystic liver disease compared with patients with polycystic liver and autosomal dominant polycystic kidney disease. Liver Int. 28, 264–270 (2008).
Van, K. L. & Hockerstedt, K. Treatment of polycystic liver disease. Br. J. Surg. 96, 1379–1380 (2009).
Ruggenenti, P. et al. Safety and efficacy of long-acting somatostatin treatment in autosomal-dominant polycystic kidney disease. Kidney Int. 68, 206–216 (2005).
Hogan, M. C. et al. Somatostatin analog therapy for severe polycystic liver disease: results after 2 years. Nephrol. Dial. Transplant. 27, 3532–3539 (2012).
Russell, R. T. & Pinson, C. W. Surgical management of polycystic liver disease. World J. Gastroenterol. 13, 5052–5059 (2007).
Schnelldorfer, T., Torres, V. E., Zakaria, S., Rosen, C. B. & Nagorney, D. M. Polycystic liver disease: a critical appraisal of hepatic resection, cyst fenestration, and liver transplantation. Ann. Surg. 250, 112–118 (2009).
Nadim, M. K., Davis, C. L., Sung, R., Kellum, J. A. & Genyk, Y. S. Simultaneous liver–kidney transplantation: a survey of US transplant centers. Am. J. Transplant. 12, 3119–3127 (2012).
Davis, C. L. Impact of pretransplant renal failure: when is listing for kidney-liver indicated? Liver Transpl. 11, S35–S44 (2005).
Drenth, J. P., Chrispijn, M., Nagorney, D. M., Kamath, P. S. & Torres, V. E. Medical and surgical treatment options for polycystic liver disease. Hepatology 52, 2223–2230 (2010).
Fong, T. L., Bunnapradist, S., Jordan, S. C., Selby, R. R. & Cho, Y. W. Analysis of the United Network for Organ Sharing database comparing renal allografts and patient survival in combined liver–kidney transplantation with the contralateral allografts in kidney alone or kidney–pancreas transplantation. Transplantation 76, 348–353 (2003).
Kirchner, G. I. et al. Outcome and quality of life in patients with polycystic liver disease after liver or combined liver–kidney transplantation. Liver Transpl. 12, 1268–1277 (2006).
Torra, R. et al. Autosomal dominant polycystic kidney disease with anticipation and Caroli's disease associated with a PKD1 mutation. Kidney Int. 52, 33–38 (1997).
Shedda, S. & Robertson, A. Caroli's syndrome and adult polycystic kidney disease. ANZ J. Surg. 77, 292–294 (2007).
Jordon, D., Harpaz, N. & Thung, S. N. Caroli's disease and adult polycystic kidney disease: a rarely recognized association. Liver 9, 30–35 (1989).
Aguilar, M., Meterissian, S., Levesque, S. & Andonian, S. Nephrectomy in patients with Caroli's and ADPKD may be associated with increased morbidity. Can. Urol. Assoc. J. 5, E19–E22 (2011).
Habib, S. et al. Caroli's disease and orthotopic liver transplantation. Liver Transpl. 12, 416–421 (2006).
Yamamoto, T. et al. Kidney volume changes in patients with autosomal dominant polycystic kidney disease after renal transplantation. Transplantation 93, 794–798 (2012).
Temmerman, F. et al. Safety and efficacy of different lanreotide doses in the treatment of polycystic liver disease: pooled analysis of individual patient data. Aliment. Pharmacol. Ther. 38, 397–406 (2013).
Pirson, Y., Chauveau, D. & Torres, V. Management of cerebral aneurysms in autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 13, 269–276 (2002).
Irazabal, M. V. et al. Extended follow-up of unruptured intracranial aneurysms detected by presymptomatic screening in patients with autosomal dominant polycystic kidney disease. Clin. J. Am. Soc. Nephrol. 6, 1274–1285 (2011).
Chauveau, D. et al. Intracranial aneurysms in autosomal dominant polycystic kidney disease. Kidney Int. 45, 1140–1146 (1994).
Rinkel, G. J. Natural history, epidemiology and screening of unruptured intracranial aneurysms. J. Neuroradiol. 35, 99–103 (2008).
Broderick, J. P. et al. Greater rupture risk for familial as compared to sporadic unruptured intracranial aneurysms. Stroke 40, 1952–1957 (2009).
Wiebers, D. O. et al. Unruptured intracranial aneurysms: natural history, clinical outcome, and risks of surgical and endovascular treatment. Lancet 362, 103–110 (2003).
Ishibashi, T. et al. Unruptured intracranial aneurysms: incidence of rupture and risk factors. Stroke 40, 313–316 (2009).
Vlak, M. H., Algra, A., Brandenburg, R. & Rinkel, G. J. Prevalence of unruptured intracranial aneurysms, with emphasis on sex, age, comorbidity, country, and time period: a systematic review and meta-analysis. Lancet Neurol. 10, 626–636 (2011).
Li, L. M., Bulters, D. O. & Kirollos, R. W. A mathematical model of utility for single screening of asymptomatic unruptured intracranial aneurysms at the age of 50 years. Acta Neurochir. (Wien.) 154, 1145–1152 (2012).
Schrier, R. W. et al. Repeat imaging for intracranial aneurysms in patients with autosomal dominant polycystic kidney disease with initially negative studies: a prospective ten-year follow-up. J. Am. Soc. Nephrol. 15, 1023–1028 (2004).
Belz, M. M. et al. Recurrence of intracranial aneurysms in autosomal-dominant polycystic kidney disease. Kidney Int. 63, 1824–1830 (2003).
Gibbs, G. F. et al. Follow-up of intracranial aneurysms in autosomal-dominant polycystic kidney disease. Kidney Int. 65, 1621–1627 (2004).
Oliveras, A. et al. Stroke in renal transplant recipients: epidemiology, predictive risk factors and outcome. Clin. Transplant. 17, 1–8 (2003).
Abedini, S. et al. Cerebrovascular events in renal transplant recipients. Transplantation 87, 112–117 (2009).
Hadimeri, H., Norden, G., Friman, S. & Nyberg, G. Autosomal dominant polycystic kidney disease in a kidney transplant population. Nephrol. Dial. Transplant. 12, 1431–1436 (1997).
Fick, G. M., Johnson, A. M., Hammond, W. S. & Gabow, P. A. Causes of death in autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 5, 2048–2056 (1995).
Yoo, D. J. Agodoa, L., Yuan, C. M., Abbott, K. C. & Nee, R. Risk of intracranial hemorrhage associated with autosomal dominant polycystic kidney disease in patients with end stage renal disease. BMC Nephrology 15, 39–46 (2014).
Ravine, D. et al. Evaluation of ultrasonographic diagnostic criteria for autosomal dominant polycystic kidney disease 1. Lancet 343, 824–827 (1994).
Pei, Y. et al. Unified criteria for ultrasonographic diagnosis of ADPKD. J. Am. Soc. Nephrol. 20, 205–212 (2009).
Peral, B. et al. Evidence of linkage disequilibrium in the Spanish polycystic kidney disease I population. Am. J. Hum. Genet. 54, 899–908 (1994).
Zhao, X. et al. Molecular diagnostics in autosomal dominant polycystic kidney disease: utility and limitations. Clin. J. Am. Soc. Nephrol. 3, 146–152 (2008).
Rossetti, S. et al. Comprehensive molecular diagnostics in autosomal dominant polycystic kidney disease. J. Am. Soc. Nephrol. 18, 2143–2160 (2007).
Garcia-Gonzalez, M. A. et al. Evaluating the clinical utility of a molecular genetic test for polycystic kidney disease. Mol. Genet. Metab. 92, 160–167 (2007).
Harris, P. C. & Rossetti, S. Molecular diagnostics for autosomal dominant polycystic kidney disease. Nat. Rev. Nephrol. 6, 197–206 (2010).
Audrezet, M. P. et al. Autosomal dominant polycystic kidney disease: comprehensive mutation analysis of PKD1 and PKD2 in 700 unrelated patients. Hum. Mutat. 33, 1239–1250 (2012).
Barua, M. et al. Family history of renal disease severity predicts the mutated gene in ADPKD. J. Am. Soc. Nephrol. 20, 1833–1838 (2009).
Fitzpatrick, P. M. et al. Long-term outcome of renal transplantation in autosomal dominant polycystic kidney disease. Am. J. Kidney Dis. 15, 535–543 (1990).
Florijn, K. W., Chang, P. C., van der Woude, F. J., van Bockel, J. H. & van Saase, J. L. Long-term cardiovascular morbidity and mortality in autosomal dominant polycystic kidney disease patients after renal transplantation. Transplantation 57, 73–81 (1994).
Valderrábano, F., Jones, E. H. & Mallick, N. P. Report on management of renal failure in Europe, XXIV, 1993. Nephrol. Dial.Transplant. 10 (Suppl. 5), 1–25 (1995).
Pirson, Y., Christophe, J. L. & Goffin, E. Outcome of renal replacement therapy in autosomal dominant polycystic kidney disease. Nephrol. Dial. Transplant. 11 (Suppl. 6), 24–28 (1996).
Thaysen, J. H., Thomsen, H. S., Sass, A. & Kristensen, J. K. Volume changes in polycystic kidneys during chronic dialysis and after renal transplantation. Acta Med. Scand. 217, 197–204 (1985).
Ishikawa, I., Tateishi, K., Kitada, H. & Shinoda, A. Regression of adult type polycystic kidneys during chronic intermittent hemodialysis. Is it a universal phenomenon? Nephron 36, 147 (1984).
Ho, T. A. et al. Autosomal dominant polycystic kidney disease is associated with central and nephrogenic defects in osmoregulation. Kidney Int. 82, 1121–1129 (2012).
Ecder, T. & Schrier, R. W. Hypertension in autosomal-dominant polycystic kidney disease: early occurrence and unique aspects. J. Am. Soc. Nephrol. 12, 194–200 (2001).
Ecder, T. & Schrier, R. W. Cardiovascular abnormalities in autosomal-dominant polycystic kidney disease. Nat. Rev. Nephrol. 5, 221–228 (2009).
Hossack, K. F., Leddy, C. L., Johnson, A. M., Schrier, R. W. & Gabow, P. A. Echocardiographic findings in autosomal dominant polycystic kidney disease. N. Engl. J. Med. 319, 907–912 (1988).
Lumiaho, A. et al. Mitral valve prolapse and mitral regurgitation are common in patients with polycystic kidney disease type 1. Am. J. Kidney Dis. 38, 1208–1216 (2001).
Qian, Q., Hartman, R. P., King, B. F. & Torres, V. E. Increased occurrence of pericardial effusion in patients with autosomal dominant polycystic kidney disease. Clin. J. Am. Soc. Nephrol. 2, 1223–1227 (2007).
Ducloux, D. et al. Polycystic kidney disease as a risk factor for post-transplant diabetes mellitus. Nephrol. Dial. Transplant. 14, 1244–1246 (1999).
de Mattos, A. M. et al. Autosomal-dominant polycystic kidney disease as a risk factor for diabetes mellitus following renal transplantation. Kidney Int. 67, 714–720 (2005).
Hamer, R. A., Chow, C. L., Ong, A. C. & McKane, W. S. Polycystic kidney disease is a risk factor for new-onset diabetes after transplantation. Transplantation 83, 36–40 (2007).
Pietrzak-Nowacka, M. et al. Autosomal dominant polycystic kidney disease is not a risk factor for post-transplant diabetes mellitus. Matched-pair design multicenter study. Arch. Med. Res. 39, 312–319 (2008).
Ruderman, I. et al. New onset diabetes after kidney transplantation in autosomal dominant polycystic kidney disease: a retrospective cohort study. Nephrology (Carlton) 17, 89–96 (2012).
Sharp, C. K., Zeligman, B. E., Johnson, A. M., Duley, I. & Gabow, P. A. Evaluation of colonic diverticular disease in autosomal dominant polycystic kidney disease without end-stage renal disease. Am. J. Kidney Dis. 34, 863–868 (1999).
Lederman, E. D., McCoy, G., Conti, D. J. & Lee, E. C. Diverticulitis and polycystic kidney disease. Am. Surg. 66, 200–203 (2000).
Sarkio, S., Halme, L., Kyllonen, L. & Salmela, K. Severe gastrointestinal complications after 1,515 adult kidney transplantations. Transpl. Int. 17, 505–510 (2004).
Andreoni, K. A. et al. Increased incidence of gastrointestinal surgical complications in renal transplant recipients with polycystic kidney disease. Transplantation 67, 262–266 (1999).
Bretagnol, A. et al. Autosomal dominant polycystic kidney disease: risk factor for nonmelanoma skin cancer following kidney transplantation. Transpl. Int. 23, 878–886 (2010).
Wetmore, J. B. et al. Polycystic kidney disease and cancer after renal transplantation. J. Am. Soc. Nephrol. http://dx.doi.org/10.1681/ASN.2013101122.
Kasiske, B. L., Snyder, J. J., Gilbertson, D. T. & Wang, C. Cancer after kidney transplantation in the United States. Am. J. Transplant. 4, 905–913 (2004).
Otley, C. C. et al. Skin cancer in organ transplant recipients: effect of pretransplant end-organ disease. J. Am. Acad. Dermatol. 53, 783–790 (2005).
Shillingford, J. M. et al. The mTOR pathway is regulated by polycystin-1, and its inhibition reverses renal cystogenesis in polycystic kidney disease. Proc. Natl. Acad. Sci. USA 103, 5466–5471 (2006).
Qian, Q. et al. Sirolimus reduces polycystic liver volume in ADPKD patients. J. Am. Soc. Nephrol. 19, 631–638 (2008).
Serra, A. L. et al. Sirolimus and kidney growth in autosomal dominant polycystic kidney disease. N. Engl. J. Med. 363, 820–829 (2010).
Walz, G. et al. Everolimus in patients with autosomal dominant polycystic kidney disease. N. Engl. J. Med. 363, 830–840 (2010).
Canaud, G. et al. Therapeutic mTOR inhibition in autosomal dominant polycystic kidney disease: What is the appropriate serum level? Am. J. Transplant. 10, 1701–1706 (2010).
Novalic, Z. et al. Dose-dependent effects of sirolimus on mTOR signaling and polycystic kidney disease. J. Am. Soc. Nephrol. 23, 842–853 (2012).
Chrispijn, M. et al. Everolimus does not further reduce polycystic liver volume when added to long acting octreotide: results from a randomized controlled trial. J. Hepatol. 59, 153–159 (2013).
Torres, V. E. et al. Prospects for mTOR inhibitor use in patients with polycystic kidney disease and hamartomatous diseases. Clin. J. Am. Soc. Nephrol. 5, 1312–1329 (2010).
Spees, E. K., Orlowski, J. P., Fitting, K. M., Temple, D. M. & Reynolds, C. P. Successful use of cadaver kidneys from diabetic donors for transplantation. Transplant. Proc. 22, 378–379 (1990).
Siegal, B. The polycystic kidney donor. Transplantation 54, 1131 (1992).
Canaud, G. et al. Therapeutic mTOR inhibition in autosomal dominant polycystic kidney disease: What is the appropriate serum level? Am. J. Transplant. 10, 1701–1706 (2010).
Howard, R. J. et al. Development of polycystic disease in a kidney 10 years after transplantation. Transplantation 68, 1620 (1999).
Koene, R. A. Should polycystic kidneys ever be used for renal transplantation? Nephrol. Dial. Transplant. 16, 227–229 (2001).
Acknowledgements
The authors thank M. Jadoul and Z. Hassoun for critically reviewing the manuscript before submission, and R. Lhommel for providing images of PET–CT.
Author information
Authors and Affiliations
Contributions
N.K., O.D. and Y.P. researched the data for the article. N.K. and Y.P. wrote the manuscript. All authors contributed to a discussion of the article content and edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Kanaan, N., Devuyst, O. & Pirson, Y. Renal transplantation in autosomal dominant polycystic kidney disease. Nat Rev Nephrol 10, 455–465 (2014). https://doi.org/10.1038/nrneph.2014.104
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrneph.2014.104
This article is cited by
-
Elevated hemoglobin levels in renal transplant recipients with polycystic kidney disease versus other etiologies: exploring mechanisms and implications for outcomes
Journal of Nephrology (2024)
-
Kidney transplant patient with immunoglobulin A nephropathy subsequently diagnosed as concurrent autosomal dominant polycystic kidney disease during 17-year follow-up
CEN Case Reports (2020)
-
Use of mammalian target of rapamycin inhibitors in patient with autosomal dominant polycystic kidney disease: an updated meta-analysis
International Urology and Nephrology (2019)
-
Long-term functional results of aspiration and sclerotherapy with ethanol in patients with autosomal dominant polycystic kidney disease: a non-randomized pilot clinical study
International Urology and Nephrology (2016)
-
ADPKD: clinical issues before and after renal transplantation
Journal of Nephrology (2016)