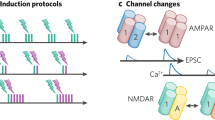
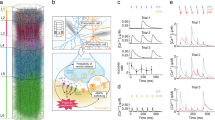
Key Points
-
Activity-dependent plasticity of synaptic strength and membrane excitability are important for learning and memory. It is, however, not well understood how activation of the subcellular signalling cascades necessary for neural plasticity is controlled by ongoing neuronal activity; neither is it known how the plasticity in turn shapes neuronal network dynamics. Several computational approaches used in systems biology are of particular relevance for analysing the molecular mechanisms underlying neuronal plasticity, and for predicting which experiments would be most crucial in improving our understanding.
-
One important methodological approach is to simulate biochemical reactions using ordinary differential equations that describe how molecule concentrations change over time. Several parameters in these models correspond to experimentally measurable values such as reaction rates and affinities. Simulations of signalling networks have shown that properties emerge that are not present in individual pathways, such as the generation of distinct outputs depending on input strength and duration, threshold switches and self-sustaining feedback activation.
-
Stochastic effects need to be taken into account when simulating molecular systems in small volumes such as a dendritic spine or the postsynaptic density. Stochastic models have shown that the conclusions reached from deterministic models might be invalid for small volumes or for systems with few molecules.
-
The spatial morphology of neurons needs to be considered in modelling because, for example, diffusion of some molecules combined with local reactions or anchoring of molecules can create microdomains or concentration gradients of these molecules, which can lead to unexpected behaviour.
-
New techniques in live-cell imaging allow high-resolution data in the spatial and temporal domain to be obtained. These imaging data are important for constraining model parameters during the development of both deterministic and stochastic models.
-
In the future, novel multi-scale approaches will move the field forward. Integration of biochemical signalling cascades within multi-compartmental neuronal models of electrical activity can address how activation of signalling pathways are controlled by neuronal activity, and also how activation of subcellular kinases and phosphatases changes membrane excitability or membrane potential through the control of ion channels and receptors.
Abstract
Synaptic plasticity is thought to underlie learning and memory, but the complexity of the interactions between the ion channels, enzymes and genes that are involved in synaptic plasticity impedes a deep understanding of this phenomenon. Computer modelling has been used to investigate the information processing that is performed by the signalling pathways involved in synaptic plasticity in principal neurons of the hippocampus, striatum and cerebellum. In the past few years, new software developments that combine computational neuroscience techniques with systems biology techniques have allowed large-scale, kinetic models of the molecular mechanisms underlying long-term potentiation and long-term depression. We highlight important advancements produced by these quantitative modelling efforts and introduce promising approaches that use advancements in live-cell imaging.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$189.00 per year
only $15.75 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Coba, M. P. et al. Neurotransmitters drive combinatorial multistate postsynaptic density networks. Sci. Signal. 2, ra19 (2009).
Collins, M. O. et al. Proteomic analysis of in vivo phosphorylated synaptic proteins. J. Biol. Chem. 280, 5972–5982 (2005).
Daoudal, G. & Debanne, D. Long-term plasticity of intrinsic excitability: learning rules and mechanisms. Learn. Mem. 10, 456–465 (2003).
Debanne, D. Associative synaptic plasticity in hippocampus and visual cortex: cellular mechanisms and functional implications. Rev. Neurosci. 7, 29–46 (1996).
Citri, A. & Malenka, R. C. Synaptic plasticity: multiple forms, functions, and mechanisms. Neuropsychopharmacology 33, 18–41 (2008). An extensive and recent overview of synaptic plasticity in the nervous system.
Lynch, G., Larson, J., Kelso, S., Barrionuevo, G. & Schottler, F. Intracellular injections of EGTA block induction of hippocampal long-term potentiation. Nature 305, 719–721 (1983).
Malenka, R. C., Kauer, J. A., Zucker, R. S. & Nicoll, R. A. Postsynaptic calcium is sufficient for potentiation of hippocampal synaptic transmission. Science 242, 81–84 (1988).
Bear, M. F., Cooper, L. N. & Ebner, F. F. A physiological basis for a theory of synapse modification. Science 237, 42–48 (1987).
Lisman, J. A mechanism for the Hebb and the anti-Hebb processes underlying learning and memory. Proc. Natl Acad. Sci. USA 86, 9574–9578 (1989).
Nevian, T. & Sakmann, B. Spine Ca2+ signaling in spike-timing-dependent plasticity. J. Neurosci. 26, 11001–11013 (2006).
Adermark, L. & Lovinger, D. M. Retrograde endocannabinoid signaling at striatal synapses requires a regulated postsynaptic release step. Proc. Natl Acad. Sci. USA 104, 20564–20569 (2007).
Feldman, D. E. Synaptic mechanisms for plasticity in neocortex. Annu. Rev. Neurosci. 32, 33–55 (2009).
Abel, T. et al. Genetic demonstration of a role for PKA in the late phase of LTP and in hippocampus-based long-term memory. Cell 88, 615–626 (1997). In this study, transgenic mice with forebrain-specific, conditionally expressed inhibition of PKA were used to show a correlation between deficits in late-phase LTP and spatial navigation.
Shema, R., Sacktor, T. C. & Dudai, Y. Rapid erasure of long-term memory associations in the cortex by an inhibitor of PKM ζ. Science 317, 951–953 (2007). This study showed that long-term associative memories in the rat neocortex were disrupted following local application of an inhibitor of protein kinase Mζ, suggesting a constitutively active kinase might be necessary for long-term memory.
Yao, Y. et al. PKM ζ maintains late long-term potentiation by N-ethylmaleimide-sensitive factor/GluR2-dependent trafficking of postsynaptic AMPA receptors. J. Neurosci. 28, 7820–7827 (2008).
Kelleher, R. J. III, Govindarajan, A., Jung, H. Y., Kang, H. & Tonegawa, S. Translational control by MAPK signaling in long-term synaptic plasticity and memory. Cell 116, 467–479 (2004).
Sweatt, J. D. The neuronal MAP kinase cascade: a biochemical signal integration system subserving synaptic plasticity and memory. J. Neurochem. 76, 1–10 (2001).
Apergis-Schoute, A. M., Debiec, J., Doyere, V., LeDoux, J. E. & Schafe, G. E. Auditory fear conditioning and long-term potentiation in the lateral amygdala require ERK/MAP kinase signaling in the auditory thalamus: a role for presynaptic plasticity in the fear system. J. Neurosci. 25, 5730–5739 (2005).
Ajay, S. M. & Bhalla, U. S. A role for ERKII in synaptic pattern selectivity on the time-scale of minutes. Eur. J. Neurosci. 20, 2671–2680 (2004).
Shouval, H. Z., Bear, M. F. & Cooper, L. N. A unified model of NMDA receptor-dependent bidirectional synaptic plasticity. Proc. Natl Acad. Sci. USA 99, 10831–10836 (2002).
Song, S., Miller, K. D. & Abbott, L. F. Competitive Hebbian learning through spike-timing-dependent synaptic plasticity. Nature Neurosci. 3, 919–926 (2000).
van Rossum, M. C., Bi, G. Q. & Turrigiano, G. G. Stable Hebbian learning from spike timing-dependent plasticity. J. Neuroscience 20, 8812–8821 (2000).
Morrison, A., Diesmann, M. & Gerstner, W. Phenomenological models of synaptic plasticity based on spike timing. Biol. Cybern. 98, 459–478 (2010).
Holmes, W. R. & Levy, W. B. Insights into associative long-term potentiation from computational models of NMDA receptor-mediated calcium influx and intracellular calcium changes. J. Neurophysiol. 63, 1148–1168 (1990).
Schiegg, A., Gerstner, W., Ritz, R. & Leo van Hemmen, J. Intracellular Ca2+ stores can account for the time course of LTP Induction: a model of Ca2+ dynamics in dendritic spines. J. Neurophysiol. 74, 1046–1055 (1995).
Gamble, E. & Koch, C. The dynamics of free calcium in dendritic spines in response to repetitive synaptic input. Science 236, 1311–1315 (1987).
Zador, A., Koch, C. & Brown, T. H. Biophysical model of a Hebbian synapse. Proc. Natl Acad. Sci. USA 87, 6718–6722 (1990).
Kitano, H. Systems biology: a brief overview. Science 295, 1662–1664 (2002).
Jeong, H., Tombor, B., Albert, R., Oltvai, Z. N. & Barabasi, A. L. The large-scale organization of metabolic networks. Nature 407, 651–654 (2000).
Bullmore, E. & Sporns, O. Complex brain networks: graph theoretical analysis of structural and functional systems. Nature Rev. Neurosci. 10, 186–198 (2009).
Ma'ayan, A. Insights into the organization of biochemical regulatory networks using graph theory analyses. J. Biol. Chem. 284, 5451–5455 (2009).
Lockhart, D. J. & Winzeler, E. A. Genomics, gene expression and DNA arrays. Nature 405, 827–836 (2000).
Tyers, M. & Mann, M. From genomics to proteomics. Nature 422, 193–197 (2003).
Rual, J. F. et al. Towards a proteome-scale map of the human protein–protein interaction network. Nature 437, 1173–1178 (2005).
Wenk, M. R. The emerging field of lipidomics. Nature Rev. Drug Discov. 4, 594–610 (2005).
Watts, D. J. & Strogatz, S. H. Collective dynamics of 'small-world' networks. Nature 393, 440–442 (1998).
Noorbakhsh, F., Overall, C. M. & Power, C. Deciphering complex mechanisms in neurodegenerative diseases: the advent of systems biology. Trends Neurosci. 32, 88–100 (2009).
Bromberg, K. D., Ma'ayan, A., Neves, S. R. & Iyengar, R. Design logic of a cannabinoid receptor signaling network that triggers neurite outgrowth. Science 320, 903–909 (2008).
Taylor, I. W. et al. Dynamic modularity in protein interaction networks predicts breast cancer outcome. Nature Biotech. 27, 199–204 (2009).
Husi, H., Ward, M. A., Choudhary, J. S., Blackstock, W. P. & Grant, S. G. Proteomic analysis of NMDA receptor–adhesion protein signaling complexes. Nature Neurosci. 3, 661–669 (2000).
Ryan, T. J. & Grant, S. G. The origin and evolution of synapses. Nature Rev. Neurosci. 10, 701–712 (2009).
Ferrell, J. E. Jr. Self-perpetuating states in signal transduction: positive feedback, double-negative feedback and bistability. Curr. Opin. Cell Biol. 14, 140–148 (2002). An excellent explanation of the difference between bistability and hysteresis, and discussion of the circuit motifs required to produce bistability. This paper also presents experimental efforts that test modelling predictions and engineer bistable circuits in bacteria.
Wolf, D. M. & Arkin, A. P. Motifs, modules and games in bacteria. Curr. Opin. Microbiol. 6, 125–134 (2003).
Ingram, P. J., Stumpf, M. P. & Stark, J. Network motifs: structure does not determine function. BMC Genomics 7, 108 (2006).
Novak, B. & Tyson, J. J. Design principles of biochemical oscillators. Nature Rev. Mol. Cell Biol. 9, 981–991 (2008).
Sauro, H. M. & Kholodenko, B. N. Quantitative analysis of signaling networks. Prog. Biophys. Mol. Biol. 86, 5–43 (2004).
Megason, S. G. & Fraser, S. E. Imaging in systems biology. Cell 130, 784–795 (2007).
Smolen, P., Baxter, D. A. & Byrne, J. H. Modeling transcriptional control in gene networks — methods, recent results, and future directions. Bull. Math. Biol. 62, 247–292 (2000).
Heinrich, R., Neel, B. G. & Rapoport, T. A. Mathematical models of protein kinase signal transduction. Mol. Cell 9, 957–970 (2002).
Bhalla, U. S. & Iyengar, R. Emergent properties of networks of biological signaling pathways. Science 283, 381–387 (1999). The first modelling paper to take a systems biology approach by simulating interactions between several pathways known to be important for synaptic plasticity.
Kholodenko, B. N. Negative feedback and ultrasensitivity can bring about oscillations in the mitogen-activated protein kinase cascades. Eur. J. Biochem. 267, 1583–1588 (2000).
Markevich, N. I., Hoek, J. B. & Kholodenko, B. N. Signaling switches and bistability arising from multisite phosphorylation in protein kinase cascades. J. Cell Biol. 164, 353–359 (2004).
Kolch, W., Calder, M. & Gilbert, D. When kinases meet mathematics: the systems biology of MAPK signalling. FEBS Lett. 579, 1891–1895 (2005).
Hornberg, J. J. et al. Principles behind the multifarious control of signal transduction. ERK phosphorylation and kinase/phosphatase control. FEBS J. 272, 244–258 (2005).
Gillespie, D. T. Stochastic simulation of chemical kinetics. Annu. Rev. Phys. Chem. 58, 35–55 (2007). A review of the exact stochastic simulation algorithm, as well as recent developments in approximate strategies for simulating such systems.
Stein, M., Gabdoulline, R. R. & Wade, R. C. Bridging from molecular simulation to biochemical networks. Curr. Opin. Struct. Biol. 17, 166–172 (2007).
Smolen, P., Baxter, D. A. & Byrne, J. H. Interlinked dual-time feedback loops can enhance robustness to stochasticity and persistence of memory. Phys. Rev. E. Stat. Nonlin. Soft. Matter Phys. 79, 031902 (2009).
Brandman, O., Ferrell, J. E. Jr, Li, R. & Meyer, T. Interlinked fast and slow positive feedback loops drive reliable cell decisions. Science 310, 496–498 (2005).
Takahashi, K., Arjunan, S. N. & Tomita, M. Space in systems biology of signaling pathways — towards intracellular molecular crowding in silico. FEBS Lett. 579, 1783–1788 (2005). A review of different methods for simulating spatial aspects of neurons.
Coggan, J. S. et al. Evidence for ectopic neurotransmission at a neuronal synapse. Science 309, 446–451 (2005).
Shimizu, T. S. et al. Molecular model of a lattice of signalling proteins involved in bacterial chemotaxis. Nature Cell Biol. 2, 792–796 (2000).
Lipkow, K., Andrews, S. S. & Bray, D. Simulated diffusion of phosphorylated CheY through the cytoplasm of Escherichia coli. J. Bacteriol. 187, 45–53 (2005).
Hattne, J., Fange, D. & Elf, J. Stochastic reaction-diffusion simulation with MesoRD. Bioinformatics. 21, 2923–2924 (2005).
Blackwell, K. T. An efficient stochastic diffusion algorithm for modeling second messengers in dendrites and spines. J. Neurosci. Methods 157, 142–153 (2006).
Zhong, H. et al. A spatial focusing model for G protein signals. Regulator of G protein signaling (RGS) protien-mediated kinetic scaffolding. J. Biol. Chem. 278, 7278–7284 (2003).
Terrin, A. et al. PGE1 stimulation of HEK293 cells generates multiple contiguous domains with different [cAMP]: role of compartmentalized phosphodiesterases. J. Cell Biol. 175, 441–451 (2006).
Oliveira, R. F., Kim, M., Zaccolo, M. & Blackwell, K. T. The role of anchoring proteins and type 4 phosphodiesterases in generating microdomains of cAMP. Society for Neuroscience Annual Meeting (San Diego, California). Abstract 788.3 (2007).
Lindskog, M., Kim, M., Wikstrom, M. A., Blackwell, K. T. & Kotaleski, J. H. Transient calcium and dopamine increase PKA activity and DARPP-32 phosphorylation. PLoS Comput. Biol. 2, e119 (2006).
Kotaleski, J. H., Lester, D. S. & Blackwell, K. T. Subcellular interactions between parallel fibre and climbing fibre signals in Purkinje cells predict sensitivity of classical conditioning to interstimulus interval. Integr. Physiol. Behav. Sci. 37, 265–292 (2002).
Miller, P., Zhabotinsky, A. M., Lisman, J. E. & Wang, X. J. The stability of a stochastic CaMKII switch: dependence on the number of enzyme molecules and protein turnover. PLoS Biol. 3, e107 (2005). An analysis of a model of the CAMK2 switch implicated in long-term memory. Using Monte Carlo simulations, the lifetimes of phosphorylation states of the CAMK2 holoenzyme were analysed.
Hayer, A. & Bhalla, U. S. Molecular switches at the synapse emerge from receptor and kinase traffic. PLoS Comput. Biol. 1, 137–154 (2005).
Ajay, S. M. & Bhalla, U. S. A propagating ERKII switch forms zones of elevated dendritic activation correlated with plasticity. HFSP J. 1, 49–66 (2007). A combined modelling and experimental study aimed at explaining how MAPK1 activation can spread along dendrites of stimulated hippocampal CA1 pyramidal neurons.
Blackwell, K. T. Paired turbulence and light do not produce a supralinear calcium increase in Hermissenda. J. Comput. Neurosci. 17, 81–99 (2004).
Neves, S. R. et al. Cell shape and negative links in regulatory motifs together control spatial information flow in signaling networks. Cell 133, 666–680 (2008).
Kubota, Y. & Bower, J. M. Transient versus asymptotic dynamics of CaM kinase II: possible roles of phosphatase. J. Comput. Neurosci. 11, 263–279 (2001).
Lisman, J. E. & Zhabotinsky, A. M. A model of synaptic memory: a CaMKII/PP1 switch that potentiates transmission by organizing an AMPA receptor anchoring assembly. Neuron 31, 191–201 (2001).
Graupner, M. & Brunel, N. STDP in a bistable synapse model based on CaMKII and associated signaling pathways. PLoS Comput. Biol. 3, e221 (2007).
De Koninck, P. & Schulman, H. Sensitivity of CaM kinase II to the frequency of Ca2+ oscillations. Science 279, 227–230 (1998).
Holmes, W. R. Models of calmodulin trapping and CaM kinase II activation in a dendritic spine. J. Comput. Neurosci. 8, 65–85 (2000).
Dosemeci, A. & Albers, R. W. A mechanism for synaptic frequency detection through autophosphorylation of CaM kinase II. Biophys. J. 70, 2493–2501 (1996).
Ito, M. The molecular organization of cerebellar long-term depression. Nature Rev. Neurosci. 3, 896–902 (2002).
Schreurs, B. G., Oh, M. M. & Alkon, D. L. Pairing-specific long-term depression of Purkinje cell excitatory postsynaptic potentials results from a classical conditioning procedure in rabbit. J. Neurophysiol. 75, 1051–1060 (1996).
Kuroda, S., Schweighofer, N. & Kawato, M. Exploration of signal transduction pathways in cerebellar long-term depression by kinetic simulation. J. Neurosci. 21, 5693–5702 (2001).
Daniel, H., Hemart, N., Jaillard, D. & Crepel, F. Coactivation of metabotropic glutamate receptors and of voltage-gated calcium channels induces long-term depression in cerebellar Purkinje cells in vitro. Exp. Brain Res. 90, 327–331 (1992).
Gormezano, I., Kehoe, E. J. & Marshall, B. J. Twenty years of classical conditioning research with the rabbit. Prog. Psychobiol. Physiol. Psychol. 10, 192–275 (1983).
Nishi, A., Snyder, G. L. & Greengard, P. Bidirectional regulation of DARPP-32 phosphorylation by dopamine. J. Neurosci. 17, 8147–8155 (1997).
Nishi, A. et al. Amplification of dopaminergic signaling by a positive feedback loop. Proc. Natl Acad. Sci. USA 97, 12840–12845 (2000).
Snyder, G. L. et al. Regulation of AMPA receptor dephosphorylation by glutamate receptor agonists. Neuropharmacology 45, 703–713 (2003).
Fernandez, E., Schiappa, R., Girault, J. A. & Le, N. N. DARPP-32 is a robust integrator of dopamine and glutamate signals. PLoS Comput. Biol. 2, e176 (2006).
Nishi, A. et al. Regulation of DARPP-32 dephosphorylation at PKA- and Cdk5-sites by NMDA and AMPA receptors: distinct roles of calcineurin and protein phosphatase-2A. J. Neurochem. 81, 832–841 (2002).
Ahn, J. H. et al. The B”/PR72 subunit mediates Ca2+-dependent dephosphorylation of DARPP-32 by protein phosphatase 2A. Proc. Natl Acad. Sci. USA 104, 9876–9881 (2007).
Bhalla, U. S. Mechanisms for temporal tuning and filtering by postsynaptic signaling pathways. Biophys. J. 83, 740–752 (2002).
Bhalla, U. S. Signaling in small subcellular volumes. II. Stochastic and diffusion effects on synaptic network properties. Biophys. J. 87, 745–753 (2004).
Andrews, S. S. & Bray, D. Stochastic simulation of chemical reactions with spatial resolution and single molecule detail. Phys. Biol. 1, 137–151 (2004).
Takahashi, K., Kaizu, K., Hu, B. & Tomita, M. A multi-algorithm, multi-timescale method for cell simulation. Bioinformatics. 20, 538–546 (2004).
Keller, D. X., Franks, K. M., Bartol, T. M. Jr & Sejnowski, T. J. Calmodulin activation by calcium transients in the postsynaptic density of dendritic spines. PLoS ONE. 3, e2045 (2008). A Monte Carlo model of spine Ca2+ dynamics and successive activation of calmodulin, and the first spatial stochastic model to include signalling molecules downstream of Ca2+.
Ray, S. & Bhalla, U. S. PyMOOSE: interoperable scripting in python for MOOSE. Front. Neuroinformatics 2, 6 (2008).
Aslam, N., Kubota, Y., Wells, D. & Shouval, H. Z. Translational switch for long-term maintenance of synaptic plasticity. Mol. Syst. Biol. 5, 284 (2009).
Hodgkin, A. L. & Huxley, A. F. A quantitative description of membrane current and its application to conduction and excitation in nerve. J. Physiol. 117, 500–544 (1952).
Rall, W. Branching dendritic trees and motoneuron membrane resistivity. Exp. Neurol. 1, 491–527 (1959).
Giepmans, B. N., Adams, S. R., Ellisman, M. H. & Tsien, R. Y. The fluorescent toolbox for assessing protein location and function. Science 312, 217–224 (2006).
Lippincott-Schwartz, J. & Patterson, G. H. Development and use of fluorescent protein markers in living cells. Science 300, 87–91 (2003).
Carlson, H. J. & Campbell, R. E. Genetically encoded FRET-based biosensors for multiparameter fluorescence imaging. Curr. Opin. Biotechnol. 20, 19–27 (2009).
Lee, S. J., Escobedo-Lozoya, Y., Szatmari, E. M. & Yasuda, R. Activation of CaMKII in single dendritic spines during long-term potentiation. Nature 458, 299–304 (2009).
Harvey, C. D., Yasuda, R., Zhong, H. & Svoboda, K. The spread of ras activity triggered by activation of a single dendritic spine. Science 321, 136–140 (2008). This study investigated the compartmentalization and spread of signalling molecules activated by synaptic activity, highlighting a key role for RAF in synaptic plasticity.
Perkel, D. J., Petrozzino, J. J., Nicoll, R. A. & Connor, J. A. The role of Ca2+ entry via synaptically activated NMDA receptors in the induction of long-term potentiation. Neuron 11, 817–823 (1993).
Bear, M. F. & Kirkwood, A. Neocortical long-term potentiation. Curr. Opin. Neurobiol. 3, 197–202 (1993).
Otmakhov, N., Griffith, L. C. & Lisman, J. E. Postsynaptic inhibitors of calcium/calmodulin-dependent protein kinase type II block induction but not maintenance of pairing-induced long-term potentiation. J. Neurosci. 17, 5357–5365 (1997).
Lisman, J., Schulman, H. & Cline, H. The molecular basis of CaMKII function in synaptic and behavioural memory. Nature Rev. Neurosci. 3, 175–190 (2002).
Wong, S. T. et al. Calcium-stimulated adenylyl cyclase activity is critical for hippocampus-dependent long-term memory and late phase LTP. Neuron 23, 787–798 (1999).
Mons, N. & Cooper, D. M. Selective expression of one Ca2+-inhibitable adenylyl cyclase in dopaminergically innervated rat brain regions. Brain Res. Mol. Brain Res. 22, 236–244 (1994).
Svenningsson, P. et al. DARPP-32: an integrator of neurotransmission. Annu. Rev. Pharmacol. Toxicol. 44, 269–296 (2004).
Mulkey, R. M., Endo, S., Shenolikar, S. & Malenka, R. C. Involvement of a calcineurin/inhibitor-1 phosphatase cascade in hippocampal long-term depression. Nature 369, 486–488 (1994).
Acknowledgements
J. H. K. was supported by the Swedish Research Council and the Parkinson's Foundation, and K. T. B. was supported by an HFSP programme grant, the joint NSF-NIH CRCNS programme through NIH grant R01 AA16022 and RO1 AA18060. The authors thank T. Abel and R. Evans for comments on an earlier version of this manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Dynamics
-
Time-dependent changes in the activity or quantity of a variable.
- Kinetics
-
The rates at which reactions or diffusion occur.
- Systems biology
-
An inter-disciplinary field that focuses on the relationships and interactions between components of complex biological systems.
- Current clamp mode
-
Used for measuring membrane potential in response to current injection.
- Markov kinetic model
-
A description of ion channel gating in which the change in channel conformation from one state to another depends only on the present state and not on previous states.
- Cable equation
-
Describes the flow of electrical current along a wire, dendrite or axon.
- Low-pass filter
-
A filter that reduces the amplitude of high-frequency signals while leaving low-frequency signals unaltered.
- Resistive–capacitive models
-
Modelling a small neuronal compartment as a resistive element in parallel with a capacitor, and modelling dendrites as a connected set of these elements.
- Network or graph theory
-
The study of the interconnectedness of related items. The nodes or vertices of a graph are individual items, and two nodes are linked with an edge if they influence each other.
- Power law
-
The relationship between the dependent variable, y, and the independent variable, x, is described by y = x^(−k).
- Bistability
-
The existence of two stable or equilibrium states for the same input. Hysteresis is required for a system to achieve bistability.
- Boolean logic network
-
A network with nodes that take one of two values — for example, activated or non-activated genes. Dynamics of the network are simulated using logical operations coupled with explicit time delay.
- Exact stochastic simulation algorithm
-
An algorithm for the numerical solution of the state of a biochemical reaction system, which therefore produces a realistic actualization of the biochemical reactions.
- Stochastic simulations
-
Simulations that use random-number generators — that is, Monte Carlo methods that are governed by rules of chance.
- Deterministic model
-
A model that calculates quantities using differential equations and algebraic equations.
- Glutamate uncaging
-
The process by which chemically caged glutamate is released by focal light. It is used to study the effects of postsynaptic activation with high temporal and spatial control.
- Partial differential equation
-
An equation that describes how the change in one variable depends on several other variables such as time and space.
- Molecular crowding
-
A phenomenon in which the volume occupied by molecules is so large that molecules cannot diffuse freely in the cytoplasm. Reactions are either impeded or enhanced depending on the proximity of reacting molecules.
- Stochastic resonance
-
A mechanism by which noise added to a system enhances the signal to noise ratio.
Rights and permissions
About this article
Cite this article
Kotaleski, J., Blackwell, K. Modelling the molecular mechanisms of synaptic plasticity using systems biology approaches. Nat Rev Neurosci 11, 239–251 (2010). https://doi.org/10.1038/nrn2807
Issue Date:
DOI: https://doi.org/10.1038/nrn2807
This article is cited by
-
A calcium-based plasticity model for predicting long-term potentiation and depression in the neocortex
Nature Communications (2022)
-
A computational model of dopaminergic modulation of hippocampal Schaffer collateral-CA1 long-term plasticity
Journal of Computational Neuroscience (2022)
-
A Modular Workflow for Model Building, Analysis, and Parameter Estimation in Systems Biology and Neuroscience
Neuroinformatics (2022)
-
Noise-induced collective dynamics in the small-world network of photosensitive neurons
Journal of Biological Physics (2022)
-
Stochastic reaction, stochastic diffusion
ChemTexts (2020)