Key Points
-
Heat shock protein 90 (HSP90) is an essential, abundant and ubiquitous eukaryotic chaperone that plays crucial roles in the folding of its client proteins. Fungal Hsp90 has been shown to stabilize client proteins, buffering or potentiating the phenotypic impact of mutations and thereby acting as an evolutionary capacitor during fungal evolution.
-
In cellular timescales, fungal Hsp90 has been shown to interact with and modulate the activities of client proteins. These clients include key regulators such as protein kinases and transcription factors that control fungal growth, environmental adaptation and pathogenicity.
-
Fungal Hsp90 activity is tightly regulated and is induced in response to heat shock and other proteotoxic stresses. Hsp90 synthesis is controlled by an autoregulatory circuit involving heat shock transcription factor 1 (Hsf1), and Hsp90 binding specificity is modulated by post-transcriptional modification.
-
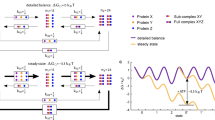
Straightforward mathematical modelling predicts that the degree to which Hsp90 binds specific client proteins depends on Hsp90 availability and the relative affinities of the Hsp90 chaperone for these client proteins. This prediction is consistent with the experimental observation that the fungal Hsp90 interactome displays considerable environmental plasticity.
-
This plasticity implies that environmental challenges promote transient changes in the profile of regulators bound by Hsp90 and, hence, modulate the activities of the corresponding signalling pathways. We propose that Hsp90 acts as a biological transistor that tunes the activity of fungal signalling networks to environmental conditions.
Abstract
Heat shock protein 90 (HSP90) is an essential, abundant and ubiquitous eukaryotic chaperone that has crucial roles in protein folding and modulates the activities of key regulators. The fungal Hsp90 interactome, which includes numerous client proteins such as receptors, protein kinases and transcription factors, displays a surprisingly high degree of plasticity that depends on environmental conditions. Furthermore, although fungal Hsp90 levels increase following environmental challenges, Hsp90 activity is tightly controlled via post-translational regulation and an autoregulatory loop involving heat shock transcription factor 1 (Hsf1). In this Review, we discuss the roles and regulation of fungal Hsp90. We propose that Hsp90 acts as a biological transistor that modulates the activity of fungal signalling networks in response to environmental cues via this Hsf1–Hsp90 autoregulatory loop.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ritossa, F. A new puffing pattern induced by temperature shock and DNP in Drosophila. Cell. Mol. Life Sci. 18, 571–573 (1962).
Lindquist, S. Regulation of protein synthesis during heat shock. Nature 293, 311–314 (1981).
Lindquist, S. The heat-shock response. Annu. Rev. Biochem. 55, 1151–1191 (1986).
Key, J. L., Lin, C. Y. & Chen, Y. M. Heat shock proteins of higher plants. Proc. Natl Acad. Sci. USA 78, 3526–3530 (1981).
Lindquist, S. & Craig, E. A. The heat-shock proteins. Annu. Rev. Genet. 22, 631–677 (1988).
Ang, D., Liberek, K., Skowyra, D., Zylicz, M. & Georgopoulos, C. Biological role and regulation of the universally conserved heat shock proteins. J. Biol. Chem. 266, 24233–24236 (1991).
Young, J. C., Agashe, V. R., Siegers, K. & Hartl, F. U. Pathways of chaperone-mediated protein folding in the cytosol. Nature Rev. Mol. Cell Biol. 5, 781–791 (2004).
Haslbeck, M. et al. Hsp42 is the general small heat shock protein in the cytosol of Saccharomyces cerevisiae. EMBO J. 23, 638–649 (2004).
Nikolaou, E. et al. Phylogenetic diversity of stress signalling pathways in fungi. BMC Evol. Biol. 9, 44 (2009).
Nicholls, S., Leach, M. D., Priest, C. L. & Brown, A. J. Role of the heat shock transcription factor, Hsf1, in a major fungal pathogen that is obligately associated with warm-blooded animals. Mol. Microbiol. 74, 844–861 (2009).
Odds, F. C. (ed.) Candida and Candidosis (Bailliere Tindall, 1988).
Nicholls, S. et al. Activation of the heat shock transcription factor Hsf1 is essential for the full virulence of the fungal pathogen Candida albicans. Fungal Genet. Biol. 48, 297–305 (2011).
Leach, M. D., Tyc, K. M., Brown, A. J. P. & Klipp, E. Modelling the regulation of thermal adaptation in Candida albicans, a major fungal pathogen of humans. PLoS ONE 7, e32467 (2012).
Borkovich, K. A., Farrelly, F. W., Finkelstein, D. B., Taulien, J. & Lindquist, S. Hsp82 is an essential protein that is required in higher concentrations for growth of cells at higher temperatures. Mol. Cell. Biol. 9, 3919–3930 (1989).
Swoboda, R. K. et al. Structure and regulation of the HSP90 gene from the pathogenic fungus Candida albicans. Infect. Immun. 63, 4506–4514 (1995).
Kim, D.-U. et al. Analysis of a genome-wide set of gene deletions in the fission yeast Schizosaccharomyces pombe. Nature Biotech. 28, 617–623 (2010).
Taipale, M., Jarosz, D. F. & Lindquist, S. HSP90 at the hub of protein homeostasis: emerging mechanistic insights. Nature Rev. Mol. Cell Biol. 11, 515–528 (2010). An excellent review of the structural dynamics of Hsp90 and the regulation of this chaperone.
Jarosz, D. F. & Lindquist, S. Hsp90 and environmental stress transform the adaptive value of natural genetic variation. Science 330, 1820–1824 (2010).
Rutherford, S. L. & Lindquist, S. Hsp90 as a capacitor for morphological evolution. Nature 396, 336–342 (1998). The description of Hsp90 as a capacitor of evolutionary change in eukaryotes.
Cowen, L. E. & Lindquist, S. Hsp90 potentiates the rapid evolution of new traits: drug resistance in diverse fungi. Science 309, 2185–2189 (2005). A summary of the role of Hsp90 in the evolution of antifungal drug resistance.
Chen, G., Bradford, W. D., Seidel, C. W. & Li, R. Hsp90 stress potentiates rapid cellular adaptation through induction of aneuploidy. Nature 482, 246–250 (2012).
Wandinger, S. K., Richter, K. & Buchner, J. The Hsp90 chaperone machinery. J. Biol. Chem. 283, 18473–18477 (2008).
Li, J., Soroka, J. & Buchner, J. The Hsp90 chaperone machinery: conformational dynamics and regulation by co-chaperones. Biochim. Biophys. Acta 1823, 624–635 (2012). An outstanding review on the Hsp90 chaperone cycle and Hsp90 co-chaperones.
Pearl, L. H. & Prodromou, C. Structure and in vivo function of Hsp90. Curr. Opin. Struct. Biol. 10, 46–51 (2000).
Ali, M. M. et al. Crystal structure of an Hsp90-nucleotide-p23/Sba1 closed chaperone complex. Nature 440, 1013–1017 (2006). The structural characterization of an Hsp90–co-chaperone complex.
Pearl, L. H., Prodromou, C. & Workman, P. The Hsp90 molecular chaperone: an open and shut case for treatment. Biochem. J. 410, 439–453 (2008).
Southworth, D. R. & Agard, D. A. Species-dependent ensembles of conserved conformational states define the Hsp90 chaperone ATPase cycle. Mol. Cell 32, 631–640 (2008).
Panaretou, B. et al. ATP binding and hydrolysis are essential to the function of the Hsp90 molecular chaperone in vivo. EMBO J. 17, 4829–4836 (1998).
Scheibel, T., Weikl, T. & Buchner, J. Two chaperone sites in Hsp90 differing in substrate specificity and ATP dependence. Proc. Natl Acad. Sci. USA 95, 1495–1499 (1998).
Siligardi, G. et al. Co-chaperone regulation of conformational switching in the Hsp90 ATPase cycle. J. Biol. Chem. 279, 51989–51998 (2004).
Stebbins, C. E. et al. Crystal structure of an Hsp90–geldanamycin complex: targeting of a protein chaperone by an antitumor agent. Cell 89, 239–250 (1997).
Chen, D. et al. Global transcriptional responses of fission yeast to environmental stress. Mol. Biol. Cell 14, 214–229 (2003).
Steen, B. R. et al. Cryptococcus neoformans gene expression during experimental cryptococcal meningitis. Eukaryot. Cell 2, 1336–1349 (2003).
Albrecht, D., Guthke, R., Brakhage, A. A. & Kniemeyer, O. Integrative analysis of the heat shock response in Aspergillus fumigatus. BMC Genomics 11, 32 (2010).
Minchiotti, G., Gargano, S. & Maresca, B. The intron-containing hsp82 gene of the dimorphic pathogenic fungus Histoplasma capsulatum is properly spliced in severe heat shock conditions. Mol. Cell. Biol. 11, 5624–5630 (1991).
Sorger, P. K. Heat shock factor and the heat shock response. Cell 65, 363–366 (1991).
Wu, C. Heat shock transcription factors: structure and regulation. Annu. Rev. Cell Dev. Biol. 11, 441–469 (1995).
Wiederrecht, G., Seto, D. & Parker, C. S. Isolation of the gene encoding the S. cerevisiae heat shock transcription factor. Cell 54, 841–853 (1988).
Sorger, P. K. & Pelham, H. R. B. Yeast heat shock factor is an essential DNA-binding protein that exhibits temperature-dependent phosphorylation. Cell 54, 855–864 (1988).
Sakurai, H. & Ota, A. Regulation of chaperone gene expression by heat shock transcription factor in Saccharomyces cerevisiae: importance in normal cell growth, stress resistance, and longevity. FEBS Lett. 585, 2744–2748 (2011).
Jakobsen, B. K. & Pelham, H. R. Constitutive binding of yeast heat shock factor to DNA in vivo. Mol. Cell. Biol. 8, 5040–5042 (1988).
McDaniel, D. et al. Basal-level expression of the yeast HSP82 gene requires a heat shock regulatory element. Mol. Cell. Biol. 9, 4789–4798 (1989).
Sorger, P. K. & Nelson, H. C. M. Trimerization of a yeast transcriptional activator via a coiled-coil motif. Cell 59, 807–813 (1989).
Gross, D. S., English, K. E., Collins, K. W. & Lee, S. W. Genomic footprinting of the yeast HSP82 promoter reveals marked distortion of the DNA helix and constitutive occupancy of heat shock and TATA elements. J. Mol. Biol. 216, 611–631 (1990).
Sewell, A. K. et al. Mutated yeast heat shock transcription factor exhibits elevated basal transcriptional activation and confers metal resistance. J. Biol. Chem. 270, 25079–25086 (1995).
Liu, X. D. & Thiele, D. J. Oxidative stress induced heat shock factor phosphorylation and HSF-dependent activation of yeast metallothionein gene transcription. Genes Dev. 10, 592–603 (1996).
Nieto-Sotelo, J., Wiederrecht, G., Okuda, A. & Parker, C. S. The yeast heat shock transcription factor contains a transcriptional activation domain whose activity is repressed under nonshock conditions. Cell 62, 807–817 (1990).
Sorger, P. K., Lewis, M. J. & Pelham, H. R. Heat shock factor is regulated differently in yeast and HeLa cells. Nature 329, 81–84 (1987).
Morano, K. A., Santoro, N., Koch, K. A. & Thiele, D. J. A trans-activation domain in yeast heat shock transcription factor is essential for cell cycle progression during stress. Mol. Cell. Biol. 19, 402–411 (1999).
Lindquist, S. Varying patterns of protein synthesis in Drosophila during heat shock: implications for regulation. Dev. Biol. 77, 463–479 (1980).
DiDomenico, B. J., Bugaisky, G. E. & Lindquist, S. The heat shock response is self-regulated at both the transcriptional and posttranscriptional levels. Cell 31, 593–603 (1982).
Abravaya, K., Myers, M. P., Murphy, S. P. & Morimoto, R. I. The human heat shock protein Hsp70 interacts with Hsf1, the transcription factor that regulates heat shock gene expression. Genes Dev. 6, 1153–1164 (1992).
Baler, R., Welch, W. J. & Voellmy, R. Heat shock gene regulation by nascent polypeptides and denatured proteins: Hsp70 as a potential autoregulatory factor. J. Cell Biol. 117, 1151–1159 (1992).
Mosser, D. D., Duchaine, J. & Massie, B. The DNA-binding activity of the human heat shock transcription factor is regulated in vivo by Hsp70. Mol. Cell. Biol. 13, 5427–5438 (1993).
Bonner, J. J. et al. Complex regulation of the yeast heat shock transcription factor. Mol. Biol. Cell 11, 1739–1751 (2000).
Liu, X.-D., Morano, K. A. & Thiele, D. J. The yeast Hsp110 family member, Sse1, is an Hsp90 cochaperone. J. Biol. Chem. 274, 26654–26660 (1999).
Zou, J., Guo, Y., Guettouche, T., Smith, D. F. & Voellmy, R. Repression of heat shock transcription factor HSF1 activation by HSP90 (HSP90 complex) that forms a stress-sensitive complex with HSF1. Cell 94, 471–480 (1998).
Guo, Y. et al. Evidence for a mechanism of repression of heat shock factor 1 transcriptional activity by a multichaperone complex. J. Biol. Chem. 276, 45791–45799 (2001).
Duina, A. A., Kalton, H. M. & Gaber, R. F. Requirement for Hsp90 and a CyP-40-type cyclophilin in negative regulation of the heat shock response. J. Biol. Chem. 273, 18974–18978 (1998).
Gerstel, B., Tuite, M. F. & McCarthy, J. E. G. The effects of 5′-capping, 3′-polyadenylation and leader composition upon the translation and stability of mRNA in a cell-free extract derived from the yeast Saccharomyces cerevisiae. Mol. Microbiol. 6, 2339–2348 (1992).
Mollapour, M. et al. Swe1Wee1-dependent tyrosine phosphorylation of Hsp90 regulates distinct facets of chaperone function. Mol. Cell 37, 333–343 (2010).
Dougherty, J. J., Puri, R. K. & Toft, D. O. Phosphorylation in vivo of chicken oviduct progesterone receptor. J. Biol. Chem. 257, 14226–14230 (1982).
Dougherty, J. J., Rabideau, D. A., Iannotti, A. M., Sullivan, W. P. & Toft, D. O. Identification of the 90 kDa substrate of rat liver type II casein kinase with the heat shock protein which binds steroid receptors. Biochim. Biophys. Acta 927, 74–80 (1987).
Wandinger, S. K., Suhre, M. H., Wegele, H. & Buchner, J. The phosphatase Ppt1 is a dedicated regulator of the molecular chaperone Hsp90. EMBO J. 25, 367–376 (2006).
Mollapour, M. et al. Threonine 22 phosphorylation attenuates Hsp90 interaction with cochaperones and affects Its chaperone activity. Mol. Cell 41, 672–681 (2011).
Scroggins, B. T. & Neckers, L. Post-translational modification of heat-shock protein 90: impact on chaperone function. Expert Opin. Drug Discov. 2, 1403–1414 (2007).
Diezmann, S., Michaut, M., Shapiro, R. S., Bader, G. D. & Cowen, L. E. Mapping the Hsp90 genetic interaction network in Candida albicans reveals environmental contingency and rewired circuitry. PLoS Genet. 8, e1002562 (2012). The first description of the Hsp90 interactome and its plasticity in a fungal pathogen.
Retzlaff, M. et al. Hsp90 is regulated by a switch point in the C-terminal domain. EMBO Rep. 10, 1147–1153 (2009).
Scroggins, B. T. et al. An acetylation site in the middle domain of Hsp90 regulates chaperone function. Mol. Cell 25, 151–159 (2007).
Siligardi, G. et al. Regulation of Hsp90 ATPase activity by the co-chaperone Cdc37p/p50cdc37. J. Biol. Chem. 277, 20151–20159 (2002).
Roe, S. M. et al. The mechanism of Hsp90 regulation by the protein kinase-specific cochaperone p50cdc37. Cell 116, 87–98 (2004).
Panaretou, B. et al. Activation of the ATPase activity of Hsp90 by the stress-regulated cochaperone Aha1. Mol. Cell 10, 1307–1318 (2002).
Zhang, W. et al. Biochemical and structural studies of the interaction of Cdc37 with Hsp90. J. Mol. Biol. 340, 891–907 (2004).
Hernandez, M. P., Sullivan, W. P. & Toft, D. O. The assembly and intermolecular properties of the hsp70-Hop-hsp90 molecular chaperone complex. J. Biol. Chem. 277, 38294–38304 (2002).
Prodromou, C. et al. Regulation of Hsp90 ATPase activity by tetratricopeptide repeat (TPR)-domain co-chaperones. EMBO J. 18, 754–762 (1999).
Lotz, G. P., Lin, H., Harst, A. & Obermann, W. M. J. Aha1 binds to the middle domain of Hsp90, contributes to client protein activation, and stimulates the ATPase activity of the molecular chaperone. J. Biol. Chem. 278, 17228–17235 (2003).
Young, J. C. & Hartl, F. U. Polypeptide release by Hsp90 involves ATP hydrolysis and is enhanced by the co-chaperone p23. EMBO J. 19, 5930–5940 (2000).
Zhao, R. et al. Navigating the chaperone network: an integrative map of physical and genetic interactions mediated by the Hsp90 chaperone. Cell 120, 715–727 (2005). This work maps the physical and genetic interactions of Hsp90 in S. cerevisiae.
Nathan, D. F., Vos, M. H. & Lindquist, S. Identification of SSF1, CNS1, and HCH1 as multicopy suppressors of a Saccharomyces cerevisiae Hsp90 loss-of-function mutation. Proc. Natl Acad. Sci. USA 96, 1409–1414 (1999).
Tesic, M., Marsh, J. A., Cullinan, S. B. & Gaber, R. F. Functional interactions between Hsp90 and the co-chaperones Cns1 and Cpr7 in Saccharomyces cerevisiae. J. Biol. Chem. 278, 32692–32701 (2003).
Catlett, M. G. & Kaplan, K. B. Sgt1p is a unique co-chaperone that acts as a client adaptor to link Hsp90 to Skp1p. J. Biol. Chem. 281, 33739–33748 (2006).
Mayr, C., Richter, K., Lilie, H. & Buchner, J. Cpr6 and Cpr7, two closely related Hsp90-associated immunophilins from Saccharomyces cerevisiae, differ in their functional properties. J. Biol. Chem. 275, 34140–34146 (2000).
Kimura, Y. et al. Cdc37 is a molecular chaperone with specific functions in signal transduction. Genes Dev. 11, 1775–1785 (1997).
Ni, J., Gao, Y., Liu, H. & Chen, J. Candida albicans Cdc37 interacts with the Crk1 kinase and is required for Crk1 production. FEBS Lett. 561, 223–230 (2004).
Bansal, P. K., Abdulle, R. & Kitagawa, K. Sgt1 associates with Hsp90: an initial step of assembly of the core kinetochore complex. Mol. Cell. Biol. 24, 8069–8079 (2004).
Eckert, K. et al. The Pih1-Tah1 cochaperone complex inhibits Hsp90 molecular chaperone ATPase activity. J. Biol. Chem. 285, 31304–31312 (2010).
Johnson, J. & Brown, C. Plasticity of the Hsp90 chaperone machine in divergent eukaryotic organisms. Cell Stress Chaperones 14, 83–94 (2009).
Whitesell, L. & Lindquist, S. L. HSP90 and the chaperoning of cancer. Nature Rev. Cancer 5, 761–772 (2005).
Mahalingam, D. et al. Targeting HSP90 for cancer therapy. Br. J. Cancer 100, 1523–1529 (2009).
Dolgin, E. & Motluk, A. Heat shock and awe. Nature Med. 17, 646–649 (2011).
Millson, S. H. et al. A two-hybrid screen of the yeast proteome for Hsp90 interactors uncovers a novel Hsp90 chaperone requirement in the activity of a stress-activated mitogen-activated protein kinase, Slt2p (Mpk1p). Eukaryot. Cell 4, 849–860 (2005). The finding that the interaction of Hsp90 with a fungal MAPK client is influenced by environmental conditions.
McClellan, A. J. et al. Diverse cellular functions of the Hsp90 molecular chaperone uncovered using systems approaches. Cell 131, 121–135 (2007). This work reveals the plasticity of the S. cerevisiae Hsp90 interactome in response to stress.
Wu, Z., Moghaddas Gholami, A. & Kuster, B. Systematic identification of the HSP90 candidate regulated proteome. Mol. Cell. Proteomics 11, M111.016675 (2012).
Stark, C. et al. The BioGRID Interaction Database: 2011 update. Nucleic Acids Res. 39, D698–D704 (2011).
Echeverría, P. C., Bernthaler, A., Dupuis, P., Mayer, B. & Picard, D. An interaction network predicted from public data as a discovery tool: application to the Hsp90 molecular chaperone machine. PLoS ONE 6, e26044 (2011).
Singh, S. D. et al. Hsp90 governs echinocandin resistance in the pathogenic yeast Candida albicans via calcineurin. PLoS Pathog. 5, e1000532 (2009).
Imai, J. & Yahara, I. Role of HSP90 in salt stress tolerance via stabilization and regulation of calcineurin. Mol. Cell. Biol. 20, 9262–9270 (2000).
Edlind, T., Smith, L., Henry, K., Katiyar, S. & Nickels, J. Antifungal activity in Saccharomyces cerevisiae is modulated by calcium signalling. Mol. Microbiol. 46, 257–268 (2002).
Sanglard, D., Ischer, F., Marchetti, O., Entenza, J. & Bille, J. Calcineurin A of Candida albicans: involvement in antifungal tolerance, cell morphogenesis and virulence. Mol. Microbiol. 48, 959–976 (2003).
Cowen, L. E., Carpenter, A. E., Matangkasombut, O., Fink, G. R. & Lindquist, S. Genetic architecture of Hsp90-dependent drug resistance. Eukaryot. Cell 5, 2184–2188 (2006).
LaFayette, S. L. et al. PKC signaling regulates drug resistance of the fungal pathogen Candida albicans via circuitry comprised of Mkc1, calcineurin, and Hsp90. PLoS Pathog. 6, e1001069 (2010).
Cowen, L. E. et al. Harnessing Hsp90 function as a powerful, broadly effective therapeutic strategy for fungal infectious disease. Proc. Natl Acad. Sci. USA 106, 2818–2823 (2009).
Watanabe, Y., Takaesu, G., Hagiwara, M., Irie, K. & Matsumoto, K. Characterization of a serum response factor-like protein in Saccharomyces cerevisiae, Rlm1, which has transcriptional activity regulated by the Mpk1 (Slt2) mitogen-activated protein kinase pathway. Mol. Cell. Biol. 17, 2615–2623 (1997).
Heinisch, J. J., Lorberg, A., Schmitz, H. P. & Jacoby, J. J. The protein kinase C-mediated MAP kinase pathway involved in the maintenance of cellular integrity in Saccharomyces cerevisiae. Mol. Microbiol. 32, 671–680 (1999).
San Jose, C., Monge, R. A., Perez-Diaz, R., Pla, J. & Nombela, C. The mitogen-activated protein kinase homolog HOG1 gene controls glycerol accumulation in the pathogenic fungus Candida albicans. J. Bacteriol. 178, 5850–5852 (1996).
Toone, W. M. & Jones, N. Stress-activated signalling pathways in yeast. Genes Cells 3, 485–498 (1998).
Hohmann, S. Osmotic stress signaling and osmoadaptation in yeasts. Microbiol. Mol. Biol. Rev. 66, 300–372 (2002).
Smith, D. A., Nicholls, S., Morgan, B. A., Brown, A. J. & Quinn, J. A conserved stress-activated protein kinase regulates a core stress response in the human pathogen Candida albicans. Mol. Biol. Cell 15, 4179–4190 (2004).
Smith, D. A., Morgan, B. A. & Quinn, J. Stress signalling to fungal stress-activated protein kinase pathways. FEMS Microbiol. Lett. 306, 1–8 (2010).
Alonso-Monge, R. et al. Role of the mitogen-activated protein kinase Hog1p in morphogenesis and virulence of Candida albicans. J. Bacteriol. 181, 3058–3068 (1999).
Hawle, P. et al. Cdc37p is required for stress-induced high-osmolarity glycerol and protein kinase C mitogen-activated protein kinase pathway functionality by interaction with Hog1p and Slt2p (Mpk1p). Eukaryot. Cell 6, 521–532 (2007).
Nguyen, A. N. & Shiozaki, K. Heat shock-induced activation of stress MAP kinase is regulated by threonine- and tyrosine-specific phosphatases. Genes Dev. 13, 1653–1663 (1999).
Gerber, M. R., Farrell, A., Deshaies, R. J., Herskowitz, I. & Morgan, D. O. Cdc37 is required for association of the protein kinase Cdc28 with G1 and mitotic cyclins. Proc. Natl Acad. Sci. USA 92, 4651–4655 (1995).
Senn, H., Shapiro, R. S. & Cowen, L. E. Cdc28 provides a molecular link between Hsp90, morphogenesis, and cell cycle progression in Candida albicans. Mol. Biol. Cell 23, 268–283 (2012).
Shapiro, R. S. et al. Hsp90 orchestrates temperature-dependent Candida albicans morphogenesis via Ras1-PKA signaling. Curr. Biol. 19, 621–629 (2009).
Stark, C. et al. BioGRID: a general repository for interaction datasets. Nucleic Acids Res. 34, D535–D539 (2006).
Chen, K. C. et al. Integrative analysis of cell cycle control in budding yeast. Mol. Biol. Cell 15, 3841–3862 (2004).
Klipp, E., Nordlander, B., Kruger, R., Gennemark, P. & Hohmann, S. Integrative model of the response of yeast to osmotic shock. Nature Biotech. 23, 975–982 (2005).
Adrover, M. A. et al. Time-dependent quantitative multicomponent control of the G1-S network by the stress-activated protein kinase Hog1 upon osmostress. Sci. Signal. 4, ra63 (2011).
Mihalik, Á. & Csermely, P. Heat shock partially dissociates the overlapping modules of the yeast protein-protein interaction network: a systems level model of adaptation. PLoS Computat. Biol. 7, e1002187 (2011).
Ebong, I.-o. et al. Heterogeneity and dynamics in the assembly of the Heat Shock Protein 90 chaperone complexes. Proc. Natl Acad. Sci. USA 108, 17939–17944 (2011).
Proctor, C. J. & Lorimer, I. A. J. Modelling the role of the Hsp70/Hsp90 system in the maintenance of protein homeostasis. PLoS ONE 6, e22038 (2011).
Aligue, R., Akhavan-Niak, H. & Russell, P. A role for Hsp90 in cell cycle control: Wee1 tyrosine kinase activity requires interaction with Hsp90. EMBO J. 13, 6099–6106 (1994).
Shapiro, R. S. et al. Pho85, Pcl1, and Hms1 signaling governs Candida albicans morphogenesis induced by high temperature or Hsp90 compromise. Curr. Biol. 22, 461–470 (2012).
Jabra-Rizk, M. A. et al. Candida dubliniensis and Candida albicans display surface variations consistent with observed intergeneric coaggregation. Rev. Iberoam. Micol. 16, 187–193 (1999).
Vecchiarelli, A., Puliti, M., Torosantucci, A., Cassone, A. & Bistoni, F. In vitro production of tumor necrosis factor by murine splenic macrophages stimulated with mannoprotein constituents of Candida albicans cell wall. Cell. Immunol. 134, 65–76 (1991).
Garner, R., Rubanowice, K., Sawyer, R. & Hudson, J. Secretion of TNF-α by alveolar macrophages in response to Candida albicans mannan. J. Leukoc. Biol. 55, 161–168 (1994).
Pietrella, D., Bistoni, G., Corbucci, C., Perito, S. & Vecchiarelli, A. Candida albicans mannoprotein influences the biological function of dendritic cells. Cell. Microbiol. 8, 602–612 (2006).
Jarosz, D. F., Taipale, M. & Lindquist, S. Protein homeostasis and the phenotypic manifestation of genetic diversity: principles and mechanisms. Annu. Rev. Genet. 44, 189–216 (2010).
Queitsch, C., Sangster, T. A. & Lindquist, S. Hsp90 as a capacitor of phenotypic variation. Nature 417, 618–624 (2002).
Sangster, T. A., Lindquist, S. & Queitsch, C. Under cover: causes, effects and implications of Hsp90-mediated genetic capacitance. Bioessays 26, 348–362 (2004).
Sangster, T. A. et al. HSP90-buffered genetic variation is common in Arabidopsis thaliana. Proc. Natl Acad. Sci. USA 105, 2969–2974 (2008).
Sangster, T. A. et al. HSP90 affects the expression of genetic variation and developmental stability in quantitative traits. Proc. Natl Acad. Sci. USA 105, 2963–2968 (2008).
Sollars, V. et al. Evidence for an epigenetic mechanism by which Hsp90 acts as a capacitor for morphological evolution. Nature Genet. 33, 70–74 (2003).
Cowen, L. E. The evolution of fungal drug resistance: modulating the trajectory from genotype to phenotype. Nature Rev. Microbiol. 6, 187–198 (2008).
Xu, Y. & Lindquist, S. Heat-shock protein hsp90 governs the activity of pp60v-src kinase. Proc. Natl Acad. Sci. USA 90, 7074–7078 (1993).
Casanueva, M. O., Burga, A. & Lehner, B. Fitness trade-offs and environmentally induced mutation buffering in isogenic C. elegans. Science 335, 82–85 (2012).
Klipp, E. et al. (eds) Systems Biology: A Textbook (Wiley-VCH, 2009).
Herrgard, M. J. et al. A consensus yeast metabolic network reconstruction obtained from a community approach to systems biology. Nature Biotech. 26, 1155–1160 (2008).
Richter, K., Muschler, P., Hainzl, O., Reinstein, J. & Buchner, J. Sti1 is a non-competitive inhibitor of the Hsp90 ATPase. J. Biol. Chem. 278, 10328–10333 (2003).
Marsh, J. A., Kalton, H. M. & Gaber, R. F. Cns1 is an essential protein associated with the Hsp90 chaperone complex in Saccharomyces cerevisiae that can restore cyclophilin 40-dependent functions in cpr7Δ cells. Mol. Cell. Biol. 18, 7353–7359 (1998).
Acknowledgements
The authors are grateful to numerous colleagues, and to J. Heitman in particular, for insightful discussions. M.D.L. is a Sir Henry Wellcome Postdoctoral Fellow (Wellcome Trust grant 096072). E.K. is supported by grants from the German Research Council (grants GRK 1772, SFB618 and SFB740), the German Ministry of Education and Research (grants 0315786A and 0315584B) and the European Commission (grants FINSysB, PITN-GA-2008-214004 and Unicellsys HEALTH-2007-201142). L.E.C. is supported by a Career Award in the Biomedical Sciences from the Burroughs Wellcome Fund, a Canada Research Chair in Microbial Genomics and Infectious Disease, a Ministry of Research and Innovation (Ontario, Canada) Early Researcher Award, and by grants from the Natural Sciences and Engineering Research Council of Canada (Discovery Grant 355965) and the Canadian Institutes of Health Research (grants MOP-86452 and MOP-119520). A.J.P.B. is supported by grants from the UK Biotechnology and Biological Research Council (grants BB/D009308/1 and BB/F00513X/1), the Wellcome Trust (grants 080088 and 097377) and the European Commission (grants FINSysB, PITN-GA-2008-214004 and STRIFE ERC-2009-AdG-249793).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary informtation S1 (figure)
The fungal Hsp90 chaperone machine and its client proteins. (PDF 204 kb)
Related links
Glossary
- Proteotoxic stresses
-
Cellular stress conditions that prompt the accumulation of unfolded or damaged proteins, or the formation of protein aggregates.
- Heat shock elements
-
Consensus sequences that are present in the promoter regions of heat shock genes and are bound by heat shock transcription factor 1 (Hsf1), thereby activating the expression of these genes.
- Hyperphosphorylation
-
The phosphorylation of a target protein at multiple residues.
- S-nitrosylated
-
Containing a covalently attached nitrosyl group on the thiol moiety of one or more cysteine residues in a protein. These nitrosyl groups are added as a result of nitrosative stress.
- Synthetic genetic phenotypes
-
Phenotypes that are not apparent as a result of a single perturbation alone, but are revealed by combining two mutations or genetic and pharmacological perturbations.
- Chemical genomic screens
-
Screens that combine small-molecule inhibitors or activators with genome-wide mutant collections to identify mutations that confer sensitivity or resistance to these molecules.
- Filamentation
-
The formation of cells with an elongated morphology, such as hyphae, pseudohyphae and yeast cells that have not undergone cell separation.
Rights and permissions
About this article
Cite this article
Leach, M., Klipp, E., Cowen, L. et al. Fungal Hsp90: a biological transistor that tunes cellular outputs to thermal inputs. Nat Rev Microbiol 10, 693–704 (2012). https://doi.org/10.1038/nrmicro2875
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrmicro2875
This article is cited by
-
A host defense peptide mimetic, brilacidin, potentiates caspofungin antifungal activity against human pathogenic fungi
Nature Communications (2023)
-
Structural basis for activation of fungal sterol receptor Upc2 and azole resistance
Nature Chemical Biology (2022)
-
CgSTE11 mediates cross tolerance to multiple environmental stressors in Candida glabrata
Scientific Reports (2019)
-
In Vitro Interaction of Geldanamycin with Triazoles and Echinocandins Against Common and Emerging Candida Species
Mycopathologia (2019)
-
Hsf1 and Hsp90 orchestrate temperature-dependent global transcriptional remodelling and chromatin architecture in Candida albicans
Nature Communications (2016)