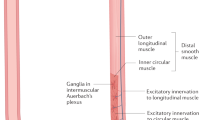
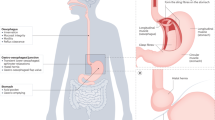
Abstract
Chronic disturbances of gastrointestinal function encompass a wide spectrum of clinical disorders that range from common conditions with mild-to-moderate symptoms to rare diseases characterized by a severe impairment of digestive function, including chronic pain, vomiting, bloating and severe constipation. Patients at the clinically severe end of the spectrum can have profound changes in gut transit and motility. In a subset of these patients, histopathological analyses have revealed abnormalities of the gut innervation, including the enteric nervous system, termed enteric neuropathies. This Review discusses advances in the diagnosis and management of the main clinical entities—achalasia, gastroparesis, intestinal pseudo-obstruction and chronic constipation—that result from enteric neuropathies, including both primary and secondary forms. We focus on the various evident neuropathologies (degenerative and inflammatory) of these disorders and, where possible, present the specific implications of histological diagnosis to contemporary treatment. This knowledge could enable the future development of novel targeted therapeutic approaches.
Key Points
-
A wide spectrum of clinical phenotypes are characterized by degrees of gut sensorimotor dysfunction; at the clinically severe end, patients can have profound changes of gut transit and motility, with or without chronic visceral dilatation
-
When histologically studied (and accepting some methodological limitations), a subset of patients with demonstrable gastrointestinal motor disturbances have evidence of disease to nerves of the enteric nervous system termed enteric neuropathies
-
Several disorders, for example, oesophageal achalasia, gastroparesis, enteric dysmotility, intestinal pseudo-obstruction and chronic constipation, can result from enteric neuropathies, including both primary and secondary forms
-
Inflammatory and degenerative cellular mechanisms can contribute to neural changes in primary and secondary neuropathies; understanding these mechanisms could promote the development of new therapeutic approaches for enteric neuropathies
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Thompson, W. G. et al. Functional bowel disorders and functional abdominal pain. Gut 45 (Suppl. 2), 43–47 (1999).
Mann, S. D., Debinski, H. S. & Kamm, M. A. Clinical characteristics of chronic idiopathic intestinal pseudo-obstruction in adults. Gut 41, 675–681 (1997).
Heneyke, S., Smith, V. V., Spitz, L. & Milla, P. J. Chronic intestinal pseudo-obstruction: treatment and long term follow up of 44 patients. Arch. Dis. Child. 81, 21–27 (1999).
Stanghellini, V. et al. Chronic intestinal pseudo-obstruction: manifestations, natural history and management. Neurogastroenterol. Motil. 19, 440–452 (2007).
De Giorgio, R., Cogliandro, R. F., Barbara, G., Corinaldesi, R. & Stanghellini, V. Chronic intestinal pseudo-obstruction: clinical features, diagnosis, and therapy. Gastroenterol. Clin. North Am. 40, 787–807 (2011).
Furness, J. B. The enteric nervous system and neurogastroenterology. Nat. Rev. Gastroenterol. Hepatol. 9, 286–294 (2012).
Knowles, C. H. et al. Gastrointestinal neuromuscular pathology: guidelines for histological techniques and reporting on behalf of the Gastro 2009 International Working Group. Acta Neuropathol. 118, 271–301 (2009).
Knowles, C. H. et al. The London Classification of gastrointestinal neuromuscular pathology: report on behalf of the Gastro 2009 International Working Group. Gut 59, 882–887 (2010).
Knowles, C. H. & Martin, J. E. Enteric neuromuscular pathology update. Gastroenterol.Clin. North Am. 40, 695–713 (2011).
Chitkara, D. K. & Di Lorenzo, C. From the bench to the 'crib'-side: implications of scientific advances to paediatric neurogastroenterology and motility. Neurogastroenterol. Motil. 18, 251–262 (2006).
Laranjeira, C. & Pachnis, V. Enteric nervous system development: recent progress and future challenges. Auton. Neurosci. 151, 61–69 (2009).
Panza, E. et al. Genetics of human enteric neuropathies. Prog. Neurobiol. 96, 176–189 (2012).
Csendes, A. et al. Histological studies of Auerbach's plexuses of the oesophagus, stomach, jejunum, and colon in patients with achalasia of the oesophagus: correlation with gastric acid secretion, presence of parietal cells and gastric emptying of solids. Gut 33, 150–154 (1992).
Goldblum, J. R., Rice, T. W. & Richter, J. E. Histopathologic features in esophagomyotomy specimens from patients with achalasia. Gastroenterology 111, 648–654 (1996).
Goldblum, J. R., Whyte, R. I., Orringer, M. B. & Appelman, H. D. Achalasia. A morphologic study of 42 resected specimens. Am. J. Surg. Pathol. 18, 327–337 (1994).
Gockel, I., Bohl, J. R., Eckardt, V. F. & Junginger, T. Reduction of interstitial cells of Cajal (ICC) associated with neuronal nitric oxide synthase (n-NOS) in patients with achalasia. Am. J. Gastroenterol. 103, 856–864 (2008).
De Giorgio, R. et al. Esophageal and gastric nitric oxide synthesizing innervation in primary achalasia. Am. J. Gastroenterol. 94, 2357–2362 (1999).
Clark, S. B., Rice, T. W., Tubbs, R. R., Richter, J. E. & Goldblum, J. R. The nature of the myenteric infiltrate in achalasia: an immunohistochemical analysis. Am. J. Surg. Pathol. 24, 1153–1158 (2000).
Di Nardo, G. et al. Review article: molecular, pathological and therapeutic features of human enteric neuropathies. Aliment. Pharmacol. Ther. 28, 25–42 (2008).
Boeckxstaens, G. E. et al. Pneumatic dilation versus laparoscopic Heller's myotomy for idiopathic achalasia. N. Engl. J. Med. 364, 1807–1816 (2011).
Grover, M. et al. Cellular changes in diabetic and idiopathic gastroparesis. Gastroenterology 140, 1575–1585 (2011).
Hirst, G. D. & Edwards, F. R. Role of interstitial cells of Cajal in the control of gastric motility. J. Pharmacol. Sci. 96, 1–10 (2004).
Liu, M. T., Kuan, Y. H., Wang, J., Hen, R. & Gershon, M. D. 5-HT4 receptor-mediated neuroprotection and neurogenesis in the enteric nervous system of adult mice. J. Neurosci. 29, 9683–9699 (2009).
Knowles, C. H. & Aziz, Q. Basic and clinical aspects of gastrointestinal pain. Pain 141, 191–209 (2009).
Rohrmann, C. A. Jr, Ricci, M. T., Krishnamurthy, S. & Schuffler, M. D. Radiologic and histologic differentiation of neuromuscular disorders of the gastrointestinal tract: visceral myopathies, visceral neuropathies, and progressive systemic sclerosis. Am. J. Roentgenol. 143, 933–941 (1984).
Stanghellini, V., Camilleri, M. & Malagelada, J. R. Chronic idiopathic intestinal pseudo-obstruction: clinical and intestinal manometric findings. Gut 28, 5–12 (1987).
Lindberg, G. et al. Full-thickness biopsy findings in chronic intestinal pseudo-obstruction and enteric dysmotility. Gut 58, 1084–1090 (2009).
Knowles, C. H. et al. Deranged smooth muscle α-actin as a biomarker of intestinal pseudo-obstruction: a controlled multinational case series. Gut 53, 1583–1589 (2004).
Cogliandro, R. F. et al. Patient-reported outcomes and gut dysmotility in functional gastrointestinal disorders. Neurogastroenterol. Motil. 23, 1084–1091 (2011).
Wingate, D., Hongo, M., Kellow, J., Lindberg, G. & Smout, A. Disorders of gastrointestinal motility: towards a new classification. J. Gastroenterol. Hepatol. 17 (Suppl.), S1–S14 (2002).
Drossman, D. A. & Dumitrascu, D. L. Rome III: New standard for functional gastrointestinal disorders. J. Gastrointest. Liver Dis. 15, 237–241 (2006).
Tornblom, H., Lindberg, G., Nyberg, B. & Veress, B. Full-thickness biopsy of the jejunum reveals inflammation and enteric neuropathy in irritable bowel syndrome. Gastroenterology 123, 1972–1979 (2002).
Moayyedi, P. et al. The American College of Gastroenterology irritable bowel syndrome monograph: translating systematic review data to clinical practice. Gastroenterology 138, 789–791 (2010).
Tack, J. et al. Functional gastroduodenal disorders. Gastroenterology 130, 1466–1479 (2006).
Stanghellini, V. et al. Natural history of chronic idiopathic intestinal pseudo-obstruction in adults: a single center study. Clin. Gastroenterol. Hepatol. 3, 449–458 (2005).
Knowles, C. H. et al. Quantitation of cellular components of the enteric nervous system in the normal human gastrointestinal tract--report on behalf of the Gastro 2009 International Working Group. Neurogastroenterol. Motil. 23, 115–124 (2011).
Mousa, H., Hyman, P. E., Cocjin, J., Flores, A. F. & Di Lorenzo, C. Long-term outcome of congenital intestinal pseudoobstruction. Dig. Dis. Sci. 47, 2298–2305 (2002).
Darnell, R. B. & Posner, J. B. Paraneoplastic syndromes affecting the nervous system. Semin. Oncol. 33, 270–298 (2006).
De Giorgio, R. & Camilleri, M. Human enteric neuropathies: morphology and molecular pathology. Neurogastroenterol. Motil. 16, 515–531 (2004).
De Giorgio, R. et al. Inflammatory neuropathies of the enteric nervous system. Gastroenterology 126, 1872–1883 (2004).
Thapar, N. Future horizons in the treatment of enteric neuropathies. J. Pediatr. Gastroenterol. Nutr. 45 (Suppl. 2), S110–S114 (2007).
Patel, M. & Yang, S. Advances in reprogramming somatic cells to induced pluripotent stem cells. Stem Cell. Rev. 6, 367–380 (2010).
Drossman, D. A. et al. What determines severity among patients with painful functional bowel disorders? Am. J. Gastroenterol. 95, 974–980 (2000).
Wingate, D. & Grundy, D. Neurogastroenterology and motility: at last, an equal partnership. Neurogastroenterol. Motil. 12, 1 (2000).
Hiatt, R. B. & Katz, L. Mast cells in inflammatory conditions of the gastrointestinal tract. Am. J. Gastroenterol. 37, 541–545 (1962).
Chadwick, V. S. et al. Activation of the mucosal immune system in irritable bowel syndrome. Gastroenterology 122, 1778–1783 (2002).
Barbara, G. et al. Activated mast cells in proximity to colonic nerves correlate with abdominal pain in irritable bowel syndrome. Gastroenterology 126, 693–702 (2004).
Veress, B., Nyberg, B., Tornblom, H. & Lindberg, G. Intestinal lymphocytic epithelioganglionitis: a unique combination of inflammation in bowel dysmotility: a histopathological and immunohistochemical analysis of 28 cases. Histopathology 54, 539–549 (2009).
Selgrad, M. et al. JC virus infects the enteric glia of patients with chronic idiopathic intestinal pseudo-obstruction. Gut 58, 25–32 (2009).
De Giorgio, R. et al. Chronic intestinal pseudo-obstruction related to viral infections. Transplant. Proc. 42, 9–14 (2010).
Zuliani, L., Graus, F., Giometto, B., Bien, C. & Vincent, A. Central nervous system neuronal surface antibody associated syndromes: review and guidelines for recognition. J. Neurol. Neurosurg. Psychiatry 83, 638–645 (2012).
Blackshaw, L. A., Brookes, S. J., Grundy, D. & Schemann, M. Sensory transmission in the gastrointestinal tract. Neurogastroenterol. Motil. 19, 1–19 (2007).
Sandgren, J. E., McPhee, M. S. & Greenberger, N. J. Narcotic bowel syndrome treated with clonidine. Resolution of abdominal pain and intestinal pseudo-obstruction. Ann. Intern. Med. 101, 331–334 (1984).
Grover, C. A., Wiele, E. D. & Close, R. J. Narcotic bowel syndrome. J. Emerg. Med. 43, 992–995 (2012).
Gosselin, R. D., Suter, M. R., Ji, R. R. & Decosterd, I. Glial cells and chronic pain. Neuroscientist 16, 519–531 (2010).
Agostini, S. et al. Evidence of central and peripheral sensitization in a rat model of narcotic bowel-like syndrome. Gastroenterology 139, 553–563 (2010).
Brint, E. K., MacSharry, J., Fanning, A., Shanahan, F. & Quigley, E. M. Differential expression of toll-like receptors in patients with irritable bowel syndrome. Am. J. Gastroenterol. 106, 329–336 (2011).
Hutchinson, M. R. et al. Opioid-induced glial activation: mechanisms of activation and implications for opioid analgesia, dependence, and reward. Scientific World Journal 7, 98–111 (2007).
Holzer, P. Opioid antagonists for prevention and treatment of opioid-induced gastrointestinal effects. Curr. Opin. Anaesthesiol. 23, 616–622 (2010).
Squires, J. E. & Squires, R. H. Jr. Munchausen syndrome by proxy: ongoing clinical challenges. J. Pediatr. Gastroenterol. Nutr. 51, 248–253 (2010).
Preston, D. M., Hawley, P. R., Lennard-Jones, J. E. & Todd, I. P. Results of colectomy for severe idiopathic constipation in women (Arbuthnot Lane's disease). Br. J. Surg. 71, 547–552 (1984).
Hutson, J. M., Chow, C. W. & Borg, J. Intractable constipation with a decrease in substance P-immunoreactive fibres: is it a variant of intestinal neuronal dysplasia? J. Pediatr. Surg. 31, 580–583 (1996).
Knowles, C. H. & Farrugia, G. Gastrointestinal neuromuscular pathology in chronic constipation. Best Pract. Res. Clin. Gastroenterol. 25, 43–57 (2011).
Smith, B., Grace, R. H. & Todd, I. P. Organic constipation in adults. Br. J. Surg. 64, 313–314 (1977).
Krishnamurthy, S., Schuffler, M. D., Rohrmann, C. A. & Pope, C. E. 2nd. Severe idiopathic constipation is associated with a distinctive abnormality of the colonic myenteric plexus. Gastroenterology 88, 26–34 (1985).
Porter, A. J., Wattchow, D. A., Hunter, A. & Costa, M. Abnormalities of nerve fibers in the circular muscle of patients with slow transit constipation. Int. J. Colorectal Dis. 13, 208–216 (1998).
Wattchow, D. et al. Regional variation in the neurochemical coding of the myenteric plexus of the human colon and changes in patients with slow transit constipation. Neurogastroenterol. Motil. 20, 1298–3105 (2008).
Knowles, C. H. et al. Smooth muscle inclusion bodies in slow transit constipation. J. Pathol. 193, 390–397 (2001).
Wedel, T. et al. Oligoneuronal hypoganglionosis in patients with idiopathic slow-transit constipation. Dis. Colon Rectum 45, 54–62 (2002).
Mitolo-Chieppa, D. et al. Cholinergic stimulation and nonadrenergic, noncholinergic relaxation of human colonic circular muscle in idiopathic chronic constipation. Dig. Dis. Sci. 43, 2719–2726 (1998).
Camilleri, M., Kerstens, R., Rykx, A. & Vandeplassche, L. A placebo-controlled trial of prucalopride for severe chronic constipation. N. Engl. J. Med. 358, 2344–2354 (2008).
Scharli, A. F. & Meier-Ruge, W. Localized and disseminated forms of neuronal intestinal dysplasia mimicking Hirschsprung's disease. J. Pediatr. Surg. 16, 164–170 (1981).
Koletzko, S. et al. Rectal biopsy for diagnosis of intestinal neuronal dysplasia in children: a prospective multicentre study on interobserver variation and clinical outcome. Gut 44, 853–861 (1999).
Knowles, C. H., Scott, M. & Lunniss, P. J. Outcome of colectomy for slow transit constipation. Ann. Surg. 230, 627–638 (1999).
Hutson, J. M. Intestinal neuronal dysplasia. Defining a new cause for chronic constipation. Aust. Fam. Physician 25, 1357 (1996).
Van den Berg, M. M. et al. Morphological changes of the enteric nervous system, interstitial cells of cajal, and smooth muscle in children with colonic motility disorders. J. Pediatr. Gastroenterol. Nutr. 48, 22–29 (2009).
Di Lorenzo, C., Solzi, G. F., Flores, A. F., Schwankovsky, L. & Hyman, P. E. Colonic motility after surgery for Hirschsprung's disease. Am. J. Gastroenterol. 95, 1759–1764 (2000).
Pensabene, L., Youssef, N. N., Griffiths, J. M. & Di Lorenzo, C. Colonic manometry in children with defecatory disorders. role in diagnosis and management. Am. J. Gastroenterol. 98, 1052–1057 (2003).
Bassotti, G., Imbimbo, B. P., Betti, C., Dozzini, G. & Morelli, A. Impaired colonic motor response to eating in patients with slow-transit constipation. Am. J. Gastroenterol. 87, 504–508 (1992).
Rao, S. S. Diagnosis and management of fecal incontinence. American College of Gastroenterology Practice Parameters Committee. Am. J. Gastroenterol. 99, 1585–1604 (2004).
Bytzer, P. et al. Prevalence of gastrointestinal symptoms associated with diabetes mellitus: a population-based survey of 15,000 adults. Arch. Intern. Med. 161, 1989–1996 (2001).
Kassander, P. Asymptomatic gastric retention in diabetics (gastroparesis diabeticorum). Ann. Intern. Med. 48, 797–812 (1958).
Harberson, J., Thomas, R. M., Harbison, S. P. & Parkman, H. P. Gastric neuromuscular pathology in gastroparesis: analysis of full-thickness antral biopsies. Dig. Dis. Sci. 55, 359–370 (2010).
Mashimo, H., Kjellin, A. & Goyal, R. K. Gastric stasis in neuronal nitric oxide synthase-deficient knockout mice. Gastroenterology 119, 766–773 (2000).
Plourde, V., Quintero, E., Suto, G., Coimbra, C. & Tache, Y. Delayed gastric emptying induced by inhibitors of nitric oxide synthase in rats. Eur. J. Pharmacol. 256, 125–129 (1994).
Orihata, M. & Sarna, S. K. Inhibition of nitric oxide synthase delays gastric emptying of solid meals. J. Pharmacol. Exp. Ther. 271, 660–670 (1994).
Anvari, M., Paterson, C. A. & Daniel, E. E. Role of nitric oxide mechanisms in control of pyloric motility and transpyloric flow of liquids in conscious dogs. Dig. Dis. Sci. 43, 506–512 (1998).
Takahashi, T., Nakamura, K., Itoh, H., Sima, A. A. & Owyang, C. Impaired expression of nitric oxide synthase in the gastric myenteric plexus of spontaneously diabetic rats. Gastroenterology 113, 1535–1544 (1997).
Wrzos, H. F., Cruz, A., Polavarapu, R., Shearer, D. & Ouyang, A. Nitric oxide synthase (NOS) expression in the myenteric plexus of streptozotocin-diabetic rats. Dig. Dis. Sci. 42, 2106–2110 (1997).
Chandrasekharan, B. & Srinivasan, S. Diabetes and the enteric nervous system. Neurogastroenterol. Motil. 19, 951–960 (2007).
Ordog, T. Interstitial cells of Cajal in diabetic gastroenteropathy. Neurogastroenterol. Motil. 20, 8–18 (2008).
He, C. L. et al. Loss of interstitial cells of cajal and inhibitory innervation in insulin-dependent diabetes. Gastroenterology 121, 427–434 (2001).
Chandrasekharan, B. et al. Colonic motor dysfunction in human diabetes is associated with enteric neuronal loss and increased oxidative stress. Neurogastroenterol. Motil. 23, 131–138 (2011).
Nakahara, M. et al. Deficiency of KIT-positive cells in the colon of patients with diabetes mellitus. J. Gastroenterol. Hepatol. 17, 666–670 (2002).
Bagyanszki, M. & Bodi, N. Diabetes-related alterations in the enteric nervous system and its microenvironment. World J. Diabetes 3, 80–93 (2012).
Pereira, E. C. et al. Biomarkers of oxidative stress and endothelial dysfunction in glucose intolerance and diabetes mellitus. Clin. Biochem. 41, 1454–1460 (2008).
Pereira, R. V. et al. L-glutamine supplementation prevents myenteric neuron loss and has gliatrophic effects in the ileum of diabetic rats. Dig. Dis. Sci. 56, 3507–3516 (2011).
Choi, K. M. et al. Heme oxygenase-1 protects interstitial cells of Cajal from oxidative stress and reverses diabetic gastroparesis. Gastroenterology 135, 2055–2064 (2008).
Negi, G., Kumar, A., Joshi, R. P. & Sharma, S. S. Oxidative stress and Nrf2 in the pathophysiology of diabetic neuropathy: old perspective with a new angle. Biochem. Biophys. Res. Commun. 408, 1–5 (2011).
Lin, Z., Forster, J., Sarosiek, I. & McCallum, R. W. Treatment of diabetic gastroparesis by high-frequency gastric electrical stimulation. Diabetes Care 27, 1071–1076 (2004).
Van der Voort, I. R. et al. Gastric electrical stimulation results in improved metabolic control in diabetic patients suffering from gastroparesis. Exp. Clin. Endocrinol. Diabetes 113, 38–42 (2005).
Abell, T. et al. Gastric electrical stimulation for medically refractory gastroparesis. Gastroenterology 125, 421–428 (2003).
Forno, L. S. Neuropathology of Parkinson's disease. J. Neuropathol. Exp. Neurol. 55, 259–272 (1996).
Wakabayashi, K. & Takahashi, H. Neuropathology of autonomic nervous system in Parkinson's disease. Eur. Neurol. 38 (Suppl. 2), 2–7 (1997).
Beach, T. G. et al. Multi-organ distribution of phosphorylated alpha-synuclein histopathology in subjects with Lewy body disorders. Acta Neuropathol. 119, 689–702 (2010).
Wakabayashi, K., Takahashi, H., Takeda, S., Ohama, E. & Ikuta, F. Parkinson's disease: the presence of Lewy bodies in Auerbach's and Meissner's plexuses. Acta Neuropathol. 76, 217–221 (1988).
Annerino, D. M. et al. Parkinson's disease is not associated with gastrointestinal myenteric ganglion neuron loss. Acta Neuropathol. 124, 665–680 (2012).
Lebouvier, T. et al. Colonic biopsies to assess the neuropathology of Parkinson's disease and its relationship with symptoms. PLoS ONE 14, e12728 (2010).
Salat-Foix, D. & Suchowersky, O. The management of gastrointestinal symptoms in Parkinson's disease. Expert Rev. Neurother. 12, 239–248 (2012).
Goetze, O. et al. Predictors of gastric emptying in Parkinson's disease. Neurogastroenterol. Motil. 18, 369–375 (2006).
Soykan, I., Sarosiek, I. & McCallum, R. W. The effect of chronic oral domperidone therapy on gastrointestinal symptoms, gastric emptying, and quality of life in patients with gastroparesis. Am. J. Gastroenterol. 92, 976–980 (1997).
Djaldetti, R., Koren, M., Ziv, I., Achiron, A. & Melamed, E. Effect of cisapride on response fluctuations in Parkinson's disease. Mov. Disord. 10, 81–84 (1995).
Asai, H. et al. Increased gastric motility during 5-HT4 agonist therapy reduces response fluctuations in Parkinson's disease. Parkinsonism Relat. Disord. 11, 499–502 (2005).
Pfeiffer, R. F. Autonomic dysfunction in Parkinson's disease. Expert Rev. Neurother. 12, 697–706 (2012).
Zangaglia, R. et al. Macrogol for the treatment of constipation in Parkinson's disease. A randomized placebo-controlled study. Mov. Disord. 22, 1239–1244 (2007).
Cassani, E. et al. Use of probiotics for the treatment of constipation in Parkinson's disease patients. Minerva Gastroenterol. Dietol. 57, 117–121 (2011).
Nguyen-tat, M. et al. Severe paraneoplastic gastroparesis associated with anti-Hu antibodies preceding the manifestation of small-cell lung cancer. Z. Gastroenterol. 46, 274–278 (2008).
Lennon, V. A. et al. Enteric neuronal autoantibodies in pseudoobstruction with small-cell lung carcinoma. Gastroenterology 100, 137–142 (1991).
Lucchinetti, C. F., Kimmel, D. W. & Lennon, V. A. Paraneoplastic and oncologic profiles of patients seropositive for type 1 antineuronal nuclear autoantibodies. Neurology 50, 652–657 (1998).
Lee, H. R., Lennon, V. A., Camilleri, M. & Prather, C. M. Paraneoplastic gastrointestinal motor dysfunction: clinical and laboratory characteristics. Am. J. Gastroenterol. 96, 373–379 (2001).
Pardi, D. S. et al. Paraneoplastic dysmotility: loss of interstitial cells of Cajal. Am. J. Gastroenterol. 97, 1828–1833 (2002).
Vernino, S. et al. Autoantibodies to ganglionic acetylcholine receptors in autoimmune autonomic neuropathies. N. Engl. J. Med. 343, 847–855 (2000).
Hubball, A., Martin, J. E., Lang, B., De Giorgio, R. & Knowles, C. H. The role of humoral autoimmunity in gastrointestinal neuromuscular diseases. Prog. Neurobiol. 87, 10–20 (2009).
Sodhi, N. et al. Autonomic function and motility in intestinal pseudoobstruction caused by paraneoplastic syndrome. Dig. Dis. Sci. 34, 1937–1942 (1989).
Gerl, A. et al. Paraneoplastic chronic intestinal pseudoobstruction as a rare complication of bronchial carcinoid. Gut 33, 1000–1003 (1992).
Pande, R. & Leis, A. A. Myasthenia gravis, thymoma, intestinal pseudo-obstruction, and neuronal nicotinic acetylcholine receptor antibody. Muscle Nerve 22, 1600–1602 (1999).
Martin, A. et al. A case of paraneoplastic inflammatory neuropathy of the gastrointestinal tract related to an underlying neuroblastoma: successful management with immunosuppressive therapy. J. Pediatr. Gastroenterol. Nutr. 46, 457–460 (2008).
Schobinger-Clement, S., Gerber, H. A. & Stallmach, T. Autoaggressive inflammation of the myenteric plexus resulting in intestinal pseudoobstruction. Am. J. Surg. Pathol. 23, 602–606 (1999).
Badari, A., Farolino, D., Nasser, E., Mehboob, S. & Crossland, D. A novel approach to paraneoplastic intestinal pseudo-obstruction. Support Care Cancer 20, 425–428 (2012).
Drukker, C. A., Heij, H. A., Wijnaendts, L. C., Verbeke, J. I. & Kaspers, G. J. Paraneoplastic gastro-intestinal anti-Hu syndrome in neuroblastoma. Pediatr. Blood Cancer 52, 396–398 (2009).
Da Silveira, A. B. et al. Glial fibrillary acidic protein and S-100 colocalization in the enteroglial cells in dilated and nondilated portions of colon from chagasic patients. Hum. Pathol. 40, 244–251 (2009).
Jabari, S. et al. Selective survival of calretinin- and vasoactive-intestinal-peptide-containing nerve elements in human chagasic submucosa and mucosa. Cell Tissue Res. 349, 473–481 (2012).
Kierszenbaum, F. Chagas' disease and the autoimmunity hypothesis. Clin. Microbiol. Rev. 12, 210–223 (1999).
Da Silveira, A. B. et al. Enteroglial cells act as antigen-presenting cells in chagasic megacolon. Hum. Pathol. 42, 522–532 (2011).
Higuchi, M. D. et al. Association of an increase in CD8+ T cells with the presence of Trypanosoma cruzi antigens in chronic, human, chagasic myocarditis. Am. J. Trop. Med. Hyg. 56, 485–489 (1997).
Kierszenbaum, F. Where do we stand on the autoimmunity hypothesis of Chagas disease? Trends Parasitol. 21, 513–516 (2005).
Goin, J. C. et al. Functional implications of circulating muscarinic cholinergic receptor autoantibodies in chagasic patients with achalasia. Gastroenterology 117, 798–805 (1999).
Teixeira, A. R., Nitz, N., Guimaro, M. C., Gomes, C. & Santos-Buch, C. A. Chagas disease. Postgrad. Med. J. 82, 788–798 (2006).
Rodriques Coura, J. & de Castro, S. L. A critical review on Chagas disease chemotherapy. Mem. Inst. Oswaldo Cruz. 97, 3–24 (2002).
Crema, E., Ribeiro, L. B., Terra, J. A. Jr & Silva, A. A. Laparoscopic transhiatal subtotal esophagectomy for the treatment of advanced megaesophagus. Ann. Thorac. Surg. 80, 1196–1201 (2005).
Gama, A. H. et al. Volvulus of the sigmoid colon in Brazil: a report of 230 cases. Dis. Colon Rectum 19, 314–320 (1976).
Perlemuter, G. et al. Chronic intestinal pseudo-obstruction in systemic lupus erythematosus. Gut 43, 117–122 (1998).
Ebert, E. C. & Hagspiel, K. D. Gastrointestinal and hepatic manifestations of systemic lupus erythematosus. J. Clin. Gastroenterol. 45, 436–441 (2011).
Ferrante, M. et al. The value of myenteric plexitis to predict early postoperative Crohn's disease recurrence. Gastroenterology 130, 1595–1606 (2006).
Sokol, H. & Beaugerie, L. Inflammatory bowel disease and lymphoproliferative disorders: the dust is starting to settle. Gut 58, 1427–1436 (2009).
Smith, V. V. et al. Acquired intestinal aganglionosis and circulating autoantibodies without neoplasia or other neural involvement. Gastroenterology 112, 1366–1371 (1997).
Hanemann, C. O., Hayward, C. & Hilton, D. A. Neurofibromatosis type 1 with involvement of the enteric nerves. J. Neurol. Neurosurg. Psychiatry 78, 1163–1164 (2007).
Moline, J. & Eng, C. Multiple endocrine neoplasia type 2: an overview. Genet. Med. 13, 755–764 (2011).
Iantorno, G. et al. The enteric nervous system in chagasic and idiopathic megacolon. Am. J. Surg. Pathol. 31, 460–468 (2007).
Schuffler, M. D., Lowe, M. C. & Bill, A. H. Studies of idiopathic intestinal pseudoobstruction. I. Hereditary hollow visceral myopathy: clinical and pathological studies. Gastroenterology 73, 327–338 (1977).
McDonald, G. B., Schuffler, M. D., Kadin, M. E. & Tytgat, G. N. Intestinal pseudoobstruction caused by diffuse lymphoid infiltration of the small intestine. Gastroenterology 89, 882–889 (1985).
De Giorgio, R. et al. Idiopathic myenteric ganglionitis underlying intractable vomiting in a young adult. Eur. J. Gastroenterol. Hepatol. 12, 613–616 (2000).
De Giorgio, R. et al. Clinical and morphofunctional features of idiopathic myenteric ganglionitis underlying severe intestinal motor dysfunction: a study of three cases. Am. J. Gastroenterol. 97, 2454–2459 (2002).
Schappi, M. G., Smith, V. V., Milla, P. J. & Lindley, K. J. Eosinophilic myenteric ganglionitis is associated with functional intestinal obstruction. Gut 52, 752–755 (2003).
Ooms, A. H. et al. Eosinophilic myenteric ganglionitis as a cause of chronic intestinal pseudo-obstruction. Virchows. Arch. 460, 123–127 (2012).
Messing, B. & Joly, F. Guidelines for management of home parenteral support in adult chronic intestinal failure patients. Gastroenterology 130 (Suppl. 1), S43–S51 (2006).
Joly, F., Amiot, A. & Messing, B. Nutritional support in the severely compromised motility patient: when and how? Gastroenterol. Clin. North Am. 40, 845–851 (2011).
Davis, M. P. Drug management of visceral pain: concepts from basic research. Pain Res. Treat. http://dx.doi.org/10.1155/2012/265605.
Emmanuel, A. V., Kamm, M. A., Roy, A. J., Kerstens, R. & Vandeplassche, L. Randomised clinical trial: the efficacy of prucalopride in patients with chronic intestinal pseudo-obstruction--a double-blind, placebo-controlled, cross-over, multiple n = 1 study. Aliment. Pharmacol. Ther. 35, 48–55 (2012).
Andersson, S. et al. Gastric electrical stimulation for intractable vomiting in patients with chronic intestinal pseudoobstruction. Neurogastroenterol. Motil. 18, 823–830 (2006).
Abu-Elmagd, K. M. et al. Long-term survival, nutritional autonomy, and quality of life after intestinal and multivisceral transplantation. Ann. Surg. 256, 494–508 (2012).
Acknowledgements
The authors of this Review are supported by grants PRIN and COFIN from the Italian Ministry of University, Research and Education 2008 and 2010 and Ricerca Fondamentale Orientata funds from the University of Bologna to R. De Giorgio. He is also the recipient of research grants from Fondazione Del Monte di Bologna e Ravenna. C. Knowles is funded by the Higher Education Funding Committee of England (HEFCE).
Author information
Authors and Affiliations
Contributions
All authors contributed equally to all aspects of this article.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Knowles, C., Lindberg, G., Panza, E. et al. New perspectives in the diagnosis and management of enteric neuropathies. Nat Rev Gastroenterol Hepatol 10, 206–218 (2013). https://doi.org/10.1038/nrgastro.2013.18
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2013.18
This article is cited by
-
Regenerative medicine: current research and perspective in pediatric surgery
Pediatric Surgery International (2023)
-
Dynamic integration of enteric neural stem cells in ex vivo organotypic colon cultures
Scientific Reports (2021)
-
The association of enteric neuropathy with gut phenotypes in acute and progressive models of Parkinson’s disease
Scientific Reports (2021)
-
New cine magnetic resonance imaging parameters for the differential diagnosis of chronic intestinal pseudo-obstruction
Scientific Reports (2021)
-
Disorders of the enteric nervous system — a holistic view
Nature Reviews Gastroenterology & Hepatology (2021)