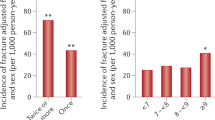
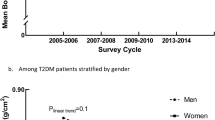
Abstract
Diabetes mellitus adversely affects the skeleton and is associated with an increased risk of osteoporosis and fragility fractures. The mechanisms underlying low bone strength are not fully understood but could include impaired accrual of peak bone mass and diabetic complications, such as nephropathy. Type 1 diabetes mellitus (T1DM) affects the skeleton more severely than type 2 diabetes mellitus (T2DM), probably because of the lack of the bone anabolic actions of insulin and other pancreatic hormones. Bone mass can remain high in patients with T2DM, but it does not protect against fractures, as bone quality is impaired. The class of oral antidiabetic drugs known as glitazones can promote bone loss and osteoporotic fractures in postmenopausal women and, therefore, should be avoided if osteoporosis is diagnosed. A physically active, healthy lifestyle and prevention of diabetic complications, along with calcium and vitamin D repletion, represent the mainstay of therapy for osteoporosis in patients with T1DM or T2DM. Assessment of BMD and other risk factors as part of the diagnostic procedure can help design tailored treatment plans. All osteoporosis drugs seem to be effective in patients with diabetes mellitus. Increased awareness of osteoporosis is needed in view of the growing and aging population of patients with diabetes mellitus.
Key Points
-
Type 1 and type 2 diabetes mellitus are associated with an increased risk of osteoporotic fractures
-
Bone formation and osteoblast function are impaired in patients with type 1 diabetes mellitus
-
BMD is increased but bone quality is reduced in patients with type 2 diabetes mellitus
-
Glitazones might promote bone loss and should be avoided in patients with osteoporosis
-
Comorbidities guide the selection of specific osteoporosis therapies
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
O'Loughlin, A., McIntosh, C., Dinneen, S. F. & O'Brien, T. Review paper: basic concepts to novel therapies: a review of the diabetic foot. Int. J. Low Extrem. Wounds 9, 90–102 (2010).
Carnevale, V., Romagnoli, E. & D'Erasmo, E. Skeletal involvement in patients with diabetes mellitus. Diabetes Metab. Res. Rev. 20, 196–204 (2004).
Schwartz, A. V. Diabetes mellitus: does it affect bone? Calcif. Tissue Int. 73, 515–519 (2003).
Sumpio, B. E. Foot ulcers. N. Engl. J. Med. 343, 787–793 (2000).
Vestergaard, P., Rejnmark, L. & Mosekilde, L. Diabetes and its complications and their relationship with risk of fractures in type 1 and 2 diabetes. Calcif. Tissue Int. 84, 45–55 (2009).
Ahmed, L. A., Joakimsen, R. M., Berntsen, G. K., Fønnebø, V. & Schirmer, H. Diabetes mellitus and the risk of non-vertebral fractures: the Tromso study. Osteoporos. Int. 17, 495–500 (2006).
Janghorbani, M., Van Dam, R. M., Willett, W. C. & Hu, F. B. Systematic review of type 1 and type 2 diabetes mellitus and risk of fracture. Am. J. Epidemiol. 166, 495–505 (2007).
Hofbauer, L. C., Brueck, C. C., Singh, S. K. & Dobnig, H. Osteoporosis in patients with diabetes mellitus. J. Bone Miner. Res. 22, 1317–1328 (2007).
Schwartz, A. V. & Sellmeyer, D. E. Women, type 2 diabetes, and fracture risk. Curr. Diab. Rep. 4, 364–369 (2004).
Strotmeyer, E. S. & Cauley, J. A. Diabetes mellitus, bone mineral density, and fracture risk. Curr. Opin. Endocrinol. Diabetes Obes. 14, 429–435 (2007).
Nagasaka, S., Murakami, T., Uchikawa, T., Ishikawa, S. E. & Saito, T. Effect of glycemic control on calcium and phosphorus handling and parathyroid hormone level in patients with non-insulin-dependent diabetes mellitus. Endocr. J. 42, 377–383 (1995).
Volpato, S., Leville, S. G., Blaum, C., Fried, L. P. & Guralnik, J. M. Risk factors for falls in older disabled women with diabetes: the women's health and aging study. J. Gerontol. A Biol. Sci. Med. Sci. 60, 1539–1545 (2005).
Holmberg, A. H. et al. Risk factors for fragility fracture in middle age. A prospective population-based study of 33,000 men and women. Osteoporos. Int. 17, 1065–1077 (2006).
Schwartz, A. V. et al. Association of BMD and FRAX score with risk of fracture in older adults with type 2 diabetes. JAMA 305, 2184–2192 (2011).
Ensrud, K. E. & Schousboe, J. T. Clinical practice. Vertebral fractures. N. Engl. J. Med. 364, 1634–1642 (2011).
Tinetti, M. E. Clinical Practice. Preventing falls in elderly persons. N. Engl. J. Med. 348, 42–49 (2003).
Bonds, D. E. et al. Risk of fracture in women with type 2 diabetes: the Women's Health Initiative Observational Study. J. Clin. Endocrinol. Metab. 91, 3404–3410 (2006).
de Liefde, I. I. et al. Bone mineral density and fracture risk in type-2 diabetes mellitus: the Rotterdam Study. Osteoporos. Int. 16, 1713–1720 (2005).
Strotmeyer, E. S. et al. Nontraumatic fracture risk with diabetes mellitus and impaired fasting glucose in older white and black adults: the health, aging and body composition study. Arch. Intern. Med. 165, 1612–1617 (2005).
Wallace, C. et al. Incidence of falls, risk factors for falls, and fall-related fractures in individuals with diabetes and prior foot ulcer. Diabetes Care 25, 1983–1986 (2002).
Chaudhary, S. B. et al. Complications of ankle fracture in patients with diabetes. J. Am. Acad. Orthop. Surg. 16, 159–170 (2008).
Wukich, D. K. & Kline, A. J. The management of ankle fractures in patients with diabetes. J. Bone Joint Surg. Am. 90, 1570–1578 (2008).
Norris, R. & Parker, M. Diabetes mellitus and hip fracture: A study of 5966 cases. Injury 42, 1313–1316 (2011).
Retzepi, M. & Donos, N. The effect of diabetes mellitus on osseous healing. Clin. Oral Implants Res. 21, 673–681 (2010).
Weber, G. et al. Bone mass in young patients with type I diabetes. Bone Miner. 8, 23–30 (1990).
Hui, S. L., Epstein, S. & Johnston, C. C. Jr. A prospective study of bone mass in patients with type I diabetes. J. Clin. Endocrinol. Metab. 60, 74–80 (1985).
Forst, T. et al. Peripheral osteopenia in adult patients with insulin-dependent diabetes mellitus. Diabet. Med. 12, 874–879 (1995).
Kayath, M. J., Dib, S. A. & Vieira, J. G. Prevalence and magnitude of osteopenia associated with insulin-dependent diabetes mellitus. J. Diabetes Complications 8, 97–104 (1994).
Compston, J. E., Smith, E. M., Matthews, C. & Schofield, P. Whole body composition and regional bone mass in women with insulin-dependent diabetes mellitus. Clin. Endocrinol. (Oxf.) 41, 289–293 (1994).
Mastrandrea, L. D. et al. Young women with type 1 diabetes have lower bone mineral density that persists over time. Diabetes Care 31, 1729–1735 (2008).
Barrett-Connor, E. & Holbrook, T. L. Sex differences in osteoporosis in older adults with non-insulin-dependent diabetes mellitus. JAMA 268, 3333–3337 (1992).
van Daele, P. L. et al. Bone density in non-insulin-dependent diabetes mellitus. The Rotterdam Study. Ann. Intern. Med. 122, 409–414 (1995).
Strotmeyer, E. S. et al. Diabetes is associated independently of body composition with BMD and bone volume in older white and black men and women: The Health, Aging, and Body Composition Study. J. Bone Miner. Res. 19, 1084–1091 (2004).
Melton, L. J. 3rd et al. A bone structural basis for fracture risk in diabetes. J. Clin. Endocrinol. Metab. 93, 4804–4809 (2008).
Vestergaard, P. Discrepancies in bone mineral density and fracture risk in patients with type 1 and type 2 diabetes—a meta-analysis. Osteoporos. Int. 18, 427–444 (2007).
Merlotti, D., Gennari, L., Dotta, F., Lauro, D. & Nuti, R. Mechanisms of impaired bone strength in type 1 and 2 diabetes. Nutr. Metab. Cardiovasc. Dis. 20, 683–690 (2010).
Vestergaard, P., Rejnmark, L. & Mosekilde, L. Relative fracture risk in patients with diabetes mellitus, and the impact of insulin and oral antidiabetic medication on relative fracture risk. Diabetologia 48, 1292–1299 (2005).
Nicodemus, K. K. & Folsom, A. R. Type 1 and type 2 diabetes and incident hip fractures in postmenopausal women. Diabetes Care 24, 1192–1197 (2001).
Melton, L. J. 3rd, Leibson, C. L., Achenbach, S. J., Therneau, T. M. & Khosla, S. Fracture risk in type 2 diabetes: update of a population-based study. J. Bone Miner. Res. 23, 1334–1342 (2008).
Loder, R. T. The influence of diabetes mellitus on the healing of closed fractures. Clin. Orthop. Relat. Res. 232, 210–216 (1988).
Stuart, M. J. & Morrey, B. F. Arthrodesis of the diabetic neuropathic ankle joint. Clin. Orthop. Relat. Res. 253, 209–211 (1990).
Papa, J., Myerson, M. & Girard, P. Salvage, with arthrodesis, in intractable diabetic neuropathic arthropathy of the foot and ankle. J. Bone Joint Surg. Am. 75, 1056–1066 (1993).
Tisdel, C. L., Marcus, R. E. & Heiple, K. G. Triple arthrodesis for diabetic peritalar neuroarthropathy. Foot Ankle Int. 16, 332–338 (1995).
Johal, K. S., Boulton, C. & Moran, C. G. Hip fractures after falls in hospital: a retrospective observational cohort study. Injury 40, 201–204 (2009).
Barrett-Connor, E., Weiss, T. W., McHorney, C. A., Miller, P. D. & Siris, E. S. Predictors of falls among postmenopausal women: results from the National Osteoporosis Risk Assessment (NORA). Osteoporos. Int. 20, 715–722 (2009).
Christensen, K., Doblhammer, G., Rau, R. & Vaupel, J. W. Ageing populations: the challenges ahead. Lancet 374, 1196–1208 (2009).
Mayne, D., Stout, N. R. & Aspray, T. J. Diabetes, falls and fractures. Age Ageing 39, 522–525 (2010).
Thacher, T. D. & Clarke, B. L. Vitamin D insufficiency. Mayo Clin. Proc. 86, 50–60 (2011).
Dobnig, H. et al. Type 2 diabetes mellitus in nursing home patients: effects on bone turnover, bone mass, and fracture risk. J. Clin. Endocrinol. Metab. 91, 3355–3363 (2006).
Kannus, P., Sievänen, H., Palvanen, M., Järvinen, T. & Parkkari, J. Prevention of falls and consequent injuries in elderly people. Lancet 366, 1885–1893 (2005).
Vischer, U. M. et al. A call to incorporate the prevention and treatment of geriatric disorders in the management of diabetes in the elderly. Diabetes Metab. 35, 168–177 (2009).
Rachner, T. D., Khosla, S. & Hofbauer, L. C. Osteoporosis: now and the future. Lancet 377, 1276–1287 (2011).
Inaba, M. et al. Influence of high glucose on 1,25-dihydroxyvitamin D3-induced effect on human osteoblast-like MG-63 cells. J. Bone Miner. Res. 10, 1050–1056 (1995).
Gopalakrishnan, V., Vignesh, R. C., Arunakaran, J., Aruldhas, M. M. & Srinivasan, N. Effects of glucose and its modulation by insulin and estradiol on BMSC differentiation into osteoblastic lineages. Biochem. Cell Biol. 84, 93–101 (2006).
Botolin, S. et al. Increased bone adiposity and peroxisomal proliferator-activated receptor-gamma2 expression in type I diabetic mice. Endocrinology 146, 3622–3631 (2005).
Lu, H., Kraut, D., Gerstenfeld, L. C. & Graves, D. T. Diabetes interferes with the bone formation by affecting the expression of transcription factors that regulate osteoblast differentiation. Endocrinology 144, 346–352 (2003).
Gunczler, P. et al. Decreased bone mineral density and bone formation markers shortly after diagnosis of clinical type 1 diabetes mellitus. J. Pediatr. Endocrinol. Metab. 14, 525–528 (2001).
Gunczler, P. et al. Decreased lumbar spine bone mass and low bone turnover in children and adolescents with insulin dependent diabetes mellitus followed longitudinally. J. Pediatr. Endocrinol. Metab. 11, 413–419 (1998).
Massé, P. G. et al. Bone metabolic abnormalities associated with well-controlled type 1 diabetes (IDDM) in young adult women: a disease complication often ignored or neglected. J. Am. Coll. Nutr. 29, 419–429 (2010).
Liu, Z. et al. A novel rat model for the study of deficits in bone formation in type-2 diabetes. Acta Orthop. 78, 46–55 (2007).
Fujii, H., Hamada, Y. & Fukagawa, M. Bone formation in spontaneously diabetic Torii-newly established model of non-obese type 2 diabetes rats. Bone 42, 372–379 (2008).
Jang, W. G. et al. Metformin induces osteoblast differentiation via orphan nuclear receptor SHP-mediated transactivation of Runx2. Bone 48, 885–893 (2011).
Kawai, M., Sousa, K. M., MacDougald, O. A. & Rosen, C. J. The many facets of PPARgamma: novel insights for the skeleton. Am. J. Physiol. Endocrinol. Metab. 299, E3–E9 (2010).
Nakashima, T. et al. Evidence for osteocyteregulation of bone homeostasis through RANKL expression. Nat. Med. 17, 1231–1234 (2011).
Xiong, J., Onal, M., Jilka, R. L., Weinstein, R. S., Manolagas, S. C. & O'Brien, C. A. Matrix-embedded cells control osteoclast formation. Nat. Med. 17, 1235–1241 (2011).
Wittrant, Y. et al. High D(+)glucose concentration inhibits RANKL-induced osteoclastogenesis. Bone 42, 1122–1130 (2008).
Kim, J. M. et al. Osteoclast precursors display dynamic metabolic shifts toward accelerated glucose metabolism at an early stage of RANKL-stimulated osteoclast differentiation. Cell. Physiol. Biochem. 20, 935–946 (2007).
Williams, J. P. et al. Regulation of osteoclastic bone resorption by glucose. Biochem. Biophys. Res. Commun. 235, 646–651 (1997).
Dienelt, A. & zur Nieden, N. I. Hyperglycemia impairs skeletogenesis from embryonic stem cells by affecting osteoblast and osteoclast differentiation. Stem Cells Dev. 20, 465–474 (2011).
Kasahara, T. et al. Malfunction of bone marrow-derived osteoclasts and the delay of bone fracture healing in diabetic mice. Bone 47, 617–625 (2010).
Thrailkill, K. M., Lumpkin, C. K. Jr, Bunn, R. C., Kemp, S. F. & Fowlkes, J. L. Is insulin an anabolic agent in bone? Dissecting the diabetic bone for clues. Am. J. Physiol. Endocrinol. Metab. 289, E735–E745 (2005).
Campos Pastor, M. M., López-Ibarra, P. J., Escobar-Jiménez, F., Serrano Pardo, M. D. & García-Cervigón, A. G. Intensive insulin therapy and bone mineral density in type 1 diabetes mellitus: a prospective study. Osteoporos. Int. 11, 455–459 (2000).
Naot, D. & Cornish, J. The role of peptides and receptors of the calcitonin family in the regulation of bone metabolism. Bone 43, 813–818 (2008).
Cornish, J. et al. Preptin, another peptide product of the pancreatic beta-cell, is osteogenic in vitro and in vivo. Am. J. Physiol. Endocrinol. Metab. 292, E117–E122 (2007).
Horcajada-Molteni, M. N. et al. Amylin and bone metabolism in streptozotocin-induced diabetic rats. J. Bone Miner. Res. 16, 958–965 (2001).
Dacquin, R. et al. Amylin inhibits bone resorption while the calcitonin receptor controls bone formation in vivo. J. Cell Biol. 164, 509–514 (2004).
Cornish, J. et al. Shared pathways of osteoblast mitogenesis induced by amylin, adrenomedullin, and IGF-1. Biochem. Biophys. Res. Commun. 318, 240–246 (2004).
Clemens, T. L. & Karsenty, G. The osteoblast: an insulin target cell controlling glucose homeostasis. J. Bone Miner. Res. 26, 677–680 (2011).
Ferron, M. et al. Insulin signaling in osteoblasts integrates bone remodeling and energy metabolism. Cell 142, 296–308 (2010).
Lee, N. K. et al. Endocrine regulation of energy metabolism by the skeleton. Cell 130, 456–469 (2007).
Oury, F. et al. Endocrine regulation of male fertility by the skeleton. Cell 144, 796–809 (2011).
Hein, G. E. Glycation endproducts in osteoporosis—is there a pathophysiologic importance? Clin. Chim. Acta 371, 32–36 (2006).
Saito, M., Fujii, K., Mori, Y. & Marumo, K. Role of collagen enzymatic and glycation induced cross-links as a determinant of bone quality in spontaneously diabetic WBN/Kob rats. Osteoporos. Int. 17, 1514–1523 (2006).
Yamamoto, M., Yamaguchi, T., Yamauchi, M. & Sugimoto, T. Low serum level of the endogenous secretory receptor for advanced glycation end products (esRAGE) is a risk factor for prevalent vertebral fractures independent of bone mineral density in patients with type 2 diabetes. Diabetes Care 32, 2263–2268 (2009).
Schwartz, A. V. et al. Pentosidine and increased fracture risk in older adults with type 2 diabetes. J. Clin. Endocrinol. Metab. 94, 2380–2386 (2009).
Bouxsein, M. L. Bone structure and fracture risk: Do they go arm in arm? J. Bone Miner. Res. 26, 1389–1391 (2011).
Seeman, E. & Delmas, P. D. Bone quality—the material and structural basis of bone strength and fragility. N. Engl. J. Med. 354, 2250–2261 (2006).
Kanis, J. A. et al. Development and use of FRAX in osteoporosis. Osteoporos. Int. 21 (Suppl. 2), S407–S413 (2010).
Raisz, L. G. Clinical practice. Screening for osteoporosis. N. Engl. J. Med. 353, 164–171 (2005).
Ebeling, P. R. Clinical practice. Osteoporosis in men. N. Engl. J. Med. 358, 1474–1482 (2008).
[No authors listed] The effect of intensive treatment of diabetes on the development and progression of long-term complications in insulin-dependent diabetes mellitus. The Diabetes Control and Complications Trial Research Group. N. Engl. J. Med. 329, 977–986 (1993).
Nathan, D. M. et al. Intensive diabetes treatment and cardiovascular disease in patients with type 1 diabetes. N. Engl. J. Med. 353, 2643–2653 (2005).
Egger, M., Davey Smith, G., Stettler, C. & Diem, P. Risk of adverse effects of intensified treatment in insulin-dependent diabetes mellitus: a meta-analysis. Diabet. Med. 14, 919–928 (1997).
[No authors listed] Lifetime benefits and costs of intensive therapy as practiced in the diabetes control and complications trial. The Diabetes Control and Complications Trial Research Group. JAMA 276, 1409–1415 (1996).
Hypoglycemia in the Diabetes Control and Complications Trial. The Diabetes Control and Complications Trial Research Group. Diabetes 46, 271–286 (1997).
Schwartz, A. V. et al. Thiazolidinedione use and bone loss in older diabetic adults. J. Clin. Endocrinol. Metab. 91, 3349–3354 (2006).
Grey, A. et al. The peroxisome proliferator-activated receptor-gamma agonist rosiglitazone decreases bone formation and bone mineral density in healthy postmenopausal women: a randomized, controlled trial. J. Clin. Endocrinol. Metab. 92, 1305–1310 (2007).
Yaturu, S., Bryant, B. & Jain, S. K. Thiazolidinedione treatment decreases bone mineral density in type 2 diabetic men. Diabetes Care 30, 1574–1576 (2007).
Meier, C. et al. Use of thiazolidinediones and fracture risk. Arch. Intern. Med. 168, 820–825 (2008).
Habib, Z. A. et al. Thiazolidinedione use and the longitudinal risk of fractures in patients with type 2 diabetes mellitus. J. Clin. Endocrinol. Metab. 95, 592–600 (2010).
Bilik, D. et al. Thiazolidinediones and fractures: evidence from translating research into action for diabetes. J. Clin. Endocrinol. Metab. 95, 4560–4565 (2010).
[No authors listed] Standards of medical care in diabetes--2011. Diabetes Care 34 (Suppl. 1), S11–S61 (2011).
Bao, X. H., Wong, V., Wang, Q. & Low, L. C. Prevalence of peripheral neuropathy with insulin-dependent diabetes mellitus. Pediatr. Neurol. 20, 204–209 (1999).
Bolland, M. J. et al. Vascular events in healthy older women receiving calcium supplementation: randomised controlled trial. BMJ 336, 262–266 (2008).
Bolland, M. J., Grey, A., Avenell, A., Gamble, G. D. & Reid, I. R. Calcium supplements with or without vitamin D and risk of cardiovascular events: reanalysis of the Women's Health Initiative limited access dataset and meta-analysis. BMJ 342, d2040 (2011).
Maxmen, A. Nutrition advice: The vitamin D-lemma. Nature 475, 23–25 (2011).
Holick, M. F. Vitamin D deficiency. N. Engl. J. Med. 357, 266–281 (2007).
Priemel, M. et al. Bone mineralization defects and vitamin D deficiency: histomorphometric analysis of iliac crest bone biopsies and circulating 25-hydroxyvitamin D in 675 patients. J. Bone Miner. Res. 25, 305–312 (2010).
Bischoff, H. A. et al. Effects of vitamin D and calcium supplementation on falls: a randomized controlled trial. J. Bone Miner. Res. 18, 343–351 (2003).
Vestergaard, P., Rejnmark, L. & Mosekilde, L. Are antiresorptive drugs effective against fractures in patients with diabetes? Calcif. Tissue Int. 88, 209–214 (2011).
Cummings, S. R. et al. Denosumab for prevention of fractures in postmenopausal women with osteoporosis. N. Engl. J. Med. 361, 756–765 (2009).
Keegan, T. H., Schwartz, A. V., Bauer, D. C., Sellmeyer, D. E. & Kelsey, J. L. Effect of alendronate on bone mineral density and biochemical markers of bone turnover in type 2 diabetic women: the fracture intervention trial. Diabetes Care 27, 1547–1553 (2004).
Favus, M. J. Bisphosphonates for osteoporosis. N. Engl. J. Med. 363, 2027–2035 (2010).
Hofbauer, L. C., Jakob, F. & Felsenberg, D. Bisphosphonates and atypical femoral fractures. N. Engl. J. Med. 363, 1084 (2010).
Acknowledgements
C. Hamann and L. C. Hofbauer are supported by grants from Elsbeth Bonhoff Foundation. C. Hamann and L. C. Hofbauer and K.-P. Günther are supported by Center for Regenerative Therapies Dresden seed grants. L. C. Hofbauer is also supported by Deutsche Forschungsgemeinschaft Transregio-67, project B2.
Author information
Authors and Affiliations
Contributions
C. Hamann and L. C. Hofbauer researched the data for and contributed equally to writing of the article. All authors provided a substantial contribution to discussions of the content and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
L. C. Hofbauer has worked as a consultant for Amgen, Merck and Novartis. He has received research grants from Amgen, Novartis and Nycomed. He has received hororaria from Amgen, Lilly, Merck, Novartis and Nycomed. The other authors declare no competing interests.
Rights and permissions
About this article
Cite this article
Hamann, C., Kirschner, S., Günther, KP. et al. Bone, sweet bone—osteoporotic fractures in diabetes mellitus. Nat Rev Endocrinol 8, 297–305 (2012). https://doi.org/10.1038/nrendo.2011.233
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2011.233
This article is cited by
-
The Role of Bone Cell Energetics in Altering Bone Quality and Strength in Health and Disease
Current Osteoporosis Reports (2023)
-
Efficacy of bisphosphonate therapy on postmenopausal osteoporotic women with and without diabetes: a prospective trial
BMC Endocrine Disorders (2022)
-
Effect of verapamil on bone mass, microstructure and mechanical properties in type 2 diabetes mellitus rats
BMC Musculoskeletal Disorders (2022)
-
Bone mass and microarchitecture in T2DM patients and corticosteroids therapy: the Bushehr Elderly Health program
Journal of Diabetes & Metabolic Disorders (2022)
-
Clinical features and burden of osteoporotic fractures among the elderly in the USA from 2016 to 2018
Archives of Osteoporosis (2022)