Key Points
-
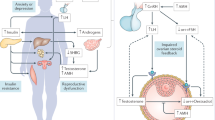
Testosterone activity, via the androgen receptor, on granulosa cells is increasingly recognized as essential for the early stages of folliculogenesis
-
Dehydroepiandrosterone (DHEA) supplementation of women with low functional ovarian reserve (LFOR) is a popular treatment for infertility, as these women almost universally have lower levels of testosterone than healthy women
-
DHEA supplementation has remained controversial primarily due to a lack of adequately powered clinical trials demonstrating its efficacy, but also owing to considerable variability in the metabolism of DHEA to testosterone
-
Approximately 10–15% of women who respond poorly to DHEA supplementation have insufficiently increased levels of testosterone—a major product of DHEA metabolism
-
Abnormally poor metabolism of DHEA to testosterone can be caused by a number of known genetic variants, as well as by endocrine and immune conditions
-
Hormonal responses to DHEA supplementation in women with LFOR can indicate genetic variants associated with poor metabolism of DHEA to testosterone and, thereby, suggest new personalized treatment options for these patients
Abstract
Hypoandrogenism in women with low functional ovarian reserve (LFOR, defined as an abnormally low number of small growing follicles) adversely affects fertility. The androgen precursor dehydroepiandrosterone (DHEA) is increasingly used to supplement treatment protocols in women with LFOR undergoing in vitro fertilization. Due to differences in androgen metabolism, however, responses to DHEA supplementation vary between patients. In addition to overall declines in steroidogenic capacity with advancing age, genetic factors, which result in altered expression or enzymatic function of key steroidogenic proteins or their upstream regulators, might further exacerbate variations in the conversion of DHEA to testosterone. In this Review, we discuss in vitro studies and animal models of polymorphisms and gene mutations that affect the conversion of DHEA to testosterone and attempt to elucidate how these variations affect female hormone profiles. We also discuss treatment options that modulate levels of testosterone by targeting the expression of steroidogenic genes. Common variants in genes encoding DHEA sulphotransferase, aromatase, steroid 5α-reductase, androgen receptor, sex-hormone binding globulin, fragile X mental retardation protein and breast cancer type 1 susceptibility protein have been implicated in androgen metabolism and, therefore, can affect levels of androgens in women. Short of screening for all potential genetic variants, hormonal assessments of patients with low testosterone levels after DHEA supplementation facilitate identification of underlying genetic defects. The genetic predisposition of patients can then be used to design individualized fertility treatments.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Weil, S. J. et al. Androgen receptor gene expression in the primate ovary: cellular localization, regulation, and functional correlations. J. Clin. Endocrinol. Metab. 83, 2479–2485 (1998).
Vendola, K. A., Zhou, J., Adesanya, O. O., Weil, S. J. & Bondy, C. A. Androgens stimulate early stages of follicular growth in the primate ovary. J. Clin. Invest. 101, 2622–2629 (1998).
Gleicher, N. et al. Hypoandrogenism in association with diminished functional ovarian reserve. Hum. Reprod. 28, 1084–1091 (2013).
Janse, F., Tanahatoe, S. J., Eijkemans, M. J. & Fauser, B. C. Testosterone concentrations, using different assays, in different types of ovarian insufficiency: a systematic review and meta-analysis. Hum. Reprod. Update 18, 405–419 (2012).
Reproductive Endocrinology and Infertility Committee et al. Advanced reproductive age and fertility. J. Obstet. Gynecol. Can. 33, 1165–1175 (2011).
Kim, C. H., Howles, C. M. & Lee, H. A. The effect of transdermal testosterone gel pretreatment on controlled ovarian stimulation and IVF outcome in low responders. Fertil. Steril. 95, 679–683 (2011).
Gleicher, N. et al. Starting and resulting testosterone levels after androgen supplementation determine at all ages in vitro fertilization (IVF) pregnancy rates in women with diminished ovarian reserve (DOR). J. Assist. Reprod. Genet. 30, 49–62 (2013).
Fanchin, R. et al. Androgens and poor responders: are we ready to take the plunge into clinical therapy? Fertil. Steril. 96, 1062–1065 (2011).
Sunkara, S. K., Coomarasamy, A., Artl, W. & Bhattacharva, S. Should androgen supplementation be used for poor ovarian response in IVF? Hum. Reprod. 27, 637–640 (2012).
Urman, B. & Yakin, K. Does dehydroepiandrosterone have any benefit in fertility treatment? Curr. Opin. Obstet. Gynecol. 24, 132–135 (2012).
Narkwichean, A., Maalouf, W., Campbell, B. K. & Jayaprakasan, K. Efficacy of dehydroepiandrosterone to improve ovarian response in women with diminished ovarian reserve: a meta-analysis. Reprod. Biol. Endocrinol. 11, 44 (2013).
Gleicher, N., Barad, D. H., Kushnir, V. A., Sen, A. & Weghofer, A. Poor responders and androgen adjuvant treatment: “Still haven't found what I'm looking for ...” Reprod. Biomed. Online 29, 650–652 (2014).
Wartofsky, L. & Handelsman, D. J. Standardization of hormonal assays for the 21st century. J. Clin. Endocrinol. Metab. 95, 5141–5143 (2010).
Gleicher, N. & Barad, D. H. Dehydroepiandrosterone (DHEA) supplementation in diminished ovarian reserve (DOR). Reprod. Biol. Endocrinol. 9, 67 (2011).
Prizant, H., Gleicher, N. & Sen, A. Androgen actions in the ovary: balance is key. J. Endocrinol. 222, R141–R151 (2014).
Arlt, W. Dehydroepiandrosterone replacement therapy. Semin. Reprod. Med. 22, 379–388 (2004).
Urbanski, H. F. et al. Androgen supplementation during aging: development of a physiologically appropriate protocol. Rejuvenation Res. 17, 150–153 (2014).
Ceci, R., Duranti, G., Rossi, A., Savini, I. & Sabatini, S. Skeletal muscle differentiation: role of dehydroepinadrosterone sulfate. Horm. Metab. Res. 43, 702–707 (2011).
Christianson, M. S. & Shen, W. Osteoporosis prevention and management: nonpharmacologic and lifestyle options. Clin. Obstet. Gynecol. 56, 703–710 (2013).
Savineau, J. P., Marthan, R. & Dumas de la Roque, E. Role of DHEA in cardiovascular diseases. Biochem. Pharmacol. 15, 718–726 (2013).
Seawalha, A. H. & Kovats, S. Dehydroepiandrosterone in systemic lupus erythematosus. Clin. Rheumatol. Rep. 10, 286–291 (2008).
Buford, T. W. & Willoughby, D. S. Impact of DHEA(S) and cortisol on immune function in aging: a brief review. Appl. Physiol. Nutr. Metab. 33, 429–433 (2008).
Forsblad-d'Elia, H., Carlsten, H., Labrie, F., Konttinen, Y. T. & Ohlsson, C. Low serum levels of sex steroids associated with disease characteristics in primary Sjogren syndrome; supplementation with dehydroepiandrosterone restores the concentrations. J. Clin. Endocrinol. Metab. 94, 2044–2051 (2009).
Kritz-Silverstein, D., von Mühlen, D., Laughlin, G. A. & Bettencourt, R. Effects of dehydroepiandrosterone supplementation on cognitive function and quality of life: the DHEA and Well-Ness (DAWN) trial. J. Am. Geriatr. Soc. 56, 1292–1298 (2008).
Gleicher, N., Weghofer, A. & Barad, D. H. The role of androgens in follicle maturation and ovulation induction: friend or foe of infertility treatment? Reprod. Biol. Endocrinol. 9, 116 (2011).
Traish, A. M., Kang, H. P., Saad, F. & Guay, A. T. Dehydroepiandrosterone (DHEA)—a precursor steroid or an active hormone in human physiology. J. Sex. Med. 8, 2960–2982 (2011).
Nippoldt, T. B. & Nair, K. S. Is there a case for DHEA replacement? Baillieres Clin. Endocrinol. Metab. 12, 507–520 (1998).
Labrie, F., Luu-The, V., Labrie, C. & Simard, J. DHEA and its transformation into androgens and estrogens in peripheral target tissues: intracrinology. Front. Neuroendocrinol. 22, 185–212 (2001).
Pluchino, N. et al. One-year therapy with 10 mg/day DHEA alone or in combination with HRT in postmenopausal women: effects on hormonal milieu. Maturitas 59, 293–303 (2008).
Labrie, F. et al. Bioavailability and metabolism of oral and percutaneous dehydroepiandrosterone in postmenopausal women. J. Steroid. Biochem. Mol. Biol. 107, 57–69 (2007).
Buster, J. E. et al. Postmenopausal steroid replacement with micronized dehydroepiandrosterone: preliminary oral bioavailability and dose proportionality studies. Am. J. Obstet. Gynecol. 166, 1163–1168 (1992).
Wolf, O. T. et al. Effects of a two-week physiological dehydroepiandrosterone substitution on cognitive performance and well-being in healthy elderly women and men. J. Clin. Endocrinol. Metab. 82, 2363–2367 (1997).
Rice, S. P. et al. Effects of dehydroepiandrosterone replacement on vascular function in primary and secondary adrenal insufficiency: a randomized crossover trial. J. Clin. Endocrinol. Metab. 94, 1966–1972 (2009).
Gurnell, E. M. et al. Long-term DHEA replacement in primary adrenal insufficiency: a randomized, controlled trial. J. Clin. Endocrinol. Metab. 93, 400–409 (2008).
Arlt, W. et al. Dehydroepiandrosterone replacement in women with adrenal insufficiency. N. Engl. J. Med. 341, 1013–1020 (1999).
Johannsson, G. et al. Low dose dehydroepiandrosterone affects behavior in hypopituitary androgen-deficient women: a placebo-controlled trial. J. Clin. Endocrinol. Metab. 87, 2046–2052 (2002).
Mainwaring, W. I. The mechanism of action of androgens. Monogr. Endocrinol. 10, 1–178 (1977).
Mo, Q., Lu, S. F., Hu, S. & Simon, N. G. DHEA and DHEA sulfate differentially regulate neural androgen receptor and its transcriptional activity. Brain Res. Mol. Brain Res. 126, 165–172 (2004).
Mo, Q., Lu, S. F. & Simon, N. G. Dehydroepiandrosterone and its metabolites: differential effects on androgen receptor trafficking and transcriptional activity. J. Steroid Biochem. Mol. Biol. 99, 50–58 (2006).
Lu, S. F., Mo, Q., Hu, S., Garippa, C. & Simon, N. G. Dehydroepiandrosterone upregulates neural androgen receptor level and transcriptional activity. J. Neurobiol. 57, 163–171 (2003).
Grino, P. B., Griffin, J. E. & Wilson, J. D. Testosterone at high concentrations interacts with the human androgen receptor similarly to dihydrotestosterone. Endocrinology 126, 1165–1172 (1990).
Longcope, C. Adrenal and gonadal androgen secretion in normal females. Clin. Endocrinol. Metab. 15, 213–228 (1986).
Labrie, F. et al. Endocrine and intracrine sources of androgens in women: inhibition of breast cancer and other roles of androgens and their precursor dehydroepiandrosterone. Endocr. Rev. 24, 152–182 (2003).
Shifren, J. L. et al. Transdermal testosterone treatment in women with impaired sexual function after oophorectomy. N. Engl. J. Med. 343, 682–688 (2000).
Sunkara, S. K., Coomarasamy, A., Arlt, W. & Bhattacharya, S. Should androgen supplementation be used for poor ovarian response in IVF? Hum. Reprod. 27, 637–640 (2012).
Cameron, D. R. & Braunstein, G. D. The use of dehydroepiandrosterone therapy in clinical practice. Treat. Endocrinol. 4, 95–114 (2005).
Dhatariya, K. K. & Nair, K. S. Dehydroepiandrosterone: is there a role for replacement? Mayo Clin. Proc. 78, 1257–1273 (2003).
Erichsen, M. M., Husebye, E. S., Michelsen, T. M., Dahl, A. A. & Lovas, K. Sexuality and fertility in women with Addison's disease. J. Clin. Endocrinol. Metab. 95, 4354–4360 (2010).
Zumoff, B., Strain, G. W., Miller, L. K. & Rosner, W. Twenty-four-hour mean plasma testosterone concentration declines with age in normal premenopausal women. J. Clin. Endocrinol. Metab. 80, 1429–1430 (1995).
Hugues, J. N. et al. Assessment of theca cell function prior to controlled ovarian stimulation: the predictive value of serum basal/stimulated steroid levels. Hum. Reprod. 25, 228–234 (2010).
Nardo, L. G., Yates, A. P., Roberts, S. A., Pemberton, P. & Laing, I. The relationships between AMH, androgens, insulin resistance and basal ovarian follicular status in non-obese subfertile women with and without polycystic ovary syndrome. Hum. Reprod. 24, 2917–2923 (2009).
Otterness, D. M. & Weinshilboum, R. Human dehydroepiandrosterone sulfotransferase: molecular cloning of cDNA and genomic DNA. Chem. Biol. Interact. 92, 145–59 (1994).
Strott, C. A. Sulfonation and molecular action. Endocr. Rev. 23, 703–732 (2002).
Longcope, C. Dehydroepiandrosterone metabolism. J. Endocrinol. 150 (Suppl. 3), S125–S127 (1996).
Reed, M. J., Purohit, A., Woo, L. W., Newman, S. P. & Potter, B. V. Steroid sulfatase: molecular biology, regulation, and inhibition. Endocr. Rev. 26, 171–202 (2005).
Asif, A. R. et al. Regulation of steroid hormone biosynthesis enzymes and organic anion transporters by forskolin and DHEA-S treatment in adrenocortical cells. Am. J. Physiol. Endocrinol. Metab. 291, E1351–E1359 (2006).
Nowell, S. & Falany, C. N. Pharmacogenetics of human cytosolic sulfotransferases. Oncogene 25, 1673–1678 (2006).
Parker, C. R. Jr. Dehydroepiandrosterone and dehydroepiandrosterone sulfate production in the human adrenal during development and aging. Steroids 64, 640–647 (1999).
Weghofer, A., Kim, A., Barad, D. H. & Gleicher, N. The impact of androgen metabolism and FMR1 genotypes on pregnancy potential and in women with dehydroepiandrosterone (DHEA) supplementation. Hum. Reprod. 27, 3287–3293 (2012).
Jaquish, C. E., Blangero, J., Haffner, S. M., Stern, M. P. & Maccluer, J. W. Quantitative genetics of dehydroepiandrosterone sulfate and its relation to possible cardiovascular disease risk factors in Mexican Americans. Hum. Hered. 46, 301–309 (1996).
An, P. et al. Genome-wide linkage scan to detect loci influencing levels of dehydroepiandrosterones in the HERITAGE Family Study. Metabolism 50, 1315–1322 (2001).
Meikle, A. W., Stringham, J. D., Woodward, M. G. & Bishop, D. T. Heritability of variation of plasma cortisol levels. Metabolism 37, 514–517 (1988).
Rotter, J. I., Wong, F. L., Lifrak, E. T. & Parker, L. N. A genetic component to the variation of dehydroepiandrosterone sulfate. Metabolism 34, 731–736 (1985).
Rice, T. et al. The Cincinnati Myocardial Infarction and Hormone Family Study: family resemblance for dehydroepiandrosterone sulfate in control and myocardial infarction families. Metabolism 42, 1284–1290 (1993).
Zhai, G. et al. Eight common genetic variants associated with serum DHEAS levels suggest a key role in ageing mechanisms. PLoS Genet. 7, e1002025 (2011).
Kumar, A., Woods, K. S., Bartolucci, A. A. & Azziz, R. Prevalence of adrenal androgen excess in patients with the polycystic ovary syndrome (PCOS). Clin. Endocrinol. (Oxf.) 62, 644–649 (2005).
Thomae, B. A., Eckloff, B. W., Freimuth, R. R., Wieben, E. D. & Weinshilboum, R. M. Human sulfotransferase SULT2A1 pharmacogenetics: genotype-to-phenotype studies. Pharmacogenomics J. 2, 48–56 (2002).
Tal, R. & Seifer, D. in Ethnic Differences in Fertility and Assisted Reproduction (ed. Sharara, F.) 73–84 (Springer, 2013).
Doldi, N., Belvisi, L., Bassan, M., Fusi, F. M. & Ferrari, A. Premature ovarian failure: steroid synthesis and autoimmunity. Gynecol. Endocrinol. 12, 23–28 (1998).
Kim, M. S., Shigenaga, J., Moser, A., Grunfeld, C. & Feingold, K. R. Suppression of DHEA sulfotransferase (Sult2A1) during the acute-phase response. Am. J. Physiol. Endocrinol. Metab. 287, E731–E738 (2004).
Parker, C. R. Jr. et al. Effects of ACTH and cytokines on dehydroepiandrosterone sulfotransferase messenger RNA in human adrenal cells. Endocr. Res. 24, 669–673 (1998).
Stankovic, A. K., Dion, L. D. & Parker, C. R. Jr. Effects of transforming growth factor-β on human fetal adrenal steroid production. Mol. Cell. Endocrinol. 99, 145–151 (1994).
Lebrethon, M. C., Jaillard, C., Naville, D., Begeot, M. & Saez, J. M. Regulation of corticotropin and steroidogenic enzyme mRNAs in human fetal adrenal cells by corticotropin, angiotensin-II and transforming growth factor β1. Mol. Cell. Endocrinol. 106, 137–143 (1994).
Lebrethon, M. C., Jaillard, C., Naville, D., Begeot, M. & Saez, J. M. Effects of transforming growth factor-β1 on human adrenocortical fasciculata-reticularis cell differentiated functions. J. Clin. Endocrinol. Metab. 79, 1033–1039 (1994).
Naz, R. K., Thurston, D. & Santoro, N. Circulating tumor necrosis factor (TNF)-α in normally cycling women and patients with premature ovarian failure and polycystic ovaries. Am. J. Reprod. Immunol. 34, 170–175 (1995).
Cianci, A. et al. Relationship between tumour necrosis factor α and sex steroid concentrations in the follicular fluid of women with immunological infertility. Hum. Reprod. 11, 265–268 (1996).
Guerra-Infante, F. M. et al. Tumor necrosis factor in peritoneal fluid from asymptomatic infertile women. Arch. Med. Res. 30, 138–143 (1999).
Horka, P. et al. Intracellular cytokine production in peripheral blood lymphocytes: a comparison of values in infertile and fertile women. Am. J. Reprod. Immunol. 65, 466–469 (2011).
Winger, E. E. et al. Treatment with adalimumab (Humira) and intravenous immunoglobulin improves pregnancy rates in women undergoing IVF. Am. J. Reprod. Immunol. 61, 113–120 (2009).
Winger, E. E. & Reed, J. L. Treatment with tumor necrosis factor inhibitors and intravenous immunoglobulin improves live birth rates in women with recurrent spontaneous abortion. Am. J. Reprod. Immunol. 60, 8–16 (2008).
Chen, S. et al. Autoantibodies to steroidogenic enzymes in autoimmune polyglandular syndrome, Addison's disease, and premature ovarian failure. J. Clin. Endocrinol. Metab. 81, 1871–1876 (1996).
Forges, T., Monnier-Barbarino, P., Faure, G. C. & Bene, M. C. Autoimmunity and antigenic targets in ovarian pathology. Hum. Reprod. Update 10, 163–175 (2004).
Reato, G. et al. Premature ovarian failure in patients with autoimmune Addison's disease: clinical, genetic, and immunological evaluation. J. Clin. Endocrinol. Metab. 96, E1255–E1261 (2011).
Falorni, A. et al. Progressive decline of residual follicle pool after clinical diagnosis of autoimmune ovarian insufficiency. Clin. Endocrinol. (Oxf.) 77, 453–458 (2012).
Canguven, O. & Albayrak, S. Do low testosterone levels contribute to the pathogenesis of asthma? Med. Hypotheses 76, 585–588 (2011).
Cutolo, M. et al. Sex hormones influence on the immune system: basic and clinical aspects in autoimmunity. Lupus 13, 635–638 (2004).
Dragojevic-Dikic, S. et al. An immunological insight into premature ovarian failure (POF). Autoimmun. Rev. 9, 771–774 (2010).
Sen, A., Kushnir, V. A., Barad, D. H. & Gleicher, N. Endocrine autoimmune disease and female infertility. Nat. Rev. Endocrinol. 10, 37–50 (2014).
Simpson, E. R. et al. Tissue-specific promoters regulate aromatase cytochrome P450 expression. J. Steroid Biochem. Mol. Biol. 44, 321–330 (1993).
Petry, C. J. et al. Association of aromatase (CYP19) gene variation with features of hyperandrogenism in two populations of young women. Hum. Reprod. 20, 1837–1843 (2005).
Zhang, X. L. et al. SNP rs2470152 in CYP19 is correlated to aromatase activity in Chinese polycystic ovary syndrome patients. Mol. Med. Rep. 5, 245–249 (2012).
Sowers, M. R., Wilson, A. L., Kardia, S. R., Chu, J. & Ferrell, R. Aromatase gene (CYP19) polymorphisms and endogenous androgen concentrations in a multiracial/multiethnic, multisite study of women at midlife. Am. J. Med. 119, S23–S30 (2006).
Kristensen, V. N. et al. Genetic variants of CYP19 (aromatase) and breast cancer risk. Oncogene 19, 1329–1333 (2000).
Haiman, C. A., Hankinson, S. E., Spiegelman, D., Brown, M. & Hunter, D. J. No association between a single nucleotide polymorphism in CYP19 and breast cancer risk. Cancer Epidemiol. Biomarkers Prev. 11, 215–216 (2002).
Michaelson-Cohen, R. et al. BRCA mutation carriers do not have compromised ovarian reserve. Int. J. Gynecol. Cancer 24, 233–237 (2014).
Titus, S. et al. Impairment of BRCA1-related DNA double-strand break repair leads to ovarian aging in mice and humans. Sci. Transl Med. 5, 172ra2 (2013).
Oktay, K. et al. Age-related decline in DNA repair function explains diminished ovarian reserve, earlier menopause, and possible oocyte vulnerability to chemotherapy in women with BRCA mutations. J. Clin. Oncol. 32, 1093–1094 (2014).
Wang, E. T. et al. BRCA1 germline mutations may be associated with reduced ovarian reserve. Fertil. Steril. 102, 1723–1728 (2014).
Chand, A. L., Simpson, E. R. & Clyne, C. D. Aromatase expression is increased in BRCA1 mutation carriers. BMC Cancer 9, 148 (2009).
Hu, Y. et al. Modulation of aromatase expression by BRCA1: a possible link to tissue-specific tumor suppression. Oncogene 24, 8343–8348 (2005).
Phillips, K. W. et al. Brca1 is expressed independently of hormonal stimulation in the mouse ovary. Lab. Invest. 76, 419–425 (1997).
Lu, Y. et al. Ubiquitination and proteasome-mediated degradation of BRCA1 and BARD1 during steroidogenesis in human ovarian granulosa cells. Mol. Endocrinol. 21, 651–663 (2007).
Oktay, K., Kim, J. Y., Barad, D. & Babayev, S. N. Association of BRCA1 mutations with occult primary ovarian insufficiency: a possible explanation for the link between infertility and breast/ovarian cancer risks. J. Clin. Oncol. 28, 240–244 (2010).
Lenie, S. & Smitz, J. Functional AR signaling is evident in an in vitro mouse follicle culture bioassay that encompasses most stages of folliculogenesis. Biol. Reprod. 80, 685–695 (2009).
Tetsuka, M. et al. Developmental regulation of androgen receptor in rat ovary. J. Endocrinol. 145, 535–543 (1995).
Kimura, S. et al. Androgen receptor function in folliculogenesis and its clinical implication in premature ovarian failure. Trends Endocrinol. Metab. 18, 183–189 (2007).
Jeppesen, J. V. et al. Which follicles make the most anti-Mullerian hormone in humans? Evidence for an abrupt decline in AMH production at the time of follicle selection. Mol. Hum. Reprod. 19, 519–527 (2013).
Mitwally, M. F., Casper, R. F. & Diamond, M. P. The role of aromatase inhibitors in ameliorating deleterious effects of ovarian stimulation on outcome of infertility treatment. Reprod. Biol. Endocrinol. 3, 54 (2005).
Simon, C., Cano, F., Valbuena, D., Remohi, J. & Pellicer, A. Clinical evidence for a detrimental effect on uterine receptivity of high serum oestradiol concentrations in high and normal responder patients. Hum. Reprod. 10, 2432–2437 (1995).
Feigenberg, T., Simon, A., Ben-Meir, A., Gielchinsky, Y. & Laufer, N. Role of androgens in the treatment of patients with low ovarian response. Reprod. Biomed. Online 19, 888–898 (2009).
Casper, R. F. & Mitwally, M. F. Review: aromatase inhibitors for ovulation induction. J. Clin. Endocrinol. Metab. 91, 760–771 (2006).
Weil, S., Vendola, K., Zhou, J. & Bondy, C. A. Androgen and follicle-stimulating hormone interactions in primate ovarian follicle development. J. Clin. Endocrinol. Metab. 84, 2951–2956 (1999).
Verpoest, W. M. et al. Aromatase inhibitors in ovarian stimulation for IVF/ICSI: a pilot study. Reprod. Biomed. Online 13, 166–172 (2006).
Kwee, J., Elting, M. E., Schats, R., McDonnell, J. & Lambalk, C. B. Ovarian volume and antral follicle count for the prediction of low and hyper responders with in vitro fertilization. Reprod. Biol. Endocrinol. 5, 9 (2007).
Garcia-Velasco, J. A. et al. The aromatase inhibitor letrozole increases the concentration of intraovarian androgens and improves in vitro fertilization outcome in low responder patients: a pilot study. Fertil. Steril. 84, 82–87 (2005).
Andersen, C. Y. & Lossl, K. Increased intrafollicular androgen levels affect human granulosa cell secretion of anti-Mullerian hormone and inhibin-B. Fertil. Steril. 89, 1760–1765 (2008).
Brueggemeier, R. W., Su, B., Sugimoto, Y., Diaz-Cruz, E. S. & Davis, D. D. Aromatase and COX in breast cancer: enzyme inhibitors and beyond. J. Steroid Biochem. Mol. Biol. 106, 16–23 (2007).
Davies, G., Martin, L. A., Sacks, N. & Dowsett, M. Cyclooxygenase-2 (COX-2), aromatase and breast cancer: a possible role for COX-2 inhibitors in breast cancer chemoprevention. Ann. Oncol. 13, 669–678 (2002).
Lu, M. et al. BRCA1 negatively regulates the cancer-associated aromatase promoters I.3 and II in breast adipose fibroblasts and malignant epithelial cells. J. Clin. Endocrinol. Metab. 91, 4514–1519 (2006).
Subbaramaiah, K., Hudis, C., Chang, S. H., Hla, T. & Dannenberg, A. J. EP2 and EP4 receptors regulate aromatase expression in human adipocytes and breast cancer cells. Evidence of a BRCA1 and p300 exchange. J. Biol. Chem. 283, 3433–3444 (2008).
Jakimiuk, A. J., Weitsman, S. R. & Magoffin, D. A. 5α-reductase activity in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 84, 2414–2418 (1999).
Russell, D. W. et al. The molecular genetics of steroid 5α-reductases. Recent Prog. Horm. Res. 49, 275–284 (1994).
Abraham, G. E. Ovarian and adrenal contribution to peripheral androgens during the menstrual cycle. J. Clin. Endocrinol. Metab. 39, 340–346 (1974).
Fassnacht, M. et al. Beyond adrenal and ovarian androgen generation: increased peripheral 5α-reductase activity in women with polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 88, 2760–2766 (2003).
Bauer, E. R., Daxenberger, A., Petri, T., Sauerwein, H. & Meyer, H. H. Characterisation of the affinity of different anabolics and synthetic hormones to the human androgen receptor, human sex hormone binding globulin and to the bovine progestin receptor. APMIS 108, 838–846 (2000).
Askew, E. B., Gampe, R. T. Jr, Stanley, T. B., Faggart, J. L. & Wilson, E. M. Modulation of androgen receptor activation function 2 by testosterone and dihydrotestosterone. J. Biol. Chem. 282, 25801–25816 (2007).
Hamel, M., Vanselow, J., Nicola, E. S. & Price, C. A. Androstenedione increases cytochrome P450 aromatase messenger ribonucleic acid transcripts in nonluteinizing bovine granulosa cells. Mol. Reprod. Dev. 70, 175–183 (2005).
Wu, Y. G., Bennett, J., Talla, D. & Stocco, C. Testosterone, not 5α-dihydrotestosterone, stimulates LRH-1 leading to FSH-independent expression of Cyp19 and P450scc in granulosa cells. Mol. Endocrinol. 25, 656–668 (2011).
Beigi, A., Sobhi, A. & Zarrinkoub, F. Finasteride versus cyproterone acetate–estrogen regimens in the treatment of hirsutism. Int. J. Gynaecol. Obstet. 87, 29–33 (2004).
Tolino, A. et al. Finasteride in the treatment of hirsutism: new therapeutic perspectives. Fertil. Steril. 66, 61–65 (1996).
Goodarzi, M. O. et al. Variants in the 5α-reductase type 1 and type 2 genes are associated with polycystic ovary syndrome and the severity of hirsutism in affected women. J. Clin. Endocrinol. Metab. 91, 4085–40891 (2006).
Makridakis, N. M., di Salle, E. & Reichardt, J. K. Biochemical and pharmacogenetic dissection of human steroid 5α-reductase type II. Pharmacogenetics 10, 407–413 (2000).
Graupp, M., Wehr, E., Schweighofer, N., Pieber, T. R. & Obermayer-Pietsch, B. Association of genetic variants in the two isoforms of 5α-reductase, SRD5A1 and SRD5A2, in lean patients with polycystic ovary syndrome. Eur. J. Obstet. Gynecol. Reprod. Biol. 157, 175–179 (2011).
Munzker, J. et al. Testosterone to dehyrotestosterone ratio as a new biomarker for an adverse metabolic phenotype in the polycystic ovary syndrome J. Clin. Endocrinol. Metab. 100, 653–660 (2015).
Ueshiba, H. et al. Serum androgen levels in hyperthyroid women. Exp. Clin. Endocrinol. Diabetes 105, 359–362 (1997).
Buchanan, G. et al. Structural and functional consequences of glutamine tract variation in the androgen receptor. Hum. Mol. Genet. 13, 1677–16792 (2004).
Brinkmann, A. O. et al. The human androgen receptor: domain structure, genomic organization and regulation of expression. J. Steroid Biochem. 34, 307–310 (1989).
Kazemi-Esfarjani, P., Trifiro, M. A. & Pinsky, L. Evidence for a repressive function of the long polyglutamine tract in the human androgen receptor: possible pathogenetic relevance for the (CAG)n-expanded neuronopathies. Hum. Mol. Genet. 4, 523–527 (1995).
Chamberlain, N. L., Driver, E. D. & Miesfeld, R. L. The length and location of CAG trinucleotide repeats in the androgen receptor N-terminal domain affect transactivation function. Nucleic Acids Res. 22, 3181–3186 (1994).
Tut, T. G., Ghadessy, F. J., Trifiro, M. A., Pinsky, L. & Yong, E. L. Long polyglutamine tracts in the androgen receptor are associated with reduced trans-activation, impaired sperm production, and male infertility. J. Clin. Endocrinol. Metab. 82, 3777–3782 (1997).
Irvine, R. A. et al. Inhibition of p160-mediated coactivation with increasing androgen receptor polyglutamine length. Hum. Mol. Genet. 9, 267–274 (2000).
Mhatre, A. N. et al. Reduced transcriptional regulatory competence of the androgen receptor in X-linked spinal and bulbar muscular atrophy. Nat. Genet. 5, 184–188 (1993).
Shiina, H. et al. Premature ovarian failure in androgen receptor-deficient mice. Proc. Natl Acad. Sci. USA 103, 224–229 (2006).
Hu, Y. C. et al. Subfertility and defective folliculogenesis in female mice lacking androgen receptor. Proc. Natl Acad. Sci. USA 101, 11209–11214 (2004).
Sen, A. & Hammes, S. R. Granulosa cell-specific androgen receptors are critical regulators of ovarian development and function. Mol. Endocrinol. 24, 1393–1403 (2010).
Walters, K. A. et al. Female mice haploinsufficient for an inactivated androgen receptor (AR) exhibit age-dependent defects that resemble the AR null phenotype of dysfunctional late follicle development, ovulation, and fertility. Endocrinology 148, 3674–3684 (2007).
Walters, K. A. et al. Subfertile female androgen receptor knockout mice exhibit defects in neuroendocrine signaling, intraovarian function, and uterine development but not uterine function. Endocrinology 150, 3274–3282 (2009).
Chatterjee, S. et al. Longer CAG repeat length in the androgen receptor gene is associated with premature ovarian failure. Hum. Reprod. 24, 3230–3235 (2009).
Aruna, M. et al. Role of androgen receptor CAG repeat polymorphism and X-inactivation in the manifestation of recurrent spontaneous abortions in Indian women. PLoS ONE 6, e17718 (2011).
Misao, R., Nakanishi, Y., Fujimoto, J., Ichigo, S. & Tamaya, T. Expression of sex hormone-binding globulin and corticosteroid-binding globulin mRNAs in corpus luteum of human subjects. Horm. Res. 48, 191–195 (1997).
Noe, G. Sex hormone binding globulin expression and colocalization with estrogen receptor in the human fallopian tube. J. Steroid Biochem. Mol. Biol. 68, 111–117 (1999).
Forges, T., Gerard, A., Hess, K., Monnier-Barbarino, P. & Gerard, H. Expression of sex hormone-binding globulin (SHBG) in human granulosa-lutein cells. Mol. Cell. Endocrinol. 219, 61–68 (2004).
Forges, T., Gerard, A., Monnier-Barbarino, P. & Gerard, H. Immunolocalization of sex hormone-binding globulin (SHBG) in human ovarian follicles and corpus luteum. Histochem. Cell Biol. 124, 285–290 (2005).
Larrea, F. et al. Evidence that human placenta is a site of sex hormone-binding globulin gene expression. J. Steroid Biochem. Mol. Biol. 46, 497–505 (1993).
Moore, K. H., Bertram, K. A., Gomez, R. R., Styner, M. J. & Matej, L. A. Sex hormone binding globulin mRNA in human breast cancer: detection in cell lines and tumor samples. J. Steroid Biochem. Mol. Biol. 59, 297–304 (1996).
Rosner, W., Hryb, D. J., Khan, M. S., Nakhla, A. M. & Romas, N. A. Androgen and estrogen signaling at the cell membrane via G-proteins and cyclic adenosine monophosphate. Steroids 64, 100–106 (1999).
Hammes, A. et al. Role of endocytosis in cellular uptake of sex steroids. Cell 122, 751–762 (2005).
Gomez, J. M. Serum leptin, insulin-like growth factor-I components and sex-hormone binding globulin. Relationship with sex, age and body composition in healthy population. Protein Pept. Lett. 14, 708–711 (2007).
Estour, B. et al. Sex hormone binding globulin in women with anorexia nervosa. Clin. Endocrinol. (Oxf.) 24, 571–576 (1986).
O'Dea, J. P. et al. Effect of dietery weight loss on sex steroid binding sex steroids, and gonadotropins in obese postmenopausal women. J. Lab. Clin. Med. 93, 1004–1008 (1979).
Toscano, V. et al. Steroidal and non-steroidal factors in plasma sex hormone binding globulin regulation. J. Steroid Biochem. Mol. Biol. 43, 431–437 (1992).
Haffner, S. M. Sex hormone-binding protein, hyperinsulinemia, insulin resistance and noninsulin-dependent diabetes. Horm. Res. 45, 233–237 (1996).
Preziosi, P. et al. Interrelation between plasma sex hormone-binding globulin and plasma insulin in healthy adult women: the telecom study. J. Clin. Endocrinol. Metab. 76, 283–287 (1993).
Nestler, J. E. et al. A direct effect of hyperinsulinemia on serum sex hormone-binding globulin levels in obese women with the polycystic ovary syndrome. J. Clin. Endocrinol. Metab. 72, 83–89 (1991).
Fritz, M. A. & Speroff, L. Clinical Gynecologic Endocrinology and Infertility (Lippincott Williams & Wilkins, 2011).
Lecomte, P., Lecureuil, N., Lecureuil, M., Osorio Salazar, C. & Valat, C. Age modulates effects of thyroid dysfunction on sex hormone binding globulin (SHBG) levels. Exp. Clin. Endocrinol. Diabetes 103, 339–342 (1995).
Tagawa, N. et al. Serum dehydroepiandrosterone, dehydroepiandrosterone sulfate, and pregnenolone sulfate concentrations in patients with hyperthyroidism and hypothyroidism. Clin. Chem. 46, 523–528 (2000).
Xita, N. & Tsatsoulis, A. Genetic variants of sex hormone-binding globulin and their biological consequences. Mol. Cell. Endocrinol. 316, 60–65 (2010).
Hogeveen, K. N., Talikka, M. & Hammond, G. L. Human sex hormone-binding globulin promoter activity is influenced by a (TAAAA)n repeat element within an Alu sequence. J. Biol. Chem. 276, 36383–36390 (2001).
Haiman, C. A. et al. Common genetic variation in the sex steroid hormone-binding globulin (SHBG) gene and circulating SHBG levels among postmenopausal women: the multiethnic cohort. J. Clin. Endocrinol. Metab. 90, 2198–2204 (2005).
Xita, N., Tsatsoulis, A., Chatzikyriakidou, A. & Georgiou, I. Association of the (TAAAA)n repeat polymorphism in the sex hormone-binding globulin (SHBG) gene with polycystic ovary syndrome and relation to SHBG serum levels. J. Clin. Endocrinol. Metab. 88, 5976–5980 (2003).
Ferk, P., Teran, N. & Gersak, K. The (TAAAA)n microsatellite polymorphism in the SHBG gene influences serum SHBG levels in women with polycystic ovary syndrome. Hum. Reprod. 22, 1031–1036 (2007).
Janse, F. et al. Similar phenotype characteristics comparing familial and sporadic premature ovarian failure. Menopause 17, 758–765 (2010).
Sutton-Tyrrell, K. et al. Sex-hormone-binding globulin and the free androgen index are related to cardiovascular risk factors in multiethnic premenopausal and perimenopausal women enrolled in the Study of Women Across the Nation (SWAN). Circulation 111, 1242–1249 (2005).
Morales, A. J., Haubrich, R. H., Hwang, J. Y., Asakura, H. & Yen, S. S. The effect of six months treatment with a 100 mg daily dose of dehydroepiandrosterone (DHEA) on circulating sex steroids, body composition and muscle strength in age-advanced men and women. Clin. Endocrinol. (Oxf.) 49, 421–432 (1998).
Kim, M. & Ceman, S. Fragile X mental retardation protein: past, present and future. Curr. Protein Pept. Sci. 13, 358–371 (2012).
Sullivan, S. D., Welt, C. & Sherman, S. FMR1 and the continuum of primary ovarian insufficiency. Semin. Reprod. Med. 29, 299–307 (2011).
Pirozzi, F., Tabolacci, E. & Neri, G. The FRAXopathies: definition, overview, and update. Am. J. Med. Genet. A 155A, 1803–1816 (2011).
Willemsen, R., Levenga, J. & Oostra, B. A. CGG repeat in the FMR1 gene: size matters. Clin. Genet. 80, 214–225 (2011).
Gleicher, N., Weghofer, A., Lee, I. H. & Barad, D. H. FMR1 genotype with autoimmunity-associated polycystic ovary-like phenotype and decreased pregnancy chance. PLoS ONE 5, e15303 (2010).
Gleicher, N., Weghofer, A., Lee, I. H. & Barad, D. H. Association of FMR1 genotypes with in vitro fertilization (IVF) outcomes based on ethnicity/race. PLoS ONE 6, e18781 (2011).
Gleicher, N., Kim, A., Weghofer, A. & Barad, D. H. Differences in ovarian aging patterns between races are associated with ovarian genotypes and sub-genotypes of the FMR1 gene. Reprod. Biol. Endocrinol. 10, 77 (2012).
Kushnir, V. A., Himaya, E., Barad, D. H., Weghofer, N. & Gleicher, N. Early decline in functional ovarian reserve in young oocyte donors based on FMR1 genotypes and sub-genotypes. Fertil. Steril. 100 (Suppl.), S114 (2013).
Geva, E., Amit, A., Lerner-Geva, L. & Lessing, J. B. Autoimmunity and reproduction. Fertil. Steril. 67, 599–611 (1997).
Gleicher, N., Weghofer, A., Oktay, K. & Barad, D. H. Do etiologies of premature ovarian aging (POA) mimic those of premature ovarian failure (POF). Hum. Reprod. 24, 2395–2400 (2009).
Shi, X., Li, N., Liao, C., Shu, Q. & Zhu, F. Glucocorticoid or androgen for autoimmune premature ovary failure in mice. Zhong Nan Da Xue Xue Bao Yi Xue Ban 34, 576–581 (2009).
Acknowledgements
The authors acknowledge R. Tal for critically reviewing the manuscript.
Author information
Authors and Affiliations
Contributions
A.S.-T. and N.G. researched data for the article, provided substantial contributions to discussions of the content, wrote the article and reviewed and/or edited the manuscript before submission. A.S., D.H.B and V.K. reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
N.G. and D.H.B. are listed as co-inventors on four awarded US patents, which claim therapeutic benefits of DHEA and other androgens in women with low functional ovarian reserve (US 7615544: 'Method of improving cumulative embryo score and quantity of fertilized oocytes, increasing euploidy rate and of normalizing ovarian function using an androgen such as dehydroepiandrosterone'; US 8067400: 'Androgen treatment in females'; US 8501719: 'Androgen treatment in females'; US 8501718: 'Androgen treatment in females'). N.G. and D.H.B. are also listed as co-inventors on two awarded US patents, which are not related to the subject of this Review (US 8629120: 'Method of treatment related to the FMR1 gene'; and US 8951724: 'Detection of infertility risk and premature ovarian aging, addressing various claimed effects and diagnostic functions of the FMR1 gene on ovaries and female fertility'). N.G. owns shares in Fertility Nutraceuticals, a company that sells DHEA products, and both N.G. and D.H.B. receive patent royalties from this company. N.G. is also the owner of The Center for Human Reproduction, New York, NY, USA. A.S. T., A.S. and V.K. declare no competing interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Shohat-Tal, A., Sen, A., Barad, D. et al. Genetics of androgen metabolism in women with infertility and hypoandrogenism. Nat Rev Endocrinol 11, 429–441 (2015). https://doi.org/10.1038/nrendo.2015.64
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2015.64
This article is cited by
-
Dehydroepiandrosterone supplementation and the impact of follicular fluid metabolome and cytokinome profiles in poor ovarian responders
Journal of Ovarian Research (2023)
-
Effect of basal serum testosterone level on the ovarian response and the cumulative live birth rate in infertile women undergoing in vitro fertilization
Journal of Assisted Reproduction and Genetics (2023)
-
Effects of dehydroepiandrosterone (DHEA) supplementation on sexual function in premenopausal infertile women
Endocrine (2019)
-
Effect of race and ethnicity on utilization and outcomes of assisted reproductive technology in the USA
Reproductive Biology and Endocrinology (2017)
-
Potential therapeutic applications of human anti-Müllerian hormone (AMH) analogues in reproductive medicine
Journal of Assisted Reproduction and Genetics (2017)