Key Points
-
Impaired psychological well-being and depression or anxiety affect about 5–10% of hypothyroid patients on levothyroxine, despite normal TSH levels
-
Persistent symptoms might be explained by factors unrelated to thyroid disease, unrecognized autoimmune disease, or inability of levothyroxine to restore T3 levels in serum and all target tissues
-
Randomized clinical trials have in general not shown superiority of levothyroxine plus liothyronine combination therapy over levothyroxine monotherapy
-
Some reports suggest a preference of patients for levothyroxine plus liothyronine combination therapy over levothyroxine monotherapy, which might be associated with weight loss
-
Levothyroxine remains the standard treatment modality for hypothyroidism, but levothyroxine plus liothyronine combination treatment might be offered to selected patients as an experimental modality, according to guidelines published in 2012
Abstract
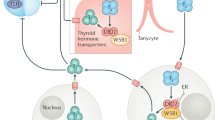
Impaired psychological well-being, depression or anxiety are observed in 5–10% of hypothyroid patients receiving levothyroxine, despite normal TSH levels. Such complaints might hypothetically be related to increased free T4 and decreased free T3 serum concentrations, which result in the abnormally low free T4:free T3 ratios observed in 30% of patients on levothyroxine. Evidence is mounting that levothyroxine monotherapy cannot assure a euthyroid state in all tissues simultaneously, and that normal serum TSH levels in patients receiving levothyroxine reflect pituitary euthyroidism alone. Levothyroxine plus liothyronine combination therapy is gaining in popularity; although the evidence suggests it is generally not superior to levothyroxine monotherapy, in some of the 14 published trials this combination was definitely preferred by patients and associated with improved metabolic profiles. Disappointing results with combination therapy could be related to use of inappropriate levothyroxine and liothyronine doses, resulting in abnormal serum free T4:free T3 ratios. Alternatively, its potential benefit might be confined to patients with specific genetic polymorphisms in thyroid hormone transporters and deiodinases that affect the intracellular levels of T3 available for binding to T3 receptors. Levothyroxine monotherapy remains the standard treatment for hypothyroidism. However, in selected patients, new guidelines suggest that experimental combination therapy might be considered.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Jackson, I. M. & Cobb, W. E. Why does anyone still use desiccated thyroid USP? Am. J. Med. 64, 284–288 (1978).
Singer, P. A. et al. Treatment guidelines for patients with hyperthyroidism and hypothyroidism. Standards of Care Committee, American Thyroid Association. JAMA 273, 808–812 (1995).
Vanderpump, M. P. J., Ahlquist, J. A. O., Franklyn, J. A. & Clayton, R. N., Consensus statement for good practice and audit measures in the management of hypothyroidism and hyperthyroidism. The Research Unit of the Royal College of Physicians of London, the Endocrinology and Diabetes Committee of the Royal College of Physicians of London, and the Society for Endocrinology. BMJ 313, 539–544 (1996).
Baskin, H. J. et al. American Association of Clinical Endocrinologists medical guidelines for clinical practice for the evaluation and treatment of hyperthyroidism and hypothyroidism. Endocr. Pract. 8, 457–469 (2002).
Royal College of Physicians. The diagnosis and management of primary hypothyroidism, revised 14 June 2011 [online], (2011).
Garber, J. R. et al. Clinical practice guidelines for hypothyroidism in adults: cosponsored by the American Association of Clinical Endocrinologists and the American Thyroid Association. Thyroid 22, 1200–1235 (2012).
Brenta, G. et al. Clinical practice guidelines for the management of hypothyroidism. Arq. Bras. Endocrinol. Metabol. 57, 265–291 (2013).
Wiersinga, W. M., Duntas L., Fadeyev, V., Nygaard, B. & Vanderpump, M. P. 2012 ETA guidelines: the use of L-T4 + L-T3 in the treatment of hypothyroidism. Eur. Thyroid J. 1, 55–71 (2012).
Mitchell, A. L., Hickey, B., Hickey, J. L. & Pearce, S. H. Trends in thyroid hormone prescribing and consumption in the UK. BMC Public Health 9, 132 (2009).
de Jong, N. W. & Baljet, G. M. Use of T4, T4 + T3, and T3 in the Dutch population in the period 2005–2011. Eur. Thyroid J. 1, 135–136 (2012).
Ord, W. M. Report of a committee of the Clinical Society of London nominated December 14, 1883, to investigate the subject of myxoedema. Trans. Clin. Soc. Lond. 8 (Suppl.), 21 (1888).
Murray, G. R. Note on the treatment of myxoedema by hypodermic injections of an extract of the thyroid gland of a sheep. Br. Med. J. 2, 796–797 (1891).
Fox, E. L. A case of myxoedema treated by taking extract of thyroid by the mouth. Br. Med. J. 2, 941 (1892).
Kendall, E. C. The isolation in crystalline form of the compound containing iodin, which occurs in the thyroid: its chemical nature and physiological activity. JAMA 64, 2042–2043 (1915).
Harrington, C. R. & Barger, G. Chemistry of thyroxine: constitution and synthesis of thyroxine. Biochem. J. 21, 169–183 (1927).
Sawin, C. S. The heritage of the thyroid: a brief history. In Werner & Ingbar's The Thyroid: a Fundamental and Clinical Text. 10th edn (eds Braverman, L. E. & Cooper, D. S.) 1–4 (Lippincott Williams & Wilkins, 2013).
Gross, J. & Pitt-Rivers, R. V. 3:5:3′-triiodothyronine. I. Isolation from thyroid gland and synthesis. Biochem. J. 53, 645–650 (1953).
Roche, J., Lissitzky, S. & Michel, R. Sur la présence de triiodothyronine dans la thyroglobuline et sur sa biosynthèse [French]. C. R. Hebd. Seances Acad. Sci. 234, 1228–1230 (1952).
MacGregor, A. G. Why does anybody use thyroid B.P.? Lancet 1, 329–332 (1961).
Braverman, L. E., Ingbar, S. H. & Sterling, K. Conversion of thyroxine (T4) to triiodothyronine (T3) in athyreotic human subjects. J. Clin. Invest. 49, 855–864 (1970).
Kaufman, S. C., Gross, T. P. & Kennedy, D. L. Thyroid hormone use: trends in the United States from 1960 through 1988. Thyroid 1, 285–291 (1991).
Bianco, A. C. et al. Biochemistry, cellular and molecular biology and physiological roles of the iodothyronine selenodeiodinases. Endocr. Rev. 23, 38–89 (2002).
Romijn, J. A., Smit, J. W. & Lamberts, S. W. Intrinsic imperfections of endocrine replacement therapy. Eur. J. Endocrinol. 149, 91–97 (2003).
Escobar-Morreale, H. F., Obregón, M. J., Escobar del Rey, F. & Morreale de Escobar, G. Replacement therapy for hypothyroidism with thyroxine alone does not ensure euthyroidism in all tissues, as studied in thyroidectomized rats. J. Clin. Invest. 96, 2828–2838 (1995).
Escobar-Morreale, H. F., Escobar del Rey, F., Obregón, M. J. & Morreale de Escobar, G. Only the combined treatment with thyroxine and triiiodothyronine ensures euthyroidism in all tissues of thyroidectomized rat. Endocrinology 137, 2490–2502 (1996).
Hypo maar niet Happy: belangenvereniging voor mensen met hypothyreoïdie [online], (2013).
Grozinsky-Glasberg, S., Fraser, A., Nahshoni, E., Weizman, A. & Leibovici, L. Thyroxine–triiodothyronine combination therapy versus thyroxine monotherapy for clinical hypothyroidism: meta-analysis of randomized controlled trials. J. Clin. Endocrinol. Metab. 91, 2592–2599 (2006).
Ivkovic, S. Polyneuropathy after radioactive iodine treatment of hyperthyroidism and beneficial effect of combined T4/T3 therapy of hypothyroidism. Eur. Thyroid J. 1, 129–131 (2012).
Biondi, B. & Wartofsky, L. Combination treatment with T4 and T3: toward personalized replacement therapy in hypothyroidism? J. Clin. Endocrinol. Metab. 97, 2256–2271 (2012).
Antonica, F. et al. Generation of functional thyroid from embryonic stem cells. Nature 491, 66–71 (2012).
Ladenson, P. W. Psychological wellbeing in patients. Clin. Endocrinol. (Oxf.) 57, 575–576 (2002).
Saravanan, P. et al. Psychological well-being in patients on 'adequate' doses of L-thyroxine: results of a large, controlled community-based questionnaire study. Clin. Endocrinol. (Oxf.) 57, 577–585 (2002).
Wekking, E. M. et al. Cognitive functioning and well-being in euthyroid patients on thyroxine replacement therapy for primary hypothyroidism. Eur. J. Endocrinol. 153, 747–753 (2005).
Panicker, V. et al. A paradoxical difference in relationship between anxiety, depression and thyroid function in subjects on and not on T4: findings from the HUNT study. Clin. Endocrinol. (Oxf.) 71, 574–580 (2009).
Samuels, M. H., Schuff, K. G., Carlson, N. E., Carello P. & Janowsky, J. S. Health status, psychological symptoms, mood, and cognition in L-thyroxine-treated hypothyroid subjects. Thyroid 17, 249–258 (2007).
Quinque, E. M., Villringer, A., Kratzsch, L. & Karger, S. Patient-reported outcomes in adequately treated hypothyroidism—insights from the German versions of ThyDQoL, ThySRQ and ThyTSQ. Health Qual. Life Outcomes 11, 68 (2013).
Somers, E. C., Thomas, S. L., Smeeth, L. & Hall, A. J. Are individuals with an autoimmune disease at higher risk of a second autoimmune disorder? Am. J. Epidemiol. 169, 749–755 (2009).
Boelaert, K. et al. Prevalence and relative risk of other autoimmune diseases in subjects with autoimmune thyroid disease. Am. J. Med. 123, 183.e1–e9 (2010).
Weetman, A. P. Diseases associated with thyroid autoimmunity: explanations for the expanding spectrum. Clin. Endocrinol. (Oxf.) 74, 411–418 (2011).
Engum, A., Bjøro, T., Mykletun, A. & Dahl, A. A. Thyroid autoimmunity, depression and anxiety: are there any connections? An epidemiological study of a large population. J. Psychosom. Res. 59, 263–268 (2005).
Pop, V. J. et al. Are autoimmune thyroid dysfunction and depression related? J. Clin. Endocrinol. Metab. 83, 3194–3197 (1998).
Watt, T. et al. Is thyroid autoimmunity per se a determinant of quality of life in patients with autoimmune hypothyroidism? Eur. Thyroid J. 1, 186–192 (2012).
Ott, J. et al. Hashimoto's thyroiditis affects symptom load and quality of life unrelated to hypothyroidism: a prospective case–control study in women undergoing thyroidectomy for benign goiter. Thyroid 21, 161–167 (2011).
Saravanan, P., Visser, T. J. & Dayan, C. M. Psychological well-being correlates with free thyroxine but not free 3,5,3′-triiodothyronine levels in patients on thyroid hormone replacement. J. Clin. Endocrinol. Metab. 91, 3389–3393 (2006).
Carr, D., McLeod, D. T., Parry, G. & Thornes, H. M. Fine adjustment of thyroxine replacement dosage: comparison of the thyrotrophin releasing hormone test using a sensitive thyrotrophin assay with measurement of free thyroid hormones and clinical assessment. Clin. Endocrinol. (Oxf.) 28, 325–333 (1988).
Toft, A. D. & Beckett, G. J. Thyroid function tests and hypothyroidism. Measurement of serum TSH alone may not always reflect thyroid status. BMJ 326, 295–296 (2003).
Flynn, R. W. et al. Serum thyroid-stimulating hormone concentration and morbidity from cardiovascular disease and fractures in patients on long-term thyroxine therapy. J. Clin. Endocrinol. Metab. 95, 186–193 (2010).
Walsh, J. P. et al. Small changes in thyroxine dosage do not produce measurable changes in hypothyroid symptoms, well-being, or quality of life: results of a double-blind, randomized clinical trial. J. Clin. Endocrinol. Metab. 91, 2624–2630 (2006).
Woeber, K. A. Levothyroxine therapy and serum free thyroxine and free triiodothyronine concentrations. J. Endocrinol. Invest. 25, 106–109 (2002).
Jonklaas, J., Davidson, B., Bhagat, S. & Soldin, S. J. Triiodothyronine levels in athyreotic individuals during levothyroxine therapy. JAMA 299, 769–777 (2008).
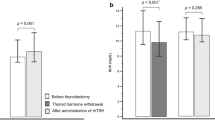
Ito, M. et al. TSH-suppressive doses of levothyroxine are required to achieve preoperative native serum triiodothyronine levels in patients who have undergone total thyroidectomy. Eur. J. Endocrinol. 167, 373–378 (2012).
Fish, L. H. et al. Replacement dose, metabolism, and bioavailability of levothyroxine in the treatment of hypothyroidism. Role of triiodothyronine in pituitary feedback in humans. N. Engl. J. Med. 316, 764–770 (1987).
Gullo, D. et al. Levothyroxine monotherapy cannot guarantee euthyroidism in all athyreotic patients. PLoS ONE 6, e22552 (2011).
Alevizaki, M., Mantzou, E., Cimponeriu, A. T., Alevizaki, C. C. & Koutras, D. A. TSH may not be a good marker for adequate thyroid hormone replacement therapy. Wien. Klin. Wochenschr. 117, 636–640 (2005).
Hoermann, R., Midgley, J. E., Larisch, R. & Dietrich, J. W. Is pituitary TSH an adequate measure of thyroid hormone-controlled homoeostasis during thyroxine treatment? Eur. J. Endocrinol. 168, 271–280 (2013).
Gereben, B. et al. Cellular and molecular basis of deiodinase-regulated thyroid hormone signaling. Endocr. Rev. 29, 898–938 (2008).
Bianco, A. C. & Casula, S. Thyroid hormone replacement therapy: three 'simple' questions, complex answers. Eur. Thyroid J. 1, 88–98 (2012).
Levitt, J. A. & Silverberg J. T4 plus T3 for hypothyroidism: a double-blind comparison with usual T4 [Abstract]. In Proc. 74th annual meeting of the American Thyroid Association (2002).
Saravanan, P., Simmons, D. J., Greenwood, R., Peters, T. J. & Dayan, C. M. Partial substitution of thyroxine (T4) with triiodothyronine in patients on T4 replacement therapy: results of a large community-based randomized controlled trial. J. Clin. Endocrinol. Metab. 90, 805–812 (2005).
Appelhof, B. C. et al. Combined therapy with levothyroxine and liothyronine in two ratios, compared with levothyroxine monotherapy in primary hypothyroidism: a double-blind, randomized, controlled clinical trial. J. Clin. Endocrinol. Metab. 90, 2666–2674 (2005).
Walsh, J. P. et al. Combined thyroxine/liothyronine treatment does not improve well-being, quality of life or cognitive function compared to thyroxine alone: a randomized controlled trial in patients with primary hypothyroidism. J. Clin. Endocrinol. Metab. 88, 4543–4550 (2003).
Clyde, P. W., Harari, A. E., Getka, E. J. & Shakir, K. M. Combined levothyroxine plus liothyronine compared with levothyroxine alone in primary hypothyroidism: a randomized controlled trial. JAMA 290, 2952–2958 (2003).
Sawka, A. M., Gerstein, H. C., Marriott, M. J., MacQueen, G. M. & Joffe, R. T. Does a combination regimen of thyroxine (T4) and 3,5,3′-triiodothyronine improve depressive symptoms better than T4 alone in patients with hypothyroidism? Results of a double-blind, randomized, controlled trial. J. Clin. Endocrinol. Metab. 88, 4551–4555 (2003).
Bunevicius, R., Kazanavicius, G., Zalinkevicius, R. & Prange, A. J. Jr. Effects of thyroxine as compared with thyroxine plus triiodothyronine in patients with hypothyroidism. N. Engl. J. Med. 340, 424–429 (1999).
Rodriguez, T., Lavis, V. R., Meininger, J. C., Kapadia, A. S & Stafford, L. F. Substitution of liothyronine at a 1:5 ratio for a portion of levothyroxine: effect on fatigue, symptoms of depression, and working memory versus treatment with thyroxine alone. Endocr. Pract. 11, 223–233 (2005).
Escobar-Morreale, H. F. et al. Thyroid hormone replacement therapy in primary hypothyroidism: a randomized trial comparing L-thyroxine plus liothyronine with L-thyroxine alone. Ann. Intern. Med. 142, 412–424 (2005).
Siegmund, W. et al. Replacement therapy with levothyroxine plus triiodothyronine (bioavailable molar ratio 14:1) is not superior to thyroxine alone to improve well-being and cognitive performance in hypothyroidism. Clin. Endocrinol. (Oxf.) 60, 750–757 (2004).
Bunevicius, R., Jakubonien, N., Jurkevicius, R., Cernicat, J., Lasas, L. & Prange. A. J. Jr. Thyroxine vs thyroxine plus triiodothyronine in treatment of hypothyroidism after thyroidectomy for Graves' disease. Endocrine 18, 129–133 (2002).
Ma, C. et al. Thyroxine alone or thyroxine plus triiodothyronine replacement therapy for hypothyroidism. Nucl. Med. Commun. 30, 586–593 (2009).
Nygaard, B., Jensen, E. W., Kvetny, J., Jarløv, A. & Faber, J. Effect of combination therapy with thyroxine (T4) and 3,5,3′-triiodothyronine versus T4 monotherapy in patients with hypothyroidism, a double-blind, randomised cross-over study. Eur. J. Endocrinol. 161, 895–902 (2009).
Schmidt, U., Nygaard, B., Jensen, E. W., Kvetny, J., Jarløv, A. & Faber, J. Peripheral markers of thyroid function: the effect of T4 monotherapy vs T4/T3 combination therapy in hypothyroid subjects in a randomized crossover study. Endocr. Connect. 2, 55–60 (2013).
Fadeyev, V. V., Morgunova, T. B., Melnichenko, G. A. & Dedov, I.I. Combined therapy with L-thyroxine and L-triiodothyronine compared to L-thyroxine alone in the treatment of primary hypothyroidism. Hormones (Athens) 9, 245–252 (2010).
Hoang, T. D., Olsen, C. H., Mai, V. Q., Clyde, P. W. & Shakir, M. K. Desiccated thyroid extract compared with levothyroxine in the treatment of hypothyroidism: a randomized, double-blind, crossover study. J. Clin. Endocrinol. Metab. 98, 1982–1990 (2013).
Escobar-Morreale, H. F., Botella-Carretero, J. I., Escobar del Rey, F. & Morreale de Escobar, G. Review: treatment of hypothyroidism with combinations of levothyroxine plus liothyronine. J. Clin. Endocrinol. Metab. 90, 4946–4954 (2005).
Wiersinga, W. M. Do we need still more trials on T4 and T3 combination therapy in hypothyroidism? Eur. J. Endocrinol. 161, 955–959 (2009).
Pilo, A. et al. Thyroidal and peripheral production of 3,5,3′-triiodothyronine in humans by multicompartmental analysis. Am. J. Physiol. 258 (Pt 1), E715–E726 (1990).
Dayan, C. M. & Panicker, V. Novel insights into thyroid hormones from the study of common genetic variation. Nat. Rev. Endocrinol. 5, 211–218 (2009).
Louwerens, M. et al. Fatigue and fatigue-related symptoms in patients treated for different causes of hypothyroidism. Eur. J. Endocrinol. 167, 809–815 (2012).
Arnaud-Lopez, L. et al. Phosphodiesterase 8B gene variants are associated with serum TSH levels and thyroid function. Am. J. Hum. Genet. 82, 1270–1280 (2008).
Shields, B. M. et al. Phosphodiesterase 8B gene polymorphism is associated with subclinical hypothyroidism in pregnancy. J. Clin. Endocrinol. Metab. 94, 4608–4612 (2009).
Medici, M. et al. A large-scale association analysis of 68 thyroid hormone pathway genes with serum TSH and FT4 levels. Eur. J. Endocrinol. 164, 781–788 (2011).
Taylor, P. N. et al. A meta-analysis of the associations between common variation in the PDE8B gene and thyroid hormone parameters, including assessment of longitudinal stability of associations over time and effect of thyroid hormone replacement. Eur. J. Endocrinol. 164, 773–780 (2011).
Peeters, R. P. et al. Polymorphisms in thyroid hormone pathway genes are associated with plasma TSH and iodothyronine levels in healthy subjects. J. Clin. Endocrinol. Metab. 88, 2880–2888 (2003).
Panicker, V. et al. A common variation in deiodinase 1 gene DIO1 is associated with the relative levels of free thyroxine and free triiodothyronine. J. Clin. Endocrinol. Metab. 93, 3075–3081 (2008).
van der Deure, W. M. et al. The effect of genetic variation in the type 1 deiodinase gene on the interindividual variation in serum thyroid hormone levels: an investigation in healthy Danish twins. Clin. Endocrinol. (Oxf.) 70, 954–960 (2009).
Panicker, V. et al. Common variation in the DIO2 gene predicts baseline psychological well-being and response to combination thyroxine plus triiodothyronine therapy in hypothyroid patients. J. Clin. Endocrinol. Metab. 94, 1623–1629 (2009).
Peeters, R. P. et al. A new polymorphism in the type II deiodinase gene is associated with circulating thyroid hormone parameters. Am. J. Physiol. Endocrinol. Metab. 289, E75–E81 (2005).
Torlontano, M. et al. Type 2 deiodinase polymorphism (threonine 92 alanine) predicts L-thyroxine dose to achieve target thyrotropin levels in thyroidectomized patients. J. Clin. Endocrinol. Metab. 93, 910–913 (2008).
Heemstra, K. A. et al. Thr92Ala polymorphism in the type 2 deiodinase is not associated with T4 dose in athyroid patients or patients with Hashimoto thyroiditis. Clin. Endocrinol. (Oxf.) 71, 279–283 (2009).
Kim, B. W. & Bianco, A. C. For some, L-thyroxine replacement might not be enough: a genetic rationale. J. Clin. Endocrinol. Metab. 94, 1521–1523 (2009).
Appelhof, B. C. et al. Polymorphisms in type 2 deiodinase are not associated with well-being, neurocognitive functioning, and preference for combined thyroxine/3,5,3′-triiodothyronine therapy. J. Clin. Endocrinol. Metab. 90, 6296–6299 (2005).
van der Deure, W. M. et al. Polymorphisms in the brain-specific thyroid hormone transporter OATP1C1 are associated with fatigue and depression in hypothyroid patients. Clin. Endocrinol. (Oxf.) 69, 804–811 (2008).
Cooper, D. S. Combined T4 and T3 therapy—back to the drawing board. JAMA 290, 3002–3004 (2003).
Celi, F. S. et al. The pharmacodynamic equivalence of levothyroxine and liothyronine: a randomized, double blind, cross-over study in thyroidectomized patients. Clin. Endocrinol. (Oxf.) 72, 709–715 (2010).
Celi, F. S. et al. Metabolic effects of liothyronine therapy in hypothyroidism: a randomized, double-blind, crossover trial of liothyronine versus levothyroxine. J. Clin. Endocrinol. Metab. 96, 3466–3474 (2011).
Yavuz, S. et al. The dynamic pituitary response to escalating-dose TRH stimulation test in hypothyroid patients treated with liothyronine or levothyroxine replacement therapy. J. Clin. Endocrinol. Metab. 98, E862–E866 (2013).
Russell, W. et al. Free triiodothyronine has a distinct circadian rhythm that is delayed but parallels thyrotropin levels. J. Clin. Endocrinol. Metab. 93, 2300–2306 (2008).
Hennemann, G., Docter, R., Visser, T. J., Postema, P. T. & Krenning, E. P. Thyroxine plus low-dose, slow-release triiodothyronine replacement in hypothyroidism: proof of principle. Thyroid 14, 271–275 (2004).
Kaplan, M. M., Sarne, D. H. & Schneider, A. B. In search of the impossible dream? Thyroid hormone replacement therapy that treats all symptoms in all hypothyroid patients. J. Clin. Endocrinol. Metab. 88, 4540–4542 (2003).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The author declares no competing financial interests.
Rights and permissions
About this article
Cite this article
Wiersinga, W. Paradigm shifts in thyroid hormone replacement therapies for hypothyroidism. Nat Rev Endocrinol 10, 164–174 (2014). https://doi.org/10.1038/nrendo.2013.258
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2013.258
This article is cited by
-
Association of TSHR gene single nucleotide intronic polymorphism with the risk of hypothyroid and hyperthyroid disorders in Yazd province
Scientific Reports (2022)
-
Profiling retrospective thyroid function data in complete thyroidectomy patients to investigate the HPT axis set point (PREDICT-IT)
Journal of Endocrinological Investigation (2021)
-
Correlation between TSH levels and quality of life among subjects with well-controlled primary hypothyroidism
Endocrine (2021)
-
Concurrent TSHR mutations and DIO2 T92A polymorphism result in abnormal thyroid hormone metabolism
Scientific Reports (2018)
-
Genome-wide analyses identify a role for SLC17A4 and AADAT in thyroid hormone regulation
Nature Communications (2018)