Key Points
-
A rise in cytoplasmic Ca2+ concentration acts as a universal intracellular messenger that drives numerous cellular responses.
-
In many cell types, a major route for Ca2+ influx is through store-operated Ca2+ release-activated Ca2+ (CRAC) channels in the plasma membrane. These ion channels are activated by the process of emptying the intracellular Ca2+ stores.
-
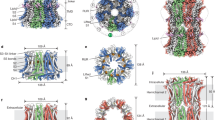
The molecular basis of this ubiquitous Ca2+ entry mechanism has recently been unravelled with the discoveries of stromal interaction molecule 1 (STIM1), a protein that senses the Ca2+ content of the stores, and ORAI1 (also known as CRACM1), which forms the CRAC channel pore.
-
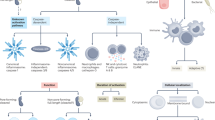
Ca2+ entry through CRAC channels activates various temporally distinct responses, ranging from exocytosis to cell growth and proliferation.
-
Aberrant CRAC channel activity has been linked to several debilitating human diseases, including certain types of immunodeficiency and autoimmunity disorders, allergy, and inflammatory bowel disease.
-
Although in its early stages, drugs targeting the CRAC channel pathway are now being developed and should prove useful in treating a range of immune and non-immune disorders.
Abstract
Elevation of cytosolic Ca2+ levels through the activation of store-operated Ca2+ release-activated Ca2+ (CRAC) channels is involved in mediating a disparate array of cellular responses. These include secretion, metabolism and gene expression, as well as cell growth and proliferation. Moreover, emerging evidence points to the involvement of aberrant CRAC channel activity in human diseases, such as certain types of immunodeficiency and autoimmunity disorders, allergy, and inflammatory bowel disease. This article summarizes recent advances in understanding the gating and function of CRAC channels, their links to human disease and key issues for the development of channel blockers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Berridge, M. J., Bootman, M. D. & Roderick, H. L. Calcium signalling: dynamics, homeostasis and remodelling. Nature Rev. Mol. Cell Biol. 4, 517–529 (2003).
Clapham, D. E. Calcium Signaling. Cell 131, 1047–1058 (2008).
Rizzuto, R. et al. Calcium and apoptosis: facts and hypotheses. Oncogene 22, 8619–8627 (2003).
Neher, E. Vesicle pools and Ca2+ microdomains: new tools for understanding their roles in neurotransmitter release. Neuron 20, 389–399 (1998). This is an authoritative description of how local Ca2+ signals arise and how they can be investigated.
Rizzuto, R. & Pozzan, T. Microdomains of intracellular calcium: molecular determinants and functional consequences. Physiol. Rev. 86, 369–408 (2006). This provides an excellent overview of microdomains and their impact on physiological processes.
Hille, B. Ionic Channels of Excitable Membranes 1–814 (Sinauer Associates, Sunderlan, Massachusetts, 2001).
Berridge, M. J. Inositol trisphosphate and calcium signalling. Nature 361, 315–325 (1993).
Putney, J. W. J. A model for receptor-regulated calcium entry. Cell Calcium 7, 1–12 (1986). This visionary paper proposes the concept of store-operated Ca2+ entry.
Hoth, M. & Penner, R. Depletion of intracellular calcium stores activates a calcium current in mast cells. Nature 355, 353–356 (1992). This is the first description of the store-operated CRAC current.
Zweifach, A. & Lewis, R. S. Mitogen-regulated Ca2+ current of T lymphocytes is activated by depletion of intracellular Ca2+ stores. Proc. Natl Acad. Sci. USA 90, 6295–6299 (1993). This paper shows that CRAC channels have a tiny conductance and are activated by a physiological agonist.
Hoth, M. & Penner, R. Calcium release-activated calcium current in rat mast cells. J. Physiol. 465, 359–386 (1993).
Zweifach, A. & Lewis, R. S. Calcium-dependent potentiation of store-operated calcium channels in T lymphocytes. J. Gen. Physiol. 107, 597–610 (1996).
Hoth, M. Calcium and barium permeation through calcium release-activated (CRAC) channels. Pflugers Arch. 430, 315–322 (1995).
Fierro, L. & Parekh, A. B. Substantial depletion of the intracellular Ca2+ stores is required for macroscopic activation of the Ca2+ release-activated Ca2+ current in rat basophilic leukaemia cells. J. Physiol. 522, 247–257 (2000).
Bakowski, D. & Parekh, A. B. Monovalent cation permeability and Ca2+ block of the store-operated calcium current ICRAC in rat basophilic leukaemia cells. Pflugers Arch. 443, 892–902 (2002).
Bakowski, D. & Parekh, A. B. Permeation through store-operated CRAC channels in divalent-free solution: potential problems and implications for putative CRAC channel genes. Cell Calcium 32, 379–391 (2002).
Prakriya, M. & Lewis, R. S. Regulation of CRAC channel activity by recruitment of silent channels to a high open-probability gating mode. J. Gen. Physiol. 128, 373–386 (2006).
Yeromin, A. V., Roos, J., Stauderman, K. A. & D., C. M. A store-operated calcium channel in Drosophila S2 cells. J. Gen. Physiol. 123, 167–182 (2004).
Lewis, R. S. The molecular choreography of a store-operated calcium channel. Nature 446, 284–287 (2007).
Parekh, A. B. & Putney, J. W. J. Store-operated calcium channels. Physiol. Rev. 85, 757–810 (2005).
Nilius, B., Owsianik, G., Voets, T. & Peters, J. A. Transient receptor potential cation channels in disease. Physiol. Rev. 87, 165–217 (2007).
Clapham, D. E., Runnels, L. W. & Struebing, C. The TRP ion channel family. Nature Rev. Neurosci. 2, 387–396 (2002).
Roos, J. et al. STIM1, an essential and conserved component of store-operated Ca2+ channel function. J. Cell Biol. 169, 435–445 (2005).
Liou, J. et al. STIM is a calcium sensor essential for calcium-store-depletion-triggered calcium infux. Curr. Biol. 15, 1235–1241 (2005). References 23 and 24 identified the importance of STIM1 in store-operated Ca2+ entry.
Cahalan, M. D. STIMulating store-operated Ca2+ entry. Nature Cell Biol. 11, 669–676 (2009).
Dziadek, M. A. & Johnstone, L. S. Biochemical properties and cellular localisation of STIM1 proteins. Cell Calcium 42, 123–132 (2007).
Spassova, M. A., Soboloff, J., He, L.-P., Dziadek, M. A. & Gill, D. L. STIM1 has a plasma membrane role in activation of store-operated calcium channels. Proc. Natl Acad. Sci. USA 103, 4040–4045 (2006).
Mercer, J. C. et al. Large store-operated calcium-selective currents due to co-expression of Orai1 or Orai2 with the intracellular calcium sensor, STIM1. J. Biol. Chem. 281, 24979–24990 (2006).
Wu, M. M., Buchanan, J., Luik, R. M. & Lewis, R. S. Ca2+ store depletion causes STIM1 to accumulate in ER regions closely associated with the plasma membrane. J. Cell Biol. 174, 803–813 (2006).
Stathopulos, P. B., Zheng, L., Li, G.-Y., Plevin, M. J. & Ikura, M. Structural and mechanistic insights into STIM1-mediated initiation of store-operated calcium entry. Cell 110, 110–122 (2008).
Zhang, S. L. et al. STIM1 is a calcium sensor that activates CRAC channels and migrates from the calcium store to the plasma membrane. Nature 437, 902–905 (2005).
Feske, S., Prakriya, M., Rao, A. & Lewis, R. S. A severe defect in CRAC calcium channel activation and altered potassium channel gating in T cells from immunodeficient patients. J. Exp. Med. 202, 651–662 (2005).
Feske, S. et al. A mutation in Orai1 causes immune deficiency by abrogating CRAC channel function. Nature 441, 179–185 (2006).
Vig, M. et al. CRACM1 is a plasma membrane protein essential for store-operated calcium entry. Science 312, 1220–1223 (2006).
Zhang, S. L. et al. Genome-wide RNAi screen of Ca2+ influx identifies genes that regulate Ca2+ release-activated Ca2+ channel activity. Proc. Natl Acad. Sci. USA 103, 9357–9362 (2006). References 33–35 show that ORAI1 is central for CRAC channel function.
Yang, J., Ellinor, P. T., Sather, W. A., Zhang, J. F. & Tsien, R. W. Molecular determinants of Ca2+ selectivity and ion permeation in L-type Ca2+ channels. Nature 366, 158–161 (1993).
Prakriya, M. et al. Orai1 is an essential pore subunit of the CRAC channel. Nature 443, 230–233 (2006).
Vig, M. et al. CRACM1 multimers form the ion-selective pore of the CRAC channel. Curr. Biol. 16, 2073–2079 (2006).
Yeromin, A. V. et al. Molecular identification of the CRAC channel by altered ion selectivity in a mutant of Orai. Nature 443, 226–229 (2006). References 37–39 demonstrated that ORAI1 forms the CRAC channel pore.
Peinelt, C. et al. Amplification of CRAC current by STIM1 and CRACM1 (Orai1). Nature Cell Biol. 8, 771–773 (2006).
Parekh, A. B. A CRAC current tango. Nature Cell Biol. 8, 655–656 (2006).
Soboloff, J. et al. Orai1 and STIM reconstitute store-operated calcium channel function. J. Biol. Chem. 281, 20661–20665 (2006).
Akabas, M. H., Stauffer, D. A., Xu, M. & Karlin, A. Acetylcholine receptor channel structure probed in cysteine-substitution mutants. Science 258, 307–310 (1992).
McNally, B. A., Yamashita, M., Engh, A. & Prakriya, M. Structural determinants of ion permeation in CRAC channels. Proc. Natl Acad. Sci. USA 106, 22516–22521 (2009).
Long, S. B., Tao, X., Campbell, E. B. & MacKinnon, R. Atomic structure of a voltage-dependent K+ channel in a lipid membrane-like environment. Nature 450, 376–382 (2007).
Gwack, Y. et al. Biochemical and functional characterization of Orai proteins. J. Biol. Chem. 282, 16232–16243 (2007).
Lis, A. et al. CRACM1, CRACM2 and CRACM3 are store-operated Ca2+ channels with distinct functional properties. Curr. Biol. 17, 794–800 (2007).
DeHaven, W. I., Smyth, J. T., Boyles, R. R. & Putney, J. W. J. Calcium inhibition and calcium potentiation of Orai1, Orai2 and Orai3 calcium release-activated calcium channels. J. Biol. Chem. 282, 17548–17556 (2007).
Gross, S. A. et al. Murine Orai2 splice variants form functional Ca2+ release-activated Ca2+ (CRAC) channels. J. Biol. Chem. 282, 19375–19384 (2007).
Parvez, S. et al. STIM2 protein mediates distinct store-dependent and store-independent modes of CRAC channel activation. FASEB J. 22, 752–761 (2008).
Brandman, O., Liou, J., Park, W. S. & Meyer, T. STIM2 is a feedback regulator that stabilizes basal cytosolic and endoplasmic reticulum calcium levels. Cell 131, 1327–1339 (2007).
Bird, G. S. et al. STIM1 is a calcium sensor specialised for digital signalling. Curr. Biol. 19, 1724–1729 (2009).
Liou, J., Fivaz, M., Inoue, T. & Meyer, T. Live-cell imaging reveals sequential oligomerization and local plasma membrane targeting of stromal interaction molecule 1 after calcium store depletion. Proc. Natl Acad. Sci. USA 104, 9301–9306 (2007).
Varnai, P., Toth, B., Toth, D. J., Hunyady, L. & Balla, T. Visualization and manipulation of plasma membrane–endoplasmic reticulum contact sites indicates the presence of additional molecular components within the STIM1–Orai1 complex. J. Biol. Chem. 282, 29678–29689 (2007).
Luik, R. M., Wang, B., Prakriya, M., Wu, M. M. & Lewis, R. S. Oligomerization of STIM1 couples ER calcium depletion to CRAC channel activation. Nature 454, 538–542 (2008).
Park, C. Y. et al. STIM1 clusters and activates CRAC channels via direct binding of a cytosolic domain of Orai1. Cell 136, 1–15 (2009).
Xu, P. et al. Aggregation of STIM1 underneath the plasma membrane induces clustering of Orai1. Biochem. Biophys. Res. Comm. 350, 969–976 (2006).
Muik, M. et al. Cytosolic homomerization and a modulatory domain within STIM1 C terminus determine coupling to ORAI1 channels. J. Biol. Chem. 284, 8421–8426. (2009).
Yuan, J. P. et al. SOAR and the polybasic STIM1 domains gate and regulate Orai channels. Nature Cell Biol. 11, 337–343 (2009).
Kawasaki, T., Lange, I. & Feske, S. A minimal regulatory domain in the C terminus of STIM1 binds to and activates ORAi1 CRAC channels. Biochem. Biophys. Res. Comm. 385, 49–54 (2009). References 56 and 58–60 provide a molecular mechanism for CRAC channel gating.
Penna, A. et al. The CRAC channel consists of a tetramer formed by STIM-induced dimerization of Orai dimers. Nature 456, 116–120 (2008).
Mignen, O., Thompson, J. L. & Shuttleworth, T. J. Orai1 subunit stoichiometry of the mammalian CRAC channel pore. J. Physiol. 586, 419–425 (2008).
Ji, W. et al. Functional stoichiometry of the unitary calcium release-activated calcium channel. Proc. Natl Acad. Sci. USA 105, 13668–13673 (2008).
Carafoli, E. Calcium signalling: a tale for all seasons. Proc. Natl Acad. Sci. USA 99, 1115–1122 (2002).
Michalak, M., Robert Parker, J. M. & Opas, M. Ca2+ signalling and calcium binding chaperones of the endoplasmic reticulum. Cell Calcium 32, 269–278 (2002).
Parekh, A. B. Functional consequences of activating store-operated CRAC channels. Cell Calcium 42, 111–121 (2007).
Parekh, A. B. Ca2+ microdomains near plasma membrane Ca2+ channels: impact on cell function. J. Physiol. 586, 3043–3054 (2008).
Gwack, Y., Feske, S., Srikanth, S., Hogan, P. G. & Rao, A. Signalling to transcription: store-operated Ca2+ entry and NFAT activation in lymphocytes. Cell Calcium 42, 145–156 (2007).
Dolmetsch, R. E., Xu, K. & Lewis, R. S. Calcium oscillations increase the efficiency and specificity of gene expression. Nature 392, 933–936 (1998).
Mogami, H., Nakano, K., Tepikin, A. V. & Petersen, O. H. Ca2+ flow via tunnels in polarized cells: recharging of apical Ca2+ stores by focal Ca2+ entry through basal membrane patch. Cell 88, 49–55 (1997).
Bautista, D. M. & Lewis, R. S. Modulation of plasma membrane calcium-ATPase activity by local calcium microdomains near CRAC channels in human T cells. J. Physiol. 556, 805–817 (2004).
Willoughby, D. & Cooper, D. M. F. Organization and Ca2+ regulation of adenylyl cyclases in cAMP microdomains. Physiol. Rev. 87, 965–1010 (2007).
Chang, W.-C., Nelson, C. & Parekh, A. B. Ca2+ influx through CRAC channels activates cytosolic phospholipase A2, leukotriene C4 secretion and expression of c-fos through ERK-dependent and independent pathways in mast cells. FASEB J. 20, E1681–E1693 (2006).
Peters-Golden, M., Gleason, M. M. & Togias, A. Cysteinyl leukotrienes: multi-functional mediators in allergic rhinitis. Clin. Exp. Allergy 36, 689–703 (2006).
Steinke, J. W. & Borish, L. Leukotriene receptors in rhinitis and sinusitis. Curr. Allergy Asthma Rep. 4, 217–223 (2004).
Chang, W. C. & Parekh, A. B. Close functional coupling between CRAC channels, arachidonic acid release and leukotriene secretion. J. Biol. Chem. 279, 29994–29999 (2004).
Chang, W. C. et al. Local calcium influx through calcium release-activated calcium (CRAC) channels stimulates production of an intracellular messenger and an intercellular pro-inflammatory signal. J. Biol. Chem. 283, 4622–4631 (2008).
DiCapite, J. L., Shirley, A., Nelson, C., Bates, G. & Parekh, A. B. Intercellular calcium wave propagation involving positive feedback between CRAC channels and cysteinyl leukotrienes. FASEB J. 23, 894–905 (2009).
Gallo, E. M., Cante-Barrett, K. & Crabtree, G. R. Lymphocyte calcium signaling from membrane to nucleus. Nature Immunol. 7, 25–32 (2006).
Ng, S.-W., Nelson, C. & Parekh, A. B. Coupling of Ca2+ microdomains to spatially and temporally distinct cellular responses by the tyrosine kinase Syk. J. Biol. Chem. 284, 24767–24772 (2009).
Di Capite, J., Ng, S.-W. & Parekh, A. B. Decoding of cytoplasmic Ca2+ oscillations through the spatial signature drives gene expression. Curr. Biol. 19, 853–858 (2009). This paper shows that local Ca2+ entry through CRAC channels and not cytosolic Ca2+ oscillations drives gene expression.
Vig, M. et al. Defective mast cell effector functions in mice lacking the CRACM1 pore subunit of store-operated calcium release-activated calcium channels. Nature Immunol. 9, 89–96 (2008).
Baba, Y., Nishida, K., Fujii, Y., Hikida, M. & Kurosaki, T. Essential function for the calcium sensor STIM1 in mast cell activation and anaphylatic responses. Nature Immunol. 9, 81–88 (2008). References 82 and 83 show that CRAC channels are essential for mast-cell function.
Gwack, Y. et al. Hair loss and defective T- and B-cell function in mice lacking ORAI1. Mol. Cell. Biol. 28, 5209–5222 (2008). A detailed characterization of the ORAI1 -knockout mouse.
Stiber, J. et al. STIM1 signalling controls store-operated calcium entry required for development and contractile function in skeletal muscle. Nature Cell Biol. 10, 688–697 (2008).
Varga-Szabo, D. et al. The calcium sensor STIM1 is an essential mediator of arterial thrombosis and ischemic brain infarction. J. Exp. Med. 205, 1583–1591 (2008).
Partiseti, M. et al. The calcium current activated by T cell receptor and store depletion in human lymphocytes is absent in a primary immunodeficiency. J. Biol. Chem. 269, 32327–32335 (1994).
Le Deist, F. et al. A primary T-cell immunodeficiency associated with defective transmembrane calcium influx. Blood 85, 1053–1062 (1995).
Feske, S. et al. Severe combined immunodeficiency due to defective binding of nuclear factor of activated T cells in T lymphocytes of two male siblings. Eur. J. Immunol. 26, 2119–2126 (1996).
Feske, S. Orai1 and STIM1 deficiency in human and mice: roles of store-operated calcium entry in immune cells and beyond. Immunol. Rev. 231, 189–209 (2009).
McCarl, C.-A. et al. Orai1 deficiency and lack of store-operated Ca2+ entry cause immunodeficiency, myopathy, and ectodermal dysplasia. J. Allergy Clin. Immunol. 124, 1311–1318 (2009).
Picard, C. et al. STIM1 mutation associated with a syndrome of immunodeficiency and autoimmunity. N. Engl. J. Med. 360, 1971–1980 (2009). This paper describes mutations in STIM1 that are linked to immunodeficiencies.
Radenne, F. et al. Quality of life in nasal polyposis. J. Allergy Clin. Immunol. 103, 79–84 (1999).
Bateman, N. D., Fahy, C. & Woolford, T. J. Nasal polyps: still more questions than answers. J. Larynol. Otol. 117, 1–9 (2003).
Pawankar, R. Nasal polyposis: an update. Curr. Opin. Allergy Clin. Immunol. 3, 1–6 (2003).
Di Capite, J., Nelson, C., Bates, G. & Parekh, A. B. Targeting Ca2+ release-activated Ca2+ channel channels and leukotriene receptors provides a novel combination strategy for treating nasal polyposis. J. Allergy Clin. Immunol. 124, 1014–1021 (2009).
Ishikawa, J. et al. A pyrazole derivative, YM-58483, potently inhibits store-operated sustained calcium influx and IL-2 production in T lymphocytes. J. Immunol. 170, 4441–4449 (2003).
Ohga, K. et al. The suppressive effects of YM-58483/BTP2, a store-oeprated Ca2+ entry blocker, on inflammatory mediator release in vitro and airway responses in vivo. Pulm. Pharmacol. Ther. 21, 360–369 (2008).
Yoshino, T. et al. YM-58483, a selective CRAC channel inhibitor prevents antigen-induced airway eosinophilia and late phase asthmatic responses via Th2 cytokine inhibition in animal models. Eur. J. Pharmacol. 560, 225–233 (2007).
Peel, S. E., Liu, B. & Hall, I. P. Orai and store-operated calcium influx in human airway smooth muscle cells. Am. J. Resp. Cell Mol. Biol. 38, 744–749 (2008).
Pirzer, U., Schoenhaar, A., Fleischer, B., Hermann, E. & Meyer zum Bueschenfelde, K. H. Reactivity of infiltrating T lymphocytes with microbial antigens in Crohn's disease. Lancet 338, 1238–1239 (1991).
Fiocchi, C. Inflammatory bowel disease:etilogy and pathogenesis. Gastroenterology 115, 182–205 (1998).
Schwarz, A. et al. Ca2+ signalling in identified T-lymphocytes from human intestinal mucosa. J. Biol. Chem. 279, 5641–5647 (2004).
Ng, S.-W., DiCapite, J. L., Singaravelu, K. & Parekh, A. B. Sustained activation of the tyrosine kinase Syk by antigen in mast cells requires local Ca2+ influx through Ca2+ release-activated Ca2+ channels. J. Biol. Chem. 283, 31348–31355 (2008).
Di Sabatino, A. et al. Targeting gut T cell Ca2+ release-activated Ca2+ channels inhibits T cell cytokine production and T-Box transcription factor T-Bet in inflammatory bowel disease. J. Immunol. 183, 3454–3462 (2009).
Yang, S., Zhang, J. J. & Huang, X. Y. Orai1 and STIM1 are critical for breast tumor cell migration and metastasis. Cancer Cell 15, 124–134 (2009). This study implicates a major role for CRAC channels in the development of breast cancer.
Vanden Abeele, F. et al. Bcl-2-dependent modulation of Ca2+ homeostasis and store-operated channels in prostate cancer cells. Cancer Cell 1, 169–179 (2002).
Prevarskaya, N., Skryma, R., Bidaux, G., Flourakis, M. & Shuba, Y. Ion channels in death and differentiation of prostate cancer cells. Cell Death Differ. 14, 1295–1304 (2007).
Franzius, D., Hoth, M. & Penner, R. Non-specific effects of calcium entry antagonists in mast cells. Pflugers Arch. 428, 433–438 (1994).
Putney, J. W. Pharmacology of capacitative calcium entry. Mol. Interv. 1, 84–94 (2001).
Djuric, S. W. et al. 3,5-Bis(trifluoromethyl)pyrazoles: a novel class of NFAT transcription factor regulator. J. Med. Chem. 43, 2975–2981 (2000).
Trevillyan, J. M. et al. Potent inhibition of NFAT activation and T cell cytokine production by novel low molecular weight pyrazole compounds. J. Biol. Chem. 276, 48118–48126 (2001).
Zitt, C. et al. Potent inhibition of Ca2+ release-activated Ca2+ channels and T-lymphocyte activation by the pyrazole derivative BTP2. J. Biol. Chem. 279, 12427–12437 (2004).
Takezawa, R. et al. A pyrazole derivative potently inhibits lymphocyte Ca2+ influx and cytokine production by facilitating transient receptor potential melastatin 4 channel activity. Mol. Pharmacol. 69, 1413–1420 (2006).
He, L. P., Hewavitharana, T., Soboloff, J., Spassova, M. A. & Gill, D. L. Functional link between store-operated and TRPC channels revealed by the 3,5-bis (trifluoromethyl)pyrazole derivative, BTP2. J. Biol. Chem. 280, 10997–11006 (2005).
Zakharov, S. I. et al. Diethylstilbestrol is a potent inhibitor of store-operated channels and capacitative Ca2+ influx. Mol. Pharmacol. 66, 702–707 (2004).
Ohana, L., Stanley, E. F. & Schlichter, L. C. The Ca2+ release-activated Ca2+ current (ICRAC) mediates store-operated Ca2+ entry in rat microglia. Channels 3, 129–139 (2009).
Rodland, K. D., Wersto, R. P., Hobson, S. & Kohn, E. C. Thapsigargin-induced gene expression in nonexcitable cells is dependent on calcium influx. Mol. Endocrinol. 11, 281–291 (1997).
Kohn, E. C. et al. A phase 1 trial of carboxyamido-triazole and paclitaxel for relapsed solid tumours: potential efficacy of the combination and demonstration of pharmacokinetic interaction. Clin. Cancer Res. 7, 1600–1609 (2001).
Hussain, M. M. et al. Phase II trial of carboxyamidotriazole in patients with relapsed epithelial ovarian cancer. J. Clin. Oncol. 21, 4356–4363 (2003).
Mignen, O. et al. Carboxyamidotriazole-induced inhibition of mitochondrial calcium import blocks capacitative calcium entry and cell proliferation in HEK-293 cells. J. Cell Sci. 118, 5615–5623 (2005).
Glitsch, M. D., Bakowski, D. & Parekh, A. B. Store-operated Ca2+ entry depends on mitochondrial Ca2+ uptake. EMBO J. 21, 6744–6754 (2002).
Gilabert, J.-A. & Parekh, A. B. Respiring mitochondria determine the pattern of activation and inactivation of the store-operated Ca2+ current ICRAC . EMBO J. 19, 6401–6407 (2000).
Hoth, M., Button, D. & Lewis, R. S. Mitochondrial control of calcium channel gating: a mechanism for sustained signaling and transcriptional activation in T lymphocytes. Proc. Natl Acad. Sci. USA 97, 10607–10612 (2000).
Parekh, A. B. Mitochondrial regulation of store-operated CRAC channels. Cell Calcium 44, 6–13 (2008).
DeHaven, W. I., Smyth, J. T., Boyles, R. R., Bird, G. S. & Putney, J. W. Complex actions of 2-aminoethyldiphenyl borate (2-APB) on store-operated calcium entry. J. Biol. Chem. 283, 19265–19273 (2008).
Peinelt, C., Lis, A., Beck, A., Fleig, A. & Penner, R. 2-aminoethoxydiphenyl borate directly facilitates and indirectly inhibits STIM1-dependent gating of CRAC channels. J. Physiol. 586, 3061–3073 (2008).
Zhang, S. L. et al. Store-dependent and independent modes of regulating Ca2+ release-activated Ca2+ channel activity of human Orai1 and Orai3. J. Biol. Chem. 283, 17662–17671 (2008).
Schindl, R. et al. 2-aminoethoxydiphenyl borate alters selectivity of Orai3 channels by increasing their pore size. J. Biol. Chem. 283, 20261–20267 (2008).
Navarro-Borelly, L. et al. STIM1–Orai1 interactions and Orai1 conformational changes revealed by live-cell FRET microscopy. J. Physiol. 586, 5383–5401 (2008).
Lefkimmiatis, K. et al. Store-operated cyclic AMP signalling mediated by STIM1. Nature Cell Biol. 11, 433–442 (2009).
Oh-Hora, M. et al. Dual functions for the endoplasmic reticulum calcium sensors STIM1 and STIM2 in T cell activation and tolerance. Nature Immunol. 9, 432–443 (2008).
Acknowledgements
Work in my laboratory is supported by the Medical Research Council and the British Heart Foundation. I thank D. Bakowski for comments on the manuscript.
Author information
Authors and Affiliations
Ethics declarations
Competing interests
The author declares no competing financial interests.
Related links
Glossary
- SERCA
-
Sarco/endoplasmic reticulum Ca2+ ATPase pump, which transports Ca2+ from the cytosol into the endoplasmic reticulum.
- Fluctuation analysis
-
A method that extracts information concerning the nature of individual channel events from statistical descriptions of the current noise.
- Transient receptor potential (TRP) ion channel family
-
A broad family of largely non-selective cation channels permeable to Na+ and Ca2+. TRP channels were first discovered in the trp mutant strain of Drosophila melanogaster.
- EF-hand domain
-
A highly conserved Ca2+ binding site found in many Ca2+-binding proteins.
- Sterile α-motif domain
-
A putative protein–protein interaction module that helps drive homo-oligomerization and hetero-oligomerization.
- Ezrin-radixin-moesin
-
A group of actin-binding proteins that link actin with plasma membrane proteins and are involved in signal transduction.
- ICRAC
-
The current that flows through Ca2+ release-activated Ca2+ (CRAC) channels.
- Muscular hypotonia
-
Decreased muscle tone.
- Ectodermal dysplasia
-
Abnormalities of ectodermal structures including hair, nails, sweat glands and teeth. These abnormalities can lead to reduced sweat production and reduced dental enamel formation.
- Puncta
-
Hot-spots or clusters of stromal interaction molecule 1 (STIM1) at endoplasmic reticulum–plasma membrane regions just below the plasma membrane.
Rights and permissions
About this article
Cite this article
Parekh, A. Store-operated CRAC channels: function in health and disease. Nat Rev Drug Discov 9, 399–410 (2010). https://doi.org/10.1038/nrd3136
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd3136
This article is cited by
-
Investigation of nanotopography on SOCE mediated cell migration via live-cell Imaging on opaque implant surface
Journal of Nanobiotechnology (2023)
-
Cholesterylation of Smoothened is a calcium-accelerated autoreaction involving an intramolecular ester intermediate
Cell Research (2022)
-
Microglia Modulate Cortical Spreading Depolarizations After Ischemic Stroke: A Narrative Review
Neurocritical Care (2022)
-
The roles of transmembrane family proteins in the regulation of store-operated Ca2+ entry
Cellular and Molecular Life Sciences (2022)
-
Direct control of store-operated calcium channels by ultrafast laser
Cell Research (2021)