Key Points
-
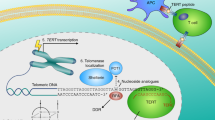
Telomeres are the linear ends of chromosomes and are important in completing DNA replication. Telomeres and telomere-associated proteins cap the chromosome ends, preventing recombination, end-to-end fusions and degradation. Telomeres progressively shorten throughout life and most pre-cancerous lesions have very short telomeres.
-
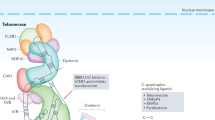
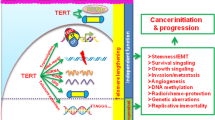
Immortalization is required for most advanced human malignancies to continue to divide. The cellular reverse transcriptase telomerase is upregulated in almost all cancers and permits the indefinite growth of human cancer cells.
-
Approaches that specifically interfere with telomerase represent a novel targeted cancer therapy forcing cells back into a senescent or apoptotic pathway.
-
Clinical trials targeting telomerase include vaccines that recognize telomerase epitopes inappropriately expressed on tumour cells and small-molecule inhibitors such as a 13-mer oligonucleotide called GRN163L
-
Other approaches in preclinical studies include a telomerase-specific oncolytic virus that exclusively replicates in and lyses cells that actively express telomerase.
-
Most cancers are heterogeneous and contain cancer stem cells as well as more differentiated cancer cells. It is now known that telomerase is expressed in at least some types of cancer stem cells and that they contain short telomeres. Therefore, telomerase inhibitors could also target cancer stem cells.
Abstract
It has been approximately a decade since telomerase was described as an almost universal marker for human cancer. Most human tumours not only express telomerase but also have very short telomeres, whereas telomerase activity is either reduced or absent in normal tissues, making the inhibition of telomerase an attractive target for cancer therapeutics. Here we review the current status of telomerase therapeutics and discuss future opportunities and challenges for telomerase research, including a possible relationship with cancer stem cells that could be a source of chemo-/radioresistance development in many advanced cancers.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Moyzis, R. K. et al. A highly conserved repetitive DNA sequence (TTAGGG)n, present at the telomeres of human chromosomes. Proc. Natl Acad. Sci. USA 85, 6622–6626 (1988).
Blackburn, E. H. Telomere states and cell fates. Nature 408, 53–56 (2000).
de Lange, T. Protection of mammalian telomeres. Oncogene 21, 532–540 (2002).
Griffith, J. D. et al. Mammalian telomeres end in a large duplex loop. Cell 97, 503–514 (1999). Provides the first direct evidence that telomeres form a duplex loop instead of ending in a linear fashion.
Blackburn, E. H. Switching and signaling at the telomere. Cell 106, 661–673 (2001).
Wright, W. E., Pereira-Smith, O. M. & Shay, J. W. Reversible cellular senescence: a two-stage model for the immortalization of normal human diploid fibroblasts. Mol. Cell. Biol. 9, 3088–3092 (1989). The first report defining two stages of cellular senescence in human cells.
Feng, J. et al. The RNA component of human telomerase. Science 269, 1236–1241 (1995). Reports the initial cloning of hTR, the functional or template RNA subunit of human telomerase.
Nakamura, T. M. et al. Telomerase catalytic subunit homologs from fission yeast and humans. Science 277, 955–959 (1997). Reports the initial cloning of hTERT, the catalytic subunit of human telomerase.
Lingner, J. & Cech, T. R. Telomerase and chromosome end maintenance. Curr. Opin. Genet. Dev. 8, 226–232 (1998).
Collins, K. & Mitchell, J. R. Telomerase in the human organism. Oncogene 21, 564–579 (2002).
Nugent, C. I. & Lundblad, V. The telomerase reverse transcriptase: components and regulation. Genes. Dev. 12, 1073–1085 (1998).
Wright, W. E., Piatyszek, M. A., Rainey, W. E., Byrd, W. & Shay, J. W. Telomerase activity in human germline and embryonic tissues and cells. Dev. Genet. 18, 173–117 (1996). The first report to demonstrate telomerase activity in human germline and embryonic tissues, and its repression during development.
Aisner, D. L., Wright, W. E. & Shay, J. W. Telomerase regulation: not just flipping the switch. Curr. Opin. Genet. Dev. 12, 80–85 (2002).
Forsyth, N. R, Wright. W. E. & Shay, J. W. Telomerase and differentiation in multicellular organisms:Turn it off, turn it on, and turn it off again. Differentiation 69, 188–197 (2002).
Bodnar, A. G. et al. Extension of life-span by introduction of telomerase into normal human cells. Science 279, 349–352 (1998). First report that the introduction of hTERT is sufficient to produce telomerase activity, maintain or elongate telomeres, and immortalize normal diploid human cells.
Shay, J. W. & Roninson, I. B. Hallmarks of senescence in carcinogenesis and cancer therapy Oncogene 23, 2919–2933 (2004).
Shay, J. W. & Wright, W. E. Senescence and immortalization: role of telomeres and telomerase. Carcinogenesis 25, 1–8 (2004).
Shay, J. W. & Wright, W. E. Hayflick, his limit, and cellular ageing. Nature Rev. Mol. Cell Biol. 1, 72–76 (2000).
Shay, J. W. & Wright, W. E. Telomeres in dyskeratosis congenita. Nature Genet. 36, 437–438 (2004).
Hanahan, D. & Weinberg, R. A. The hallmarks of cancer. Cell 100, 57–70 (2000).
Greider, C. W. & Blackburn, E. H. Identification of a specific telomere terminal transferase activity in Tetrahymena extracts. Cell 43, 405–413 (1985). First paper to identify the existence of telomerase activity approximately 10 years before the main components, TERT and TR, were cloned.
Kim, N. W. et al. Specific association of human telomerase activity with immortal cells and cancer. Science 266, 2011–2015 (1994). First report to describe TRAP (PCR-based telomerase activity assay) and to demonstrate telomerase activity in a large panel of primary human cancers but not in normal human tissues.
Shay, J. W. & Bacchetti, S. A survey of telomerase activity in human cancer. Eur. J. Cancer 5, 787–791 (1997). Provides an overview of the diagnostic potential of measuring telomerase activity in human cancer.
Shay, J. W. Telomerase in cancer: diagnostic, prognostic, and therapeutic implications. Cancer J. Sci. Am. (Suppl. 1), S26–S34 (1998).
Hodes, R. Molecular targeting of cancer: telomeres as targets. Proc. Natl Acad. Sci. USA 98, 7649–7651 (2001).
Saretzki, G. Telomerase inhibition as cancer therapy. Cancer Letter. 194, 209–219 (2003).
Kelland, L. R. Telomerase: biology and phase I trials. Lancet Oncol. 2, 95–102 (2001).
White, L. K., Wright, W. E. & Shay, J. W. Telomerase inhibitors. Trends Biotechnol. 19, 114–120 (2001).
Shay, J. W. & Wright, W. E. Mechanism-based combination telomerase inhibition therapy. Cancer Cell. 7, 1–2 (2005).
Neidle, S. & Parkinson, G. Telomere maintenance as a target for anticancer drug discovery. Nature Rev. Drug Discov. 1, 383–393 (2002).
Helder, M. N., Wisman, G. B. A. & van der Zee, A. G. J. Telomerase and telomeres: from basic biology to cancer treatment. Cancer Invest. 20, 82–101 (2002).
Blasco, M. A. Telomeres and human disease: ageing, cancer and beyond. Nature Rev. Genet. 6, 611–622 (2005).
McKenzie, K. E., Umbricht C. B. & Sukumar S. Applications of telomerase research in the fight against cancer. Mol. Med. Today 5, 114–122 (1999).
Shay, J. W. Meeting Report: The role of telomeres and telomerase in cancer. Cancer Res. 65, 3513–3517 (2005).
Gellert, G. C., Jackson, S. R. Dikmen, G., Wright, W. E. & Shay, J. W. Telomerase as a therapeutic target in cancer. Drug Discov. Today 2, 159–164 (2005).
Shay, J. W. & Wright W. E. in Telomeres 2nd Edn (Eds. deLange, T., Lundblad, V. Blackburn, E.) 81–108 (Cold Spring Harbor Laboratory, New York, 2005).
Granger M. P., Wright W. E. & Shay J. W. Telomerase in cancer and aging. Crit. Rev. Oncol. Hematol. 4, 29–40 (2002).
Shay, J. W. & Gazdar, A. F. Telomerase in the early detection of cancer. J. Clin. Pathol. 50, 106–109 (1997).
Gilley, D., Tanaka, H. & Herbert, B.-S. Telomere dysfunction in aging and cancer. Intl J. Biochem. Cell Biol. 37, 1000–1013 (2005).
Gowan, S. M. et al. A G-quadruplex-interactive potent small-molecule inhibitor of telomerase exhibiting in vitro and in vivo antitumor activity. Mol. Pharmacol. 61, 1154–1162 (2002).
Read, M. A. et al. Molecular modeling studies on G-quadruplex complexes of telomerase inhibitors: structure-activity relationships. J. Med. Chem. 42, 4538–4546 (1999).
Riou, J. F. et al. Cell senescence and telomere shortening induced by a new series of specific G-quadruplex DNA ligands. Proc. Natl Acad. Sci. USA 99, 2672–2677 (2002).
Kim, M. M. et al. A low threshold level of expression of mutant-template telomerase RNA inhibits human tumor cell proliferation. Proc. Natl Acad. Sci. USA 98, 7982–7987 (2001).
Minev, B. et al. Cytotoxic T cell immunity against telomerase reverse transcriptase in humans. Proc. Natl Acad. Sci. USA 97, 4796–4801 (2000).
Nair, S. K. et al. Induction of cytotoxic T lymphocyte responses and tumor immunity against unrelated tumors using telomerase reverse transcriptase RNA transfected dendritic cells. Nature Med. 6, 1011–1017 (2000).
Vonderheide, R. H. et al. Vaccination of cancer patients against telomerase induces functional anti-tumor CD8+ T lymphocytes. Clin. Cancer Res. 10, 828–839 (2004). First intradermal immunotherapy trial directed against telomerase in patients with breast cancer resistant to conventional cytotoxic therapy or progressive hormone-independent prostate cancer.
Vonderheide, R. H. Telomerase as a universal tumor-associated antigen for cancer immunotherapy. Oncogene 21, 674–679 (2002).
Brunsvig, P. F. et al. Telomerase peptide vaccination: a Phase I/II study in patients with non-small cell lung cancer. Cancer Immunol. Immunother. 21 Feb 2006 [epub ahead of print].
Su, Z. et al. Telomerase mRNA-tranfected dendritic cells stimulate antigen-specific CD8+ and CD4+ cell responses in patients with metastatic prostate cancer. J. Immunol. 174, 3798–3807 (2005). First immunotherapy trial directed against telomerase (hTERT) mRNA-transfected dendritic cells in patients with metastatic prostate cancer.
Chen, Z., Koeneman, K. S. & Corey, D. R. Consequences of telomerase inhibition and combination treatments for the proliferation of cancer cells. Cancer Res. 63, 5917–5925 (2003).
Gaudernack, G. et al. Clinical trials of a peptide vaccine targeting telomerase. ASCO Annu. Mtg A666 (2003).
Corey, D. R. Telomerase: an unusual target for cytotoxic agents. Chem. Res. Toxicol. 13, 957–960 (2000).
Dikmen, Z. G. et al. In vivo inhibition of lung cancer by GRN163L — a novel human telomerase inhibitor. Cancer Res. 65, 7866–7873 (2005). First demonstration that the telomerase inhibitor GRN163L prevents lung metastasis in a xenograft animal model.
Gellert, G. C., Dikmen, Z. G., Wright, W. E., Gryaznov, S. & Shay, J. W. Effects of a novel telomerase inhibitor, GRN163L, in human breast cancer. Breast Cancer Res. Treatment (in the press).
Djojosubroto, M. W. et al. Telomerase antagonist GRN163 and GRN163L inhibit tumor growth and increase chemosensitivity of human hepatoma. (in press, Hepatology, 2006)
Gryaznov, S. et al. Telomerase inhibitors – oligonucleotide phosphoramidates as potential therapeutic agents. Nucleosides Nucleotides Nucleic Acids 20, 401–410 (2001).
Herbert, B.-S. et al. Inhibition of human telomerase in immortal human cells leads to progressive telomere shortening and cell death. Proc. Natl Acad. Sci. USA 96, 14276–14281 (1999).
Herbert, B-S., Pongracz, K. Shay, J. W. & Gryaznov, S. M. Oligonucleotide N3′-P5′ phosphoramidates as efficient telomerase inhibitors Oncogene 21, 638–642 (2002).
Asai, A. et al. A novel telomerase template antagonist (GRN163) as a potential anticancer agent. Cancer Res. 63, 3931–3939 (2003).
Gu, J. et al. Tumor-specific transgene expression from the human telomerase reverse transcriptase promoter enables targeting of the therapeutic effects of the Bax gene to cancers. Cancer Res. 60, 5359–5364 (2001).
Gu, J., Andreff, M., Roth, J. A. & Fang, B. hTERT promoter induces tumor-specific Bax gene expression and cell killing in syngenic mouse tumor model and prevents systemic toxicity. Gene Ther. 9, 30–37 (2002).
Koga, S. et al. A novel telomerase-specific gene therapy: gene transfer of caspase-8 utilizing the human telomerase catalytic subunit gene promoter. Hum. Gene Ther. 11, 1397–1406 (2000).
Koga, S. et al. FADD gene therapy using the human telomerase catalytic subunit (hTERT) gene promoter to restrict induction of apoptosis to tumors in vitro and in vivo. Anticancer Res 21, 1937–1943 (2001).
Komata, T. et al. Treatment of malignant glioma cells with the transfer of constitutively active caspase-6 using the human telomerase catalytic subunit (human telomerase reverse transcriptase) gene promoter. Cancer Res. 61, 5796–5802 (2001).
Majumdar, A. S. et al. The telomerase reverse transcriptase promoter drives efficacious tumor suicide gene therapy while preventing hepatotoxicity encountered with constitutive promoters. Gene Ther. 8, 568–578 (2001).
Abdul-Ghani, R. et al. Use of transcriptional regulatory sequences of telomerase (hTER and hTERT) for selective killing of cancer cells. Mol. Ther. 2, 539–544 (2000).
Plumb, J. A. et al. Telomerase-specific suicide gene therapy vectors expressing bacterial nitroreductase sensitize human cancer cells to the pro-drug CB1954. Oncogene 20, 7797–7803 (2001).
Bilsland, A. E., Fletcher-Monaghan, A. & Keith, W. N. Properties of a telomerase-specific cre/lox switch for transcriptionally targeted cancer gene therapy. Neoplasia 10, 1–10 (2006).
Reya, T., Morrison, S. J., Clarke, M. F. & Weissman, I. L. Stem cells, cancer, and cancer stem cells. Nature 414, 105–111 (2001).
Goodell, M. A. et al. Dye efflux studies suggest that hematopoietic stem cells expressing low or undetectable levels of CD34 antigen exist in multiple species. Nature Med. 3, 1337–1345 (1997).
Wang, J. C. Y. & Dick, J. E. Cancer stem cells: lessons from leukemia. Trends Cell Biol. 1, 494–501 (2005).
Bjerkvig, R., Tysnes, B. B., Aboody, K. S., Najbauer, J. & Terzis, A. J. A. The origin of the cancer stem cells: current controversies and new insights. Nature Rev. Genet. 5, 899–904 (2005).
Welm, B. E. et al. Sca-1(pos) cells in the mouse mammary gland represent an enriched progenitor cell population. Dev. Biol. 245, 42–56 (2002).
Clayton, H., Titley, I. & Vivanco, M. Growth and differentiation of progenitor/stem cells derived from the human mammary gland. Exp. Cell Res. 297, 444–460 (2004).
Al-Hajj, M., Wicha, M. S., Benito-Hernandez, A., Morrison, S. J. & Clarke, M. F. Prospective identification of tumorigenic breast cancer cells. Proc. Natl Acad. Sci. USA 100, 3983–3988 (2003).
Ponit, D. et al. Isolation and in vitro propagation of tumorigenic breast cancer cells with stem/progenitor cell properties. Cancer Res. 65, 5506–5511 (2005). First paper to demonstrate telomerase activity in cancer stem cells
Campbell, L. J. et al. hTERT, the catalytic component of telomerase, is downregulated in the haematopoietic stem cells of patients with chronic myeloid leukaemia. Leukemia 20, 671–679 (2006).
Wang, J. C. Y. et al. Dissociation of telomerase activity and telomere length maintenance in primitive human hematopoietic cells. Proc. Natl Acad. Sci. USA 102, 14398–14403 (2005).
Shay, J. W., Pereira-Smith, O. M. & Wright, W. E. A role for both Rb and p53 in the regulation of human cellular senescence. Exp. Cell Res. 196, 33–39 (1991).
Shay, J. W., Wright, W. E. & Werbin, H. Defining the molecular mechanisms of human cell immortalization. Biochim. Biophys. Acta 1072, 1–7 (1991).
Wright, W. E. & Shay, J. W. Cellular senescence as a tumor-protection mechanism: the essential role of counting. Curr. Opin. Genet. Dev. 11, 98–103 (2001).
Zou, Y., Sfeir, A., Shay, J. W. & Wright, W. E. Does a sentinel or groups of short telomeres determine replicative senescence? Mol. Biol. Cell. 15, 3709–3718 (2004).
Goytisolo, F. A & Blasco, M. A. Many ways to telomere dysfunction: in vivo studies using mouse models. Oncogene 21, 584–591(2002).
Maser, R. S. & DePinho, R. A. Connecting chromosomes, crisis and cancer. Science 297, 565–569 (2002).
Feldser, D. M., Hackett, J. A. & Greider, C. W. Telomere dysfunction and the initiation of genome instability. Nature Rev. Cancer 3, 1–5 (2003).
Dokal, I. & Vulliamy T. in Telomeres 2nd Edn (Eds. deLange, T., Lundblad, V. Blackburn, E.) 139–161 (Cold Spring Harbor Laboratory, New York, 2005).
Gomez, D. et al. Telomerase downregulation induced by the G-quadruplex ligand 12459 in A549 cells is mediated by hTERT RNA alternative splicing. Nucl. Acids Res. 31, 371–379 (2004).
Cristofari, G. & Lingner, J. in Telomeres 2nd Edn (Eds. deLange, T., Lundblad, V. Blackburn, E.) 21–47 (Cold Spring Harbor Laboratory, New York, 2005).
Chen, J-L. & Greider, C. W. in Telomeres 2nd Edn (Eds. deLange, T., Lundblad, V. Blackburn, E.) 49–79 (Cold Spring Harbor Laboratory, New York, 2005).
Acknowledgements
The authors acknowledge support from the Southland Foundation Distinguished Chair in Geriatrics Research, the Ellison Medical Foundation, and NSCOR and National Cancer Institute grants. We also acknowledge A. Diehl for providing drafts of the figures used in this review.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Both authors are scientific advisory board members of the Geron Corp.
Related links
Glossary
- Replicative senescence
-
The process by which most normal human cells 'count' the number of times they have divided, eventually undergoing an irreversible growth arrest due to telomere shortening on a few chromosome ends.
- Mitotic catastrophe
-
A response to abnormal mitotic DNA damage, leading to cell death. Normal cells avoid mitotic catastrophe by activating different cell-cycle checkpoint genes, which allows cells to repair the damage before mitosis; this mechanism is absent in checkpoint-deficient cells with critically shortened telomeres.
- Telomerase reverse transcriptase
-
(hTERT). The catalytic subunit of telomerase (an RNA-dependent DNA polymerase) that synthesizes telomeric repeats onto the end of telomeres using the integral RNA (hTR) component as a template.
- Telomerase RNA
-
(hTR/hTERC). The integral RNA that provides an 11-bp template complementary to the telomeric repeats to be added to the chromosome.
- Crisis
-
A balance between cell growth and cell death. When cells bypass replicative senescence, telomeres continue to shorten, eventually leading to mitotic catastrophe. These cells die or, rarely, reactivate telomerase, leading to an immortalized cell line.
- Cancer stem cells
-
A small subset of tumour cells that can recreate and sustain (re-initiate, re-populate) the tumour in a functional transplant assay. It is believed that cancer stem cells have multilineage potential and might be responsible for the failure of current therapies.
- Prostate-specific antigen
-
A serine protease in the kallikrein gene family that is secreted into seminal fluid by prostatic epithelial cells and found in the serum. As it is almost exclusively a product of prostate cells, measurement in blood has proved to be exceptionally useful as a tumour marker for diagnosis of prostate cancer and monitoring the effectiveness of treatment.
- Xenograft
-
Transplantation of tissue or cells from one species to another. In cancer research, most xenografts are human cancer cell lines or human tumours that have been transplanted to immune-deficient rodents.
Rights and permissions
About this article
Cite this article
Shay, J., Wright, W. Telomerase therapeutics for cancer: challenges and new directions. Nat Rev Drug Discov 5, 577–584 (2006). https://doi.org/10.1038/nrd2081
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd2081
This article is cited by
-
Hyperpolarized δ-[1- 13C]gluconolactone imaging visualizes response to TERT or GABPB1 targeting therapy for glioblastoma
Scientific Reports (2023)
-
DNA–dependent protein kinase in telomere maintenance and protection
Cellular & Molecular Biology Letters (2020)
-
Analysis of candidate genes expected to be essential for melanoma surviving
Cancer Cell International (2020)
-
The mTOR pathway is necessary for survival of mice with short telomeres
Nature Communications (2020)
-
Telomeres and telomerase: three decades of progress
Nature Reviews Genetics (2019)