Key Points
-
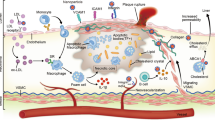
Nanomedicine — the use of nanotechnology for medical purposes — is a fast-growing field that has had numerous applications in cancer and is now increasingly being considered for atherosclerosis, the major cause of cardiovascular disease.
-
Several methods of targeting atherosclerotic plaques using nanoparticles have been explored, which can be classified into nonspecific targeting, specific targeting of the vasculature as well as specific targeting of plaque components either with synthetic nanoparticles or via interaction through a natural conduit.
-
Nanoparticle-facilitated therapeutics can potentially be applied to target the liver and change lipid levels systemically, or they can consist of direct injections of high-density lipoprotein nanoparticles to enhance the transport of cholesterol in plaques to the liver for excretion. Nanotherapy can also be used to intervene in specific atherosclerotic plaque processes by depleting monocyte recruitment or decreasing plaque inflammation and neovascularization.
-
Silencing of plaque inflammation after a clinical event — such as a myocardial infarction or stroke — using nanotherapy is a promising therapeutic paradigm as it may improve the effectiveness of subsequent (statin) therapy.
-
Medical imaging is increasingly being applied to assess atherosclerosis and to evaluate the effects of therapeutic interventions. It allows the direct and non-invasive visualization of plaque build-up and plaque characteristics in animal models as well as in patients.
-
Nanoparticle-enhanced diagnostics allow the molecular imaging of atherosclerotic plaques by several clinically available imaging modalities, including magnetic resonance imaging, computed tomography, positron emission tomography and ultrasound imaging. Multimodal nanoparticles have exhibited valuable roles in the evaluation and corroboration of targeting mechanisms.
Abstract
The use of nanotechnology for medical purposes — nanomedicine — has grown exponentially over the past few decades. This is exemplified by the US Food and Drug Administration's approval of several nanotherapies for various conditions, as well as the funding of nanomedical programmes worldwide. Although originally the domain of anticancer therapy, recent advances have illustrated the considerable potential of nanomedicine in the diagnosis and treatment of atherosclerosis. This Review elaborates on nanoparticle-targeting concepts in atherosclerotic disease, provides an overview of the use of nanomedicine in atherosclerosis, and discusses potential future applications and clinical benefits.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Change history
31 October 2011
The following sentence was inadvertently duplicated on page 835: Subsequently, several other nanomedicinal therapeutics have been approved for clinical use, including an albumin-bound nanoparticle delivering paclitaxel for the treatment of breast cancer (Abraxane; Abraxis BioScience) and liposomal amphotericin B for the treatment of fungal infections (AmBisome; Astellas Pharma). This has been removed from this both the html and pdf versions.
References
Moghimi, S. M., Hunter, A. C. & Murray, J. C. Nanomedicine: current status and future prospects. FASEB J. 19, 311–330 (2005).
Wagner, V., Dullaart, A., Bock, A. K. & Zweck, A. The emerging nanomedicine landscape. Nature Biotech. 24, 1211–1217 (2006).
Kim, B. Y., Rutka, J. T. & Chan, W. C. Nanomedicine. N. Engl. J. Med. 363, 2434–2443 (2010).
Nagarajan, R. & Hatton, T. A. (eds) Nanoparticles: Synthesis, Stabilization, Passivation, and Functionalization 2–14 (American Chemical Society, Washington DC, 2008).
Mulder, W. J. et al. Nanoparticulate assemblies of amphiphiles and diagnostically active materials for multimodality imaging. Acc. Chem. Res. 42, 904–914 (2009).
Mitragotri, S. & Lahann, J. Physical approaches to biomaterial design. Nature Mater. 8, 15–23 (2009). This is a detailed review about the influence of biomaterial design on the in vivo behaviour of the biomaterials.
Zhang, L. et al. Nanoparticles in medicine: therapeutic applications and developments. Clin. Pharmacol. Ther. 83, 761–769 (2008).
Farokhzad, O. C. & Langer, R. Impact of nanotechnology on drug delivery. ACS Nano 3, 16–20 (2009).
Davis, M. E., Chen, Z. G. & Shin, D. M. Nanoparticle therapeutics: an emerging treatment modality for cancer. Nature Rev. Drug Discov. 7, 771–782 (2008).
Allen, T. M. Long-circulating (sterically stabilized) liposomes for targeted drug delivery. Trends Pharmacol. Sci. 15, 215–220 (1994).
van Dalen, E. C., Michiels, E. M., Caron, H. N. & Kremer, L. C. Different anthracycline derivates for reducing cardiotoxicity in cancer patients. Cochrane Database Syst. Rev. CD005006 (2010).
Terreno, E., Castelli, D. D., Viale, A. & Aime, S. Challenges for molecular magnetic resonance imaging. Chem. Rev. 110, 3019–3042 (2010).
Yu, S. B. & Watson, A. D. Metal-based X-ray contrast media. Chem. Rev. 99, 2353–2378 (1999).
Chen, K. & Conti, P. S. Target-specific delivery of peptide-based probes for PET imaging. Adv. Drug Deliv. Rev. 62, 1005–1022 (2010).
Kobayashi, H., Ogawa, M., Alford, R., Choyke, P. L. & Urano, Y. New strategies for fluorescent probe design in medical diagnostic imaging. Chem. Rev. 110, 2620–2640 (2010).
Allen, T. M. & Cullis, P. R. Drug delivery systems: entering the mainstream. Science 303, 1818–1822 (2004).
Ford, E. S. et al. Explaining the decrease in U.S. deaths from coronary disease, 1980–2000. N. Engl. J. Med. 356, 2388–2398 (2007).
Roger, V. L. et al. Heart disease and stroke statistics —2011 update: a report from the American Heart Association. Circulation 123, e18–e209 (2011).
Naghavi, M. et al. From vulnerable plaque to vulnerable patient: a call for new definitions and risk assessment strategies: Part I. Circulation 108, 1664–1672 (2003).
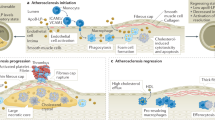
Falk, E. Pathogenesis of atherosclerosis. J. Am. Coll. Cardiol. 47, C7–C12 (2006). This is a comprehensive review on the pathogenesis of atherosclerosis.
Chiu, J. J. & Chien, S. Effects of disturbed flow on vascular endothelium: pathophysiological basis and clinical perspectives. Physiol. Rev. 91, 327–387 (2011).
Tabas, I., Williams, K. J. & Boren, J. Subendothelial lipoprotein retention as the initiating process in atherosclerosis: update and therapeutic implications. Circulation 116, 1832–1844 (2007).
Tabas, I. Macrophage death and defective inflammation resolution in atherosclerosis. Nature Rev. Immunol. 10, 36–46 (2010).
Moreno, P. R., Purushothaman, K. R., Sirol, M., Levy, A. P. & Fuster, V. Neovascularization in human atherosclerosis. Circulation 113, 2245–2252 (2006).
Moreno, P. R., Sanz, J. & Fuster, V. Promoting mechanisms of vascular health: circulating progenitor cells, angiogenesis, and reverse cholesterol transport. J. Am. Coll. Cardiol. 53, 2315–2323 (2009).
Kolodgie, F. D. et al. Elimination of neoangiogenesis for plaque stabilization: is there a role for local drug therapy? J. Am. Coll. Cardiol. 49, 2093–2101 (2007).
Libby, P. Inflammation in atherosclerosis. Nature 420, 868–874 (2002).
Hansson, G. K. & Libby, P. The immune response in atherosclerosis: a double-edged sword. Nature Rev. Immunol. 6, 508–519 (2006).
Falk, E., Shah, P. K. & Fuster, V. Coronary plaque disruption. Circulation 92, 657–671 (1995).
Coussens, L. M. & Werb, Z. Inflammation and cancer. Nature 420, 860–867 (2002).
Mantovani, A., Allavena, P., Sica, A. & Balkwill, F. Cancer-related inflammation. Nature 454, 436–444 (2008).
Ferrari, M. Cancer nanotechnology: opportunities and challenges. Nature Rev. Cancer 5, 161–171 (2005).
Peer, D. et al. Nanocarriers as an emerging platform for cancer therapy. Nature Nanotechnol. 2, 751–760 (2007).
Matsumura, Y. & Maeda, H. A new concept for macromolecular therapeutics in cancer chemotherapy: mechanism of tumoritropic accumulation of proteins and the antitumor agent smancs. Cancer Res. 46, 6387–6392 (1986).
Fang, J., Nakamura, H. & Maeda, H. The EPR effect: unique features of tumor blood vessels for drug delivery, factors involved, and limitations and augmentation of the effect. Adv. Drug Deliv. Rev. 63, 136–151 (2011).
Tarbell, J. M. Shear stress and the endothelial transport barrier. Cardiovasc. Res. 87, 320–330 (2010).
van Bochove, G. S. et al. Contrast enhancement by differently sized paramagnetic MRI contrast agents in mice with two phenotypes of atherosclerotic plaque. Contrast Media Mol. Imaging 6, 35–45 (2011).
Flogel, U. et al. In vivo monitoring of inflammation after cardiac and cerebral ischemia by fluorine magnetic resonance imaging. Circulation 118, 140–148 (2008).
Torchilin, V. P. Multifunctional nanocarriers. Adv. Drug Deliv. Rev. 58, 1532–1555 (2006).
Hansson, G. K. Inflammation, atherosclerosis, and coronary artery disease. N. Engl. J. Med. 352, 1685–1695 (2005).
Jiang, W., Kim, B. Y., Rutka, J. T. & Chan, W. C. Nanoparticle-mediated cellular response is size-dependent. Nature Nanotechnol. 3, 145–150 (2008).
O'Neil, C. P. et al. Extracellular matrix binding mixed micelles for drug delivery applications. J. Control Release 137, 146–151 (2009).
Briley-Saebo, K. C. et al. Targeted iron oxide particles for in vivo magnetic resonance detection of atherosclerotic lesions with antibodies directed to oxidation-specific epitopes. J. Am. Coll. Cardiol. 57, 337–347 (2011).
Kirpotin, D. B. et al. Antibody targeting of long-circulating lipidic nanoparticles does not increase tumor localization but does increase internalization in animal models. Cancer Res. 66, 6732–6740 (2006). This study demonstrates that ligand-directed targeting can increase cell internalization.
Bartlett, D. W., Su, H., Hildebrandt, I. J., Weber, W. A. & Davis, M. E. Impact of tumor-specific targeting on the biodistribution and efficacy of siRNA nanoparticles measured by multimodality in vivo imaging. Proc. Natl Acad. Sci. USA 104, 15549–15554 (2007).
Lanza, G. M. et al. Angiogenesis imaging with vascular-constrained particles: the why and how. Eur. J. Nucl. Med. Mol. Imaging 37 (Suppl. 1), 114–126 (2010).
Nahrendorf, M. et al. Noninvasive vascular cell adhesion molecule-1 imaging identifies inflammatory activation of cells in atherosclerosis. Circulation 114, 1504–1511 (2006). This is an example of a multimodal nanoparticle being used to detect plaque inflammation via target-specific imaging of adhesion molecules.
Libby, P. The forgotten majority: unfinished business in cardiovascular risk reduction. J. Am. Coll. Cardiol. 46, 1225–1228 (2005).
Miller, G. J. & Miller, N. E. Plasma-high-density-lipoprotein concentration and development of ischaemic heart-disease. Lancet 1, 16–19 (1975).
Gordon, D. J. & Rifkind, B. M. High-density lipoprotein — the clinical implications of recent studies. N. Engl. J. Med. 321, 1311–1316 (1989).
Navab, M., Reddy, S. T., Van Lenten, B. J. & Fogelman, A. M. HDL and cardiovascular disease: atherogenic and atheroprotective mechanisms. Nature Rev. Cardiol. 8, 222–232 (2011).
Khera, A. V. et al. Cholesterol efflux capacity, high-density lipoprotein function, and atherosclerosis. N. Engl. J. Med. 364, 127–135 (2011).
Briel, M. et al. Association between change in high density lipoprotein cholesterol and cardiovascular disease morbidity and mortality: systematic review and meta-regression analysis. BMJ 338, b92 (2009).
Degoma, E. M. & Rader, D. J. Novel HDL-directed pharmacotherapeutic strategies. Nature Rev. Cardiol. 8, 266–277 (2011).
Badimon, J. J., Badimon, L., Galvez, A., Dische, R. & Fuster, V. High density lipoprotein plasma fractions inhibit aortic fatty streaks in cholesterol-fed rabbits. Lab. Invest. 60, 455–461 (1989).
Badimon, J. J., Badimon, L. & Fuster, V. Regression of atherosclerotic lesions by high density lipoprotein plasma fraction in the cholesterol-fed rabbit. J. Clin. Invest. 85, 1234–1241 (1990).
Shah, P. K. et al. Effects of recombinant apolipoprotein A-IMilano on aortic atherosclerosis in apolipoprotein E-deficient mice. Circulation 97, 780–785 (1998).
Shah, P. K. et al. High-dose recombinant apolipoprotein A-IMilano mobilizes tissue cholesterol and rapidly reduces plaque lipid and macrophage content in apolipoprotein E-deficient mice. Potential implications for acute plaque stabilization. Circulation 103, 3047–3050 (2001).
Chiesa, G. et al. Recombinant apolipoprotein A-IMilano infusion into rabbit carotid artery rapidly removes lipid from fatty streaks. Circ. Res. 90, 974–980 (2002).
Ibanez, B. et al. Rapid change in plaque size, composition, and molecular footprint after recombinant apolipoprotein A-IMilano (ETC-216) administration: magnetic resonance imaging study in an experimental model of atherosclerosis. J. Am. Coll. Cardiol. 51, 1104–1109 (2008).
Kaul, S. et al. Rapid reversal of endothelial dysfunction in hypercholesterolemic apolipoprotein E-null mice by recombinant apolipoprotein A-IMilano-phospholipid complex. J. Am. Coll. Cardiol. 44, 1311–1319 (2004).
Nissen, S. E. et al. Effect of recombinant ApoA-I Milano on coronary atherosclerosis in patients with acute coronary syndromes: a randomized controlled trial. JAMA 290, 2292–2300 (2003). This article presents the results of a clinical trial with infusions of reconstituted HDL that demonstrated regression of coronary atherosclerosis as measured by intravascular ultrasound imaging.
Nicholls, S. J. et al. Relationship between atheroma regression and change in lumen size after infusion of apolipoprotein A-I Milano. J. Am. Coll. Cardiol. 47, 992–997 (2006).
Bloedon, L. T. et al. Safety, pharmacokinetics, and pharmacodynamics of oral apoA-I mimetic peptide D-4F in high-risk cardiovascular patients. J. Lipid Res. 49, 1344–1352 (2008).
Navab, M. et al. Structure and function of HDL mimetics. Arterioscler. Thromb. Vasc. Biol. 30, 164–168 (2010).
Vickers, K. C., Palmisano, B. T., Shoucri, B. M., Shamburek, R. D. & Remaley, A. T. MicroRNAs are transported in plasma and delivered to recipient cells by high-density lipoproteins. Nature Cell Biol. 13, 423–433 (2011).
Rensen, P. C. et al. Recombinant lipoproteins: lipoprotein-like lipid particles for drug targeting. Adv. Drug Deliv. Rev. 47, 251–276 (2001).
Yang, M. et al. Efficient cytosolic delivery of siRNA using HDL-mimicking nanoparticles. Small 7, 568–573 (2011).
McMahon, K. M. et al. Biomimetic high density lipoprotein nanoparticles for nucleic acid delivery. Nano Lett. 11, 1208–1214 (2011).
Skajaa, T. et al. High-density lipoprotein-based contrast agents for multimodal imaging of atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 30, 169–176 (2010).
Thaxton, C. S., Daniel, W. L., Giljohann, D. A., Thomas, A. D. & Mirkin, C. A. Templated spherical high density lipoprotein nanoparticles. J. Am. Chem. Soc. 131, 1384–1385 (2009).
Skajaa, T. et al. The biological properties of iron oxide core high-density lipoprotein in experimental atherosclerosis. Biomaterials 32, 206–213 (2011).
Zheng, G., Chen, J., Li, H. & Glickson, J. D. Rerouting lipoprotein nanoparticles to selected alternate receptors for the targeted delivery of cancer diagnostic and therapeutic agents. Proc. Natl Acad. Sci. USA 102, 17757–17762 (2005).
Cannon, C. P., Steinberg, B. A., Murphy, S. A., Mega, J. L. & Braunwald, E. Meta-analysis of cardiovascular outcomes trials comparing intensive versus moderate statin therapy. J. Am. Coll. Cardiol. 48, 438–445 (2006).
Armitage, J. et al. Intensive lowering of LDL cholesterol with 80 mg versus 20 mg simvastatin daily in 12,064 survivors of myocardial infarction: a double-blind randomised trial. Lancet 376, 1658–1669 (2010).
Zimmermann, T. S. et al. RNAi-mediated gene silencing in non-human primates. Nature 441, 111–114 (2006). This study demonstrates the potential of systemic lipid-lowering by liposome-encapsulated siRNA.
Semple, S. C. et al. Rational design of cationic lipids for siRNA delivery. Nature Biotech. 28, 172–176 (2010).
Love, K. T. et al. Lipid-like materials for low-dose, in vivo gene silencing. Proc. Natl Acad. Sci. USA 107, 1864–1869 (2010).
Libby, P., Ridker, P. M. & Hansson, G. K. Inflammation in atherosclerosis: from pathophysiology to practice. J. Am. Coll. Cardiol. 54, 2129–2138 (2009).
Charo, I. F. & Taub, R. Anti-inflammatory therapeutics for the treatment of atherosclerosis. Nature Rev. Drug Discov. 10, 365–376 (2011).
Poon, M. et al. Dexamethasone inhibits macrophage accumulation after balloon arterial injury in cholesterol fed rabbits. Atherosclerosis 155, 371–380 (2001).
Lobatto, M. E. et al. Multimodal clinical imaging to longitudinally assess a nanomedical anti-inflammatory treatment in experimental atherosclerosis. Mol. Pharm. 7, 2020–2029 (2010). This study shows the ability of non-invasive imaging to longitudinally monitor therapeutic response in a rabbit model of atherosclerosis after nanomedical treatment.
Mauriello, A. et al. Diffuse and active inflammation occurs in both vulnerable and stable plaques of the entire coronary tree: a histopathologic study of patients dying of acute myocardial infarction. J. Am. Coll. Cardiol. 45, 1585–1593 (2005).
McCarthy, J. R., Korngold, E., Weissleder, R. & Jaffer, F. A. A light-activated theranostic nanoagent for targeted macrophage ablation in inflammatory atherosclerosis. Small 6, 2041–2049 (2010).
Dangas, G. D. et al. In-stent restenosis in the drug-eluting stent era. J. Am. Coll. Cardiol. 56, 1897–1907 (2010).
Pendyala, L. K. et al. The first-generation drug-eluting stents and coronary endothelial dysfunction. JACC Cardiovasc. Interv. 2, 1169–1177 (2009).
Luscher, T. F. et al. Drug-eluting stent and coronary thrombosis: biological mechanisms and clinical implications. Circulation 115, 1051–1058 (2007).
Nakazawa, G. et al. The pathology of neoatherosclerosis in human coronary implants: bare-metal and drug-eluting stents. J. Am. Coll. Cardiol. 57, 1314–1322 (2011).
Danenberg, H. D. et al. Macrophage depletion by clodronate-containing liposomes reduces neointimal formation after balloon injury in rats and rabbits. Circulation 106, 599–605 (2002).
Danenberg, H. D. et al. Liposomal alendronate inhibits systemic innate immunity and reduces in-stent neointimal hyperplasia in rabbits. Circulation 108, 2798–2804 (2003).
Joner, M. et al. Site-specific targeting of nanoparticle prednisolone reduces in-stent restenosis in a rabbit model of established atheroma. Arterioscler. Thromb. Vasc. Biol. 28, 1960–1966 (2008).
Ribichini, F. et al. Immunosuppressive therapy with oral prednisone to prevent restenosis after PCI. A multicenter randomized trial. Am. J. Med. 124, 434–443 (2011).
Kolodgie, F. D. et al. Sustained reduction of in-stent neointimal growth with the use of a novel systemic nanoparticle paclitaxel. Circulation 106, 1195–1198 (2002).
Uwatoku, T. et al. Application of nanoparticle technology for the prevention of restenosis after balloon injury in rats. Circ. Res. 92, e62–e69 (2003).
Abid, M. R. et al. Forkhead transcription factors inhibit vascular smooth muscle cell proliferation and neointimal hyperplasia. J. Biol. Chem. 280, 29864–29873 (2005).
Li, J. M. et al. Local arterial nanoparticle delivery of siRNA for NOX2 knockdown to prevent restenosis in an atherosclerotic rat model. Gene Ther. 17, 1279–1287 (2010).
Lanza, G. M. et al. Targeted antiproliferative drug delivery to vascular smooth muscle cells with a magnetic resonance imaging nanoparticle contrast agent: implications for rational therapy of restenosis. Circulation 106, 2842–2847 (2002).
Cyrus, T. et al. Intramural delivery of rapamycin with αvβ3-targeted paramagnetic nanoparticles inhibits stenosis after balloon injury. Arterioscler. Thromb. Vasc. Biol. 28, 820–826 (2008).
Chan, J. M. et al. Spatiotemporal controlled delivery of nanoparticles to injured vasculature. Proc. Natl Acad. Sci. USA 107, 2213–2218 (2010).
Kaul, S. et al. Intramural delivery of recombinant apolipoprotein A-IMilano/phospholipid complex (ETC-216) inhibits in-stent stenosis in porcine coronary arteries. Circulation 107, 2551–2554 (2003).
Nakano, K. et al. Formulation of nanoparticle-eluting stents by a cationic electrodeposition coating technology: efficient nano-drug delivery via bioabsorbable polymeric nanoparticle-eluting stents in porcine coronary arteries. JACC Cardiovasc. Interv. 2, 277–283 (2009).
Polyak, B. et al. High field gradient targeting of magnetic nanoparticle-loaded endothelial cells to the surfaces of steel stents. Proc. Natl Acad. Sci. USA 105, 698–703 (2008).
Chorny, M. et al. Targeting stents with local delivery of paclitaxel-loaded magnetic nanoparticles using uniform fields. Proc. Natl Acad. Sci. USA 107, 8346–8351 (2010).
Jain, R. K., Finn, A. V., Kolodgie, F. D., Gold, H. K. & Virmani, R. Antiangiogenic therapy for normalization of atherosclerotic plaque vasculature: a potential strategy for plaque stabilization. Nature Clin. Pract. Cardiovasc. Med. 4, 491–502 (2007).
Moulton, K. S. et al. Angiogenesis inhibitors endostatin or TNP-470 reduce intimal neovascularization and plaque growth in apolipoprotein E-deficient mice. Circulation 99, 1726–1732 (1999).
Moulton, K. S. et al. Inhibition of plaque neovascularization reduces macrophage accumulation and progression of advanced atherosclerosis. Proc. Natl Acad. Sci. USA 100, 4736–4741 (2003).
Winter, P. M. et al. Endothelial αvβ3 integrin-targeted fumagillin nanoparticles inhibit angiogenesis in atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 26, 2103–2109 (2006). This was the first study to show the therapeutic potential of combined molecular imaging and drug delivery with targeted nanoparticles in a rabbit model of atherosclerosis.
Winter, P. M. et al. Antiangiogenic synergism of integrin-targeted fumagillin nanoparticles and atorvastatin in atherosclerosis. JACC Cardiovasc. Imaging 1, 624–634 (2008).
Hedman, M. et al. Safety and feasibility of catheter-based local intracoronary vascular endothelial growth factor gene transfer in the prevention of postangioplasty and in-stent restenosis and in the treatment of chronic myocardial ischemia: Phase II results of the Kuopio Angiogenesis Trial (KAT). Circulation 107, 2677–2683 (2003).
Yla-Herttuala, S., Rissanen, T. T., Vajanto, I. & Hartikainen, J. Vascular endothelial growth factors: biology and current status of clinical applications in cardiovascular medicine. J. Am. Coll. Cardiol. 49, 1015–1026 (2007).
Kim, J., Cao, L., Shvartsman, D., Silva, E. A. & Mooney, D. J. Targeted delivery of nanoparticles to ischemic muscle for imaging and therapeutic angiogenesis. Nano Lett. 11, 694–700 (2011).
Celletti, F. L. et al. Vascular endothelial growth factor enhances atherosclerotic plaque progression. Nature Med. 7, 425–429 (2001).
Peters, D. et al. Targeting atherosclerosis by using modular, multifunctional micelles. Proc. Natl Acad. Sci. USA 106, 9815–9819 (2009).
Myerson, J., He, L., Lanza, G., Tollefsen, D. & Wickline, S. Thrombin-inhibiting perfluorocarbon nanoparticles provide a novel strategy for the treatment and magnetic resonance imaging of acute thrombosis. J. Thromb. Haemost. 9, 1292–1300 (2011).
Uesugi, Y., Kawata, H., Jo, J., Saito, Y. & Tabata, Y. An ultrasound-responsive nano delivery system of tissue-type plasminogen activator for thrombolytic therapy. J. Control Release 147, 269–277 (2010).
Sanz, J. & Fayad, Z. A. Imaging of atherosclerotic cardiovascular disease. Nature 451, 953–957 (2008). This is a review on the different methods of imaging atherosclerotic cardiovascular disease.
Weissleder, R. & Mahmood, U. Molecular imaging. Radiology 219, 316–333 (2001).
Chen, I. Y. & Wu, J. C. Cardiovascular molecular imaging: focus on clinical translation. Circulation 123, 425–443 (2011).
Leuschner, F. & Nahrendorf, M. Molecular imaging of coronary atherosclerosis and myocardial infarction: considerations for the bench and perspectives for the clinic. Circ. Res. 108, 593–606 (2011). This is a review on the processes that can be targeted to enable the detection of coronary atherosclerosis by molecular imaging.
Buono, C., Anzinger, J. J., Amar, M. & Kruth, H. S. Fluorescent pegylated nanoparticles demonstrate fluid-phase pinocytosis by macrophages in mouse atherosclerotic lesions. J. Clin. Invest. 119, 1373–1381 (2009).
Louie, A. Multimodality imaging probes: design and challenges. Chem. Rev. 110, 3146–3195 (2010). This is an extensive review on the synthesis and design of multimodal imaging probes.
Mulder, W. J. et al. Magnetic and fluorescent nanoparticles for multimodality imaging. Nanomedicine (Lond.) 2, 307–324 (2007).
Underhill, H. R., Hatsukami, T. S., Fayad, Z. A., Fuster, V. & Yuan, C. MRI of carotid atherosclerosis: clinical implications and future directions. Nature Rev. Cardiol. 7, 165–173 (2010).
Voros, S. et al. Coronary atherosclerosis imaging by Coronary CT angiography: current status, correlation with intravascular interrogation and meta-analysis. JACC Cardiovasc. Imaging 4, 537–548 (2011).
Sirol, M. et al. Lipid-rich atherosclerotic plaques detected by gadofluorine-enhanced in vivo magnetic resonance imaging. Circulation 109, 2890–2896 (2004).
Ruehm, S. G., Corot, C., Vogt, P., Kolb, S. & Debatin, J. F. Magnetic resonance imaging of atherosclerotic plaque with ultrasmall superparamagnetic particles of iron oxide in hyperlipidemic rabbits. Circulation 103, 415–422 (2001). This was the first study to use USPIO-enhanced MRI of atherosclerotic plaques in a rabbit model.
Woollard, K. J. & Geissmann, F. Monocytes in atherosclerosis: subsets and functions. Nature Rev. Cardiol. 7, 77–86 (2010).
Kooi, M. E. et al. Accumulation of ultrasmall superparamagnetic particles of iron oxide in human atherosclerotic plaques can be detected by in vivo magnetic resonance imaging. Circulation 107, 2453–2458 (2003).
Tang, T. Y. et al. The ATHEROMA (Atorvastatin Therapy: Effects on Reduction of Macrophage Activity) Study. Evaluation using ultrasmall superparamagnetic iron oxide-enhanced magnetic resonance imaging in carotid disease. J. Am. Coll. Cardiol. 53, 2039–2050 (2009).
Fayad, Z. A., Razzouk, L., Briley-Saebo, K. C. & Mani, V. Iron oxide magnetic resonance imaging for atherosclerosis therapeutic evaluation: still “rusty?” J. Am. Coll. Cardiol. 53, 2051–2052 (2009).
Corti, R. et al. Effects of aggressive versus conventional lipid-lowering therapy by simvastatin on human atherosclerotic lesions: a prospective, randomized, double-blind trial with high-resolution magnetic resonance imaging. J. Am. Coll. Cardiol. 46, 106–112 (2005).
Flacke, S. et al. Novel MRI contrast agent for molecular imaging of fibrin: implications for detecting vulnerable plaques. Circulation 104, 1280–1285 (2001).
Winter, P. M. et al. Molecular imaging of angiogenesis in early-stage atherosclerosis with αvβ3-integrin-targeted nanoparticles. Circulation 108, 2270–2274 (2003).
Amirbekian, V. et al. Detecting and assessing macrophages in vivo to evaluate atherosclerosis noninvasively using molecular MRI. Proc. Natl Acad. Sci. USA 104, 961–966 (2007).
van Tilborg, G. A. et al. Annexin A5-functionalized bimodal nanoparticles for MRI and fluorescence imaging of atherosclerotic plaques. Bioconjug. Chem. 21, 1794–1803 (2010).
Cormode, D. P. et al. Comparison of synthetic high density lipoprotein (HDL) contrast agents for MR imaging of atherosclerosis. Bioconjug. Chem. 20, 937–943 (2009).
Cormode, D. P. et al. Nanocrystal core high-density lipoproteins: a multimodality contrast agent platform. Nano Lett. 8, 3715–3723 (2008). This paper reports the effect of including nanocrystals in the core of HDL to enable its detection by multiple imaging techniques without compromising the important biophysical features of HDL.
Skajaa, T. et al. Quantum dot and Cy5.5 labeled nanoparticles to investigate lipoprotein biointeractions via Förster resonance energy transfer. Nano Lett. 10, 5131–5138 (2010).
Torchilin, V. P., Frank-Kamenetsky, M. D. & Wolf, G. L. CT visualization of blood pool in rats by using long-circulating, iodine-containing micelles. Acad. Radiol. 6, 61–65 (1999).
Rabin, O., Manuel Perez, J., Grimm, J., Wojtkiewicz, G. & Weissleder, R. An X-ray computed tomography imaging agent based on long-circulating bismuth sulphide nanoparticles. Nature Mater. 5, 118–122 (2006).
Hyafil, F. et al. Noninvasive detection of macrophages using a nanoparticulate contrast agent for computed tomography. Nature Med. 13, 636–641 (2007).
Cormode, D. P. et al. Atherosclerotic plaque composition: analysis with multicolor CT and targeted gold nanoparticles. Radiology 256, 774–782 (2010).
Pan, D. et al. Computed tomography in color: NanoK-enhanced spectral CT molecular imaging. Angew. Chem. Int. Ed. Engl. 49, 9635–9639 (2010).
Rudd, J. H. et al. Imaging atherosclerotic plaque inflammation by fluorodeoxyglucose with positron emission tomography: ready for prime time? J. Am. Coll. Cardiol. 55, 2527–2535 (2010).
Fujimoto, S. et al. Molecular imaging of matrix metalloproteinase in atherosclerotic lesions: resolution with dietary modification and statin therapy. J. Am. Coll. Cardiol. 52, 1847–1857 (2008).
Rominger, A. et al. In vivo imaging of macrophage activity in the coronary arteries using 68Ga-DOTATATE PET/CT: correlation with coronary calcium burden and risk factors. J. Nucl. Med. 51, 193–197 (2010).
Kietselaer, B. L. et al. Noninvasive detection of plaque instability with use of radiolabeled annexin A5 in patients with carotid-artery atherosclerosis. N. Engl. J. Med. 350, 1472–1473 (2004).
Nahrendorf, M. et al. Nanoparticle PET-CT imaging of macrophages in inflammatory atherosclerosis. Circulation 117, 379–387 (2008). This study reports the effects of exploiting the favourable targeting properties of dextran-coated iron oxide nanoparticles for plaque imaging by PET–CT.
Tassa, C., Shaw, S. Y. & Weissleder, R. Dextran-coated iron oxide nanoparticles: a versatile platform for targeted molecular imaging, molecular diagnostics, and therapy. Acc. Chem. Res. 10 Jun 2011 (doi:10.1021/ar200084x).
Almutairi, A. et al. Biodegradable dendritic positron-emitting nanoprobes for the noninvasive imaging of angiogenesis. Proc. Natl Acad. Sci. USA 106, 685–690 (2009).
Nahrendorf, M. et al. Hybrid in vivo FMT-CT imaging of protease activity in atherosclerosis with customized nanosensors. Arterioscler. Thromb. Vasc. Biol. 29, 1444–1451 (2009).
Jaffer, F. A., Libby, P. & Weissleder, R. Optical and multimodality molecular imaging: insights into atherosclerosis. Arterioscler. Thromb. Vasc. Biol. 29, 1017–1024 (2009).
Wang, B. et al. Plasmonic intravascular photoacoustic imaging for detection of macrophages in atherosclerotic plaques. Nano Lett. 9, 2212–2217 (2009).
Lanza, G. M. et al. A novel site-targeted ultrasonic contrast agent with broad biomedical application. Circulation 94, 3334–3340 (1996).
Lindner, J. R. Molecular imaging of cardiovascular disease with contrast-enhanced ultrasonography. Nature Rev. Cardiol. 6, 475–481 (2009).
Rader, D. J. & Daugherty, A. Translating molecular discoveries into new therapies for atherosclerosis. Nature 451, 904–913 (2008).
Dobrovolskaia, M. A. & McNeil, S. E. Immunological properties of engineered nanomaterials. Nature Nanotechnol. 2, 469–478 (2007). This review describes the immunotoxicological aspects of nanotechnology-based therapeutic or diagnostic nanoparticles.
Choi, H. S. & Frangioni, J. V. Nanoparticles for biomedical imaging: fundamentals of clinical translation. Mol. Imaging 9, 291–310 (2010).
Bentzon, J. F. & Falk, E. Atherosclerotic lesions in mouse and man: is it the same disease? Curr. Opin. Lipidol. 21, 434–440 (2010).
Fitzgerald, K. T. et al. Standardization of models and methods used to assess nanoparticles in cardiovascular applications. Small 7, 705–717 (2011).
Buxton, D. B. et al. Report of the national heart, lung, and blood institute working group on the translation of cardiovascular molecular imaging. Circulation 123, 2157–2163 (2011).
Libby, P., Ridker, P. M. & Hansson, G. K. Progress and challenges in translating the biology of atherosclerosis. Nature 473, 317–325 (2011). This review discusses targets for the treatment of atherosclerosis.
Lindsay, A. C. & Choudhury, R. P. Form to function: current and future roles for atherosclerosis imaging in drug development. Nature Rev. Drug Discov. 7, 517–529 (2008). This review describes the role of atherosclerosis imaging on drug development.
Muntendam, P., McCall, C., Sanz, J., Falk, E. & Fuster, V. The BioImage Study: novel approaches to risk assessment in the primary prevention of atherosclerotic cardiovascular disease — study design and objectives. Am. Heart J. 160, 49–57 e1 (2010).
Martinez, A. W. & Chaikof, E. L. Microfabrication and nanotechnology in stent design. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 3, 256–268 (2011).
Dvir, T., Timko, B. P., Kohane, D. S. & Langer, R. Nanotechnological strategies for engineering complex tissues. Nature Nanotechnol. 6, 13–22 (2011).
Park, J. S. et al. A highly sensitive and selective diagnostic assay based on virus nanoparticles. Nature Nanotechnol. 4, 259–264 (2009).
Takahama, H. et al. Prolonged targeting of ischemic/reperfused myocardium by liposomal adenosine augments cardioprotection in rats. J. Am. Coll. Cardiol. 53, 709–717 (2009).
Nahrendorf, M. et al. Detection of macrophages in aortic aneurysms by nanoparticle positron emission tomography–computed tomography. Arterioscler. Thromb. Vasc. Biol. 31, 750–757 (2011).
Lanza, G. M. et al. Rationale for a nanomedicine approach to thrombolytic therapy. Stroke 41, S42–S44 (2010).
Yu, L. et al. Autonomic denervation with magnetic nanoparticles. Circulation 122, 2653–2659 (2010).
Choi, H. S. et al. Renal clearance of quantum dots. Nature Biotech. 25, 1165–1170 (2007). This is an excellent example of the influence of the size of a nanoparticle on its biodistribution and clearance.
Alexis, F., Pridgen, E., Molnar, L. K. & Farokhzad, O. C. Factors affecting the clearance and biodistribution of polymeric nanoparticles. Mol. Pharm. 5, 505–515 (2008).
Klibanov, A. L., Maruyama, K., Torchilin, V. P. & Huang, L. Amphipathic polyethyleneglycols effectively prolong the circulation time of liposomes. FEBS Lett. 268, 235–237 (1990).
Owens, D. E., & Peppas, N. A. Opsonization, biodistribution, and pharmacokinetics of polymeric nanoparticles. Int. J. Pharm. 307, 93–102 (2006).
Knop, K., Hoogenboom, R., Fischer, D. & Schubert, U. S. Poly(ethylene glycol) in drug delivery: pros and cons as well as potential alternatives. Angew. Chem. Int. Ed. Engl. 49, 6288–6308 (2010).
Petros, R. A. & DeSimone, J. M. Strategies in the design of nanoparticles for therapeutic applications. Nature Rev. Drug Discov. 9, 615–627 (2010). This is a review on recent advances in the design and therapeutic potential of nanoparticles.
Nel, A. E. et al. Understanding biophysicochemical interactions at the nano-bio interface. Nature Mater. 8, 543–557 (2009).
Torchilin, V. P. Recent advances with liposomes as pharmaceutical carriers. Nature Rev. Drug Discov. 4, 145–160 (2005).
Williams, K. J., Phillips, M. C. & Rodrigueza, W. V. Structural and metabolic consequences of liposome–lipoprotein interactions. Adv. Drug Deliv. Rev. 32, 31–43 (1998).
Williams, K. J., Scalia, R., Mazany, K. D., Rodrigueza, W. V. & Lefer, A. M. Rapid restoration of normal endothelial functions in genetically hyperlipidemic mice by a synthetic mediator of reverse lipid transport. Arterioscler. Thromb. Vasc. Biol. 20, 1033–1039 (2000).
Rodrigueza, W. V., Klimuk, S. K., Pritchard, P. H. & Hope, M. J. Cholesterol mobilization and regression of atheroma in cholesterol-fed rabbits induced by large unilamellar vesicles. Biochim. Biophys. Acta 1368, 306–320 (1998).
Caride, V. J. & Zaret, B. L. Liposome accumulation in regions of experimental myocardial infarction. Science 198, 735–738 (1977).
Hodis, H. N. et al. Relationship of arterial wall uptake of radiolabeled liposomes to the presence of monocyte/macrophage cells in the hypertensive and atherosclerotic arterial wall. Atherosclerosis 87, 109–117 (1991).
Torchilin, V. P. et al. Targeted accumulation of polyethylene glycol-coated immunoliposomes in infarcted rabbit myocardium. FASEB J. 6, 2716–2719 (1992).
Acknowledgements
We gratefully acknowledge D. P. Cormode for discussions and recommendations. Support for this work was partially provided by the International Atherosclerosis Society (M.E.L). This work was supported by the National Heart, Lung, and Blood Institute (NHLBI) and the US National Institutes of Health (NIH), as a Program of Excellence in Nanotechnology (PEN) Award (Contract no. HHSN268201000045C), as well as by the NIH grants R01 EB009638 (Z.A.F.) and R01 CA155432 (W.J.M.M.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Glossary
- Atherosclerosis
-
A chronic systemic inflammatory disease in which the arterial wall thickens as a result of the accumulation of fatty material within the vessel wall.
- Drug-eluting stents
-
Stents that are typically coated with antiproliferative or immunosuppressive drugs for local release to prevent restenosis within the stent.
- Apolipoprotein
-
The protein constituent of lipoproteins that provides cell specificity and is essential for their structural integrity.
- Vasa vasorum
-
A microvascular network that supplies larger blood vessels with nutrients.
- Stenosis
-
Luminal narrowing of a blood vessel.
- Atherothrombotic events
-
Clinical events such as myocardial infarction or stroke that are the result of atherosclerotic plaque disruption with superimposed thrombosis.
- Reverse cholesterol transport
-
Transport of cholesterol from peripheral cells to the liver for excretion.
- Mononuclear phagocyte system
-
A branch of the immune system that consists of phagocytic cells that can remove macromolecules from circulation.
- 18F-FDG
-
A radioactive glucose analogue that is taken up by metabolically active cells, and is frequently used for molecular imaging in combination with positron emission tomography.
- Restenosis
-
The reoccurence of stenosis after previous treatment.
Rights and permissions
About this article
Cite this article
Lobatto, M., Fuster, V., Fayad, Z. et al. Perspectives and opportunities for nanomedicine in the management of atherosclerosis. Nat Rev Drug Discov 10, 835–852 (2011). https://doi.org/10.1038/nrd3578
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrd3578
This article is cited by
-
Thylakoid engineered M2 macrophage for sonodynamic effect promoted cell therapy of early atherosclerosis
Nano Research (2024)
-
Advances in treatment strategies based on scavenging reactive oxygen species of nanoparticles for atherosclerosis
Journal of Nanobiotechnology (2023)
-
Research progress on the therapeutic effects of nanoparticles loaded with drugs against atherosclerosis
Cardiovascular Drugs and Therapy (2023)
-
Nanoparticles Treat Ischemic Stroke by Responding to Stroke Microenvironment
BioNanoScience (2023)
-
Pharmaceutical nanotechnology: from the bench to the market
Future Journal of Pharmaceutical Sciences (2022)