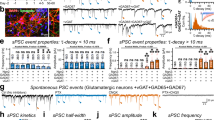
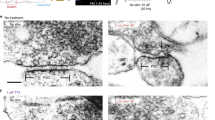
Abstract
Neurotransmitter release is triggered by membrane depolarization, Ca2+ influx and Ca2+ sensing by the release machinery, causing synaptic vesicle (SV) fusion with the plasma membrane. Interlinked is a complex membrane cycle in which vesicles are tethered to the release site, primed, fused and recycled. As many of these processes are Ca2+ dependent and simultaneously occurring, it is difficult to dissect them experimentally. This problem can be partially circumvented by controlling synaptic Ca2+ concentrations via UV photolysis of caged Ca2+. We developed a culture protocol for Ca2+ uncaging in small synapses on the basis of the generation of small glia cell islands with single neurons on top, which are sufficiently small to be covered with a UV-light flash. Neurons are loaded with the photolabile Ca2+-chelator nitrophenyl-EGTA and Ca2+ indicators, and a UV flash is used to trigger Ca2+-uncaging and SV fusion. The protocol takes three weeks to complete and provides unprecedented insights into the mechanisms of transmitter release.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Wojcik, S.M. & Brose, N. Regulation of membrane fusion in synaptic excitation-secretion coupling: speed and accuracy matter. Neuron 55, 11–24 (2007).
Zucker, R.S. & Regehr, W.G. Short-term synaptic plasticity. Annu. Rev. Physiol. 64, 355–405 (2002).
Kavalali, E.T. Multiple vesicle recycling pathways in central synapses and their impact on neurotransmission. J. Physiol. (London) 585, 669–679 (2007).
Neher, E. & Sakaba, T. Multiple roles of calcium ions in the regulation of neurotransmitter release. Neuron 59, 861–872 (2008).
Goda, Y. & Stevens, C.F. Two components of transmitter release at a central synapse. Proc. Natl. Acad. Sci. USA 91, 12942–12946 (1994).
Zucker, R.S. & Lara-Estrella, L.O. Post-tetanic decay of evoked and spontaneous transmitter release and a residual-calcium model of synaptic facilitation at crayfish neuromuscular junctions. J. Gen. Physiol. 81, 355–372 (1983).
Moulder, K.L. & Mennerick, S. Reluctant vesicles contribute to the total readily releasable pool in glutamatergic hippocampal neurons. J. Neurosci. 25, 3842–3850 (2005).
Rosenmund, C. & Stevens, C.F. Definition of the readily releasable pool of vesicles at hippocampal synapses. Neuron 16, 1197–1207 (1996).
Kaplan, J.H. & Ellis-Davies, G.C. Photolabile chelators for the rapid photorelease of divalent cations. Proc. Natl. Acad. Sci. USA 85, 6571–6575 (1988).
Ellis-Davies, G.C. & Kaplan, J.H. Nitrophenyl-EGTA, a photolabile chelator that selectively binds Ca2+ with high affinity and releases it rapidly upon photolysis. Proc. Natl. Acad. Sci. USA 91, 187–191 (1994).
Delaney, K.R. & Zucker, R.S. Calcium released by photolysis of DM-nitrophen stimulates transmitter release at squid giant synapse. J. Physiol. (Lond.) 426, 473–498 (1990).
Tsien, R.Y. & Zucker, R.S. Control of cytoplasmic calcium with photolabile tetracarboxylate 2-nitrobenzhydrol chelators. Biophys. J. 50, 843–853 (1986).
Neher, E. & Zucker, R.S. Multiple calcium-dependent processes related to secretion in bovine chromaffin cells. Neuron 10, 21–30 (1993).
Kirillova, J., Thomas, P. & Almers, W. Two independently regulated secretory pathways in mast cells. J. Physiol. (Paris) 87, 203–208 (1993).
Thomas, P., Wong, J.G. & Almers, W. Millisecond studies of secretion in single rat pituitary cells stimulated by flash photolysis of caged Ca2+. EMBO J. 12, 303–306 (1993).
Schneggenburger, R. & Neher, E. Intracellular calcium dependence of transmitter release rates at a fast central synapse. Nature 406, 889–893 (2000).
Bollmann, J.H., Sakmann, B. & Borst, J.G. Calcium sensitivity of glutamate release in a calyx-type terminal. Science 289, 953–957 (2000).
Sakaba, T. Two Ca2+-dependent steps controlling synaptic vesicle fusion and replenishment at the cerebellar basket cell terminal. Neuron 57, 406–419 (2008).
Kasai, H. Comparative biology of Ca2+-dependent exocytosis: implications of kinetic diversity for secretory function. Trends Neurosci. 22, 88–93 (1999).
Burgalossi, A. et al. SNARE protein recycling by αSNAP and βSNAP supports synaptic vesicle priming. Neuron 68, 473–487 (2010).
Bekkers, J.M. & Stevens, C.F. Excitatory and inhibitory autaptic currents in isolated hippocampal neurons maintained in cell culture. Proc. Natl. Acad. Sci. USA 88, 7834–7838 (1991).
Pyott, S.J. & Rosenmund, C. The effects of temperature on vesicular supply and release in autaptic cultures of rat and mouse hippocampal neurons. J. Physiol. (Lond.) 539, 523–535 (2002).
Segal, M.M. Epileptiform activity in microcultures containing one excitatory hippocampal neuron. J. Neurophysiol. 65, 761–770 (1991).
Rost, B.R. et al. Autaptic cultures of single hippocampal granule cells of mice and rats. Eur. J. Neurosci. 32, 939–947 (2010).
McGuinness, L. et al. Presynaptic NMDARs in the hippocampus facilitate transmitter release at theta frequency. Neuron 68, 1109–1127 (2010).
Jockusch, W.J. et al. CAPS-1 and CAPS-2 are essential synaptic vesicle priming proteins. Cell 131, 796–808 (2007).
Spijker, S. Dissection of the rodent brain regions. Neuroproteomics 57, 13–26 (2011).
Kaech, S. & Banker, G. Culturing hippocampal neurons. Nat. Protoc. 1, 2406–2415 (2006).
Goslin, K., Asmussen, H. & Banker, G. Rat hippocampal neurons in low-density cultures. in Culturing Nerve Cells (eds Banker, G. & Goslin, K.) 339–370 (MIT Press, 1998).
Rosenmund, C., Feltz, A. & Westbrook, G.L. Calcium-dependent inactivation of synaptic NMDA receptors in hippocampal neurons. J. Neurophysiol. 73, 427–430 (1995).
Malenka, R.C., Kauer, J.A., Zucker, R.S. & Nicoll, R.A. Postsynaptic calcium is sufficient for potentiation of hippocampal synaptic transmission. Science 242, 81–84 (1988).
Neveu, D. & Zucker, R.S. Long-lasting potentiation and depression without presynaptic activity. J. Neurophysiol. 75, 2157–2160 (1996).
Neveu, D. & Zucker, R.S. Postsynaptic levels of [Ca2+]i needed to trigger LTD and LTP. Neuron 16, 619–629 (1996).
Van der Kloot, W. Estimating the timing of quantal releases during end-plate currents at the frog neuromuscular junction. J. Physiol. (Lond.) 402, 595–603 (1988).
Acknowledgements
We thank A. Galinski, I. Beulshausen and M. Dörre (Göttingen, Germany) for excellent technical support, and M. Krohn, F. Schütte, R. Otremba and E. Özdemir (Göttingen, Germany) for fine mechanics support. We are particularly grateful to C. Rosenmund (Berlin, Germany) for advice on microisland culture, to T.M. Boeckers (Ulm, Germany) for providing the rabbit polyclonal antibody against ProSap-1 and to J. Sørensen (Copenhagen, Denmark) for help with the [Ca2+]i calibration. This work was supported by the Max Planck Society (Munich, Germany), the German Research Foundation (SFB889, B1) and the European Union (EUROSPIN, SynSys; Brussels, Belgium). K.M.M. is a recipient of a fellowship of the German Academic Exchange Service (DAAD; Bonn, Germany).
Author information
Authors and Affiliations
Contributions
J.-S.R. conceived the method and supervised the experiments. A.B. and W.J.J. performed experiments in the establishment of the protocol. R.N., S.Y.J. and K.M.M. performed additional experiments. A.B., J.-S.R., W.J.J., S.M.W. and N.B. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Technical drawing of the stamp used for generating the growth surface for microisland cultures. (a-b) Bottom (a) and lateral (b) views of the stamp. Insets show higher magnifications of the micro-dotted thermoplastic surface. (PDF 66 kb)
Supplementary Fig. 2
Linear relationship between F1 and F2. The isocoefficient α can be calculated from the linear relationship between F1 and F2. R is the correlation coefficient. (PDF 63 kb)
Supplementary Fig. 3
Mag-Fura-2/Fura-4F calibration curve from Supplementary Table 2. (PDF 55 kb)
Supplementary Table 1
Solutions for purity determination of NP-EGTA, Ca2+ calibration of Fura-4F, and photolysis efficiency (PLE) of NP-EGTA. (PDF 49 kb)
Supplementary Table 2
Ca2+ calibration solutions for Mag-Fura-2 and Fura-4F. (PDF 61 kb)
Supplementary Methods
Ca2+ calibration protocol. (PDF 109 kb)
Rights and permissions
About this article
Cite this article
Burgalossi, A., Jung, S., Man, Kn. et al. Analysis of neurotransmitter release mechanisms by photolysis of caged Ca2+ in an autaptic neuron culture system. Nat Protoc 7, 1351–1365 (2012). https://doi.org/10.1038/nprot.2012.074
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2012.074
This article is cited by
-
Modulation of cognition and neuronal plasticity in gain- and loss-of-function mouse models of the schizophrenia risk gene Tcf4
Translational Psychiatry (2020)
-
Cholesterol modulates presynaptic and postsynaptic properties of excitatory synaptic transmission
Scientific Reports (2020)
-
In vivo mouse and live cell STED microscopy of neuronal actin plasticity using far-red emitting fluorescent proteins
Scientific Reports (2017)
-
Photoswitchable diacylglycerols enable optical control of protein kinase C
Nature Chemical Biology (2016)
-
Rheb activation disrupts spine synapse formation through accumulation of syntenin in tuberous sclerosis complex
Nature Communications (2015)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.