Abstract
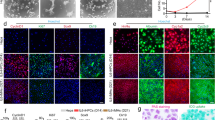
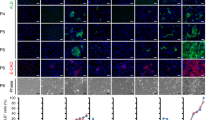
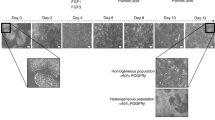
Differentiation of stem cells to hepatocytes has industrial applications, as well as the potential to develop new therapeutic strategies for liver disease. The protocol described here, sequentially using cytokines that are known to have a role in liver embryonic development, efficiently differentiates rat multipotent adult progenitor cells (rMAPCs) to hepatocyte-like cells by directing them through defined embryonic intermediates, namely, primitive streak/mesendoderm/definitive endoderm, hepatoblast and hepatocyte-like phenotype. After 20 days, the final differentiated multipotent adult progenitor cell progeny is a mixture of cells, comprising cells with the characteristics of hepatoblasts and a smaller cell fraction with the morphological and phenotypical features of mature hepatocytes, as well as other mesodermal cells and some persistent undifferentiated rMAPCs. A detailed functional characterization of the stem cell progeny is also described; this should be used to confirm that differentiated cells display the functional characteristics of mature hepatocytes, including albumin secretion, glycogen storage and several detoxifying functions such as urea production, bilirubin conjugation, glutathione S-transferase activity and cytochrome activity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Subramanian, K. et al. in Cellular Programming and Reprogramming: Methods and Protocols 1st edn. (ed. Ding, S.) 55–78 (Humana Press, 2010).
Breyer, A. et al. Multipotent adult progenitor cell isolation and culture procedures. Exp. Hematol. 34, 1596–1601 (2006).
Ulloa-Montoya, F. et al. Comparative transcriptome analysis of embryonic and adult stem cells with extended and limited differentiation capacity. Genome Biol. 8, R163 (2007).
Mitsui, K. et al. The homeoprotein Nanog is required for maintenance of pluripotency in mouse epiblast and ES cells. Cell 113, 631–642 (2003).
Kunath, T. et al. Imprinted X-inactivation in extra-embryonic endoderm cell lines from mouse blastocysts. Development 132, 1649–1661 (2005).
Debeb, B.G. et al. Isolation of Oct4-expressing extraembryonic endoderm precursor cell lines. PLoS ONE 4, e7216 (2009).
Jiang, Y. et al. Pluripotency of mesenchymal stem cells derived from adult marrow. Nature 418, 41–49 (2002).
Kogler, G. et al. A new human somatic stem cell from placental cord blood with intrinsic pluripotent differentiation potential. J. Exp. Med. 200, 123–135 (2004).
Yoon, Y.S. et al. Clonally expanded novel multipotent stem cells from human bone marrow regenerate myocardium after myocardial infarction. J. Clin. Invest. 115, 326–338 (2005).
D′Ippolito, G. et al. Marrow-isolated adult multilineage inducible (MIAMI) cells, a unique population of postnatal young and old human cells with extensive expansion and differentiation potential. J. Cell Sci. 117, 2971–2981 (2004).
De Coppi, P. et al. Isolation of amniotic stem cell lines with potential for therapy. Nat. Biotech. 25, 100–106 (2007).
Dan, Y.Y. et al. Isolation of multipotent progenitor cells from human fetal liver capable of differentiating into liver and mesenchymal lineages. Proc. Natl. Acad. Sci. USA 103, 9912–9917 (2006).
Kucia, M. et al. A population of very small embryonic-like (VSEL) CXCR4+ SSEA-1+ Oct-4+ stem cells identified in adult bone marrow. Leukemia 20, 857–869 (2006).
Anjos-Afonso, F. & Bonnet, D. Non-hematopoietic/endothelial SSEA-1+ cells defines the most primitive progenitors in the adult murine bone marrow mesenchymal compartment. Blood 109, 1298–1306 (2007).
Beltrami, A.P. et al. Multipotent cells can be generated in vitro from several adult human organs (heart, liver, and bone marrow). Blood 110, 3438–3446 (2007).
Schwartz, R.E. et al. Multipotent adult progenitor cells from bone marrow differentiate into functional hepatocyte-like cells. J. Clin. Invest. 109, 1291–1302 (2002).
Lee, O.K. et al. Isolation of multipotent mesenchymal stem cells from umbilical cord blood. Blood 103, 1669–1675 (2004).
Chien, C.C. et al. In vitro differentiation of human placenta-derived multipotent cells into hepatocyte-like cells. Stem Cells 24, 1759–1768 (2006).
Snykers, S. et al. Sequential exposure to cytokines reflecting embryogenesis: the key for in vitro differentiation of adult bone marrow stem cells into functional hepatocyte-like cells. Toxicol. Sci. 94, 330–341 (2006).
Snykers, S., De Kock, J., Rogiers, V. & Vanhaecke, T. In vitro differentiation of embryonic and adult stem cells into hepatocytes: state of the art. Stem Cells 27, 577–605 (2009).
Banas, A. et al. Adipose tissue-derived mesenchymal stem cells as a source of human hepatocytes. Hepatology 46, 219–228 (2007).
Lee, K.D. et al. In vitro hepatic differentiation of human mesenchymal stem cells. Hepatology 40, 1275–1284 (2004).
Grünewald, J.O., Walker, J.C. & Strope, E.R. Photooxidation of xanthobilirubic acid in aqueous solution: product and mechanism studies. Photochem. Photobiol. 24, 29–40 (1976).
Seifter, S., Dayton, S., Novic, B. & Muntwyler, E. The estimation of glycogen with the anthrone reagent. Arch. Biochem. 25, 191–200 (1950).
Habig, W.H., Pabst, M.J. & Jakoby, W.B. Glutathione S-transferases. The first enzymatic step in mercapturic acid formation. J. Biol. Chem. 249, 7130–7139 (1974).
Muraca, M. & Blanckaert, N. Liquid-chromatographic assay and identification of mono- and diester conjugates of bilirubin in normal serum. Clin. Chem. 29, 1767–1771 (1983).
Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 72, 248–254 (1976).
Sancho-Bru, P. et al. Stem and progenitor cells for liver repopulation: can we standardise the process from bench to bedside? Gut 58, 594–603 (2009).
Acknowledgements
We thank F. Nevens, J. van Pelt, P. Windmolders, I. Vander Elst and M. Zeegers (Hepatology Lab, K.U. Leuven) for their help with functional assays and T. Notelaers (SCIL) for help with differentiation studies. This work was supported by grants to C.V. from the NIH (RO1-HL67932, RO1 DK58295, RO1-HL073221 and U19-DK61244), the Alpha 1 Foundation, FWO G.0667.07 Odysseus award and a K.U. Leuven SCIL Center of Excellence Award. C.V. also received research funding from Athersys Inc. (Cleveland, OH, USA). P.R. was funded by IWT and VVGE. P.S.-B. was funded by FWO (KAN 1.5163.09) and Agència de Gestió d'Ajuts Universitaris i de Recerca, Beatriu de Pinós (2006 BP-A 10104). K.P. was funded by FWO, BAEF and VVGE.
Author information
Authors and Affiliations
Contributions
P.R. and P.S.-B. prepared the manuscript. K.P. and C.V. designed the protocol. P.R., P.S.-B. and K.P. conducted the experiments, RT-qPCR, immunocytochemistry and functional assays. C.V. supervised the project.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Roelandt, P., Sancho-Bru, P., Pauwelyn, K. et al. Differentiation of rat multipotent adult progenitor cells to functional hepatocyte-like cells by mimicking embryonic liver development. Nat Protoc 5, 1324–1336 (2010). https://doi.org/10.1038/nprot.2010.80
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2010.80
This article is cited by
-
Directed Differentiation of Adult Liver Derived Mesenchymal Like Stem Cells into Functional Hepatocytes
Scientific Reports (2018)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.