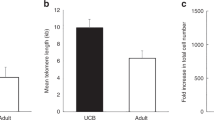
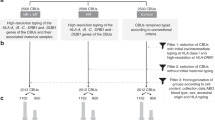
Abstract
Umbilical cord blood is a key source of stem cells for transplantation and regenerative medicine. To maximize each cord blood sample, it is important to analyze its cellular populations. Many cord blood banks focus on counts of total nucleated cells and/or cells that carry the CD34 antigen, but this limited focus does not give a true estimation of cord blood content, quality and appropriateness for use. This protocol is the first of its kind to enable a comprehensive investigation of cord blood cellular populations. Using multicolor flow cytometry it is possible to examine expression of 26 antigens—including hematopoietic markers CD45 and CD34, immune markers CD19 and CD3, and HLA—using a total of only 1 × 106 cells from each unit for both pre- and post-processing. The samples are stained, lysed, washed and analyzed flow cytometrically. This method also provides valuable information beyond cord blood composition and quality; for example, it can also be used to assess whether maternal factors affect CD34+ cell numbers. Collection, processing, cryopreservation and initial flow cytometry take ∼5 h.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gluckman, E. et al. Hematopoietic reconstitution in a patient with Fanconi's anemia by means of umbilical-cord blood from an HLA-identical sibling. N. Engl. J. Med. 321, 1174–1178 (1989).
Escolar, M.L. et al. Transplantation of umbilical-cord blood in babies with infantile Krabbe's disease. N. Engl. J. Med. 352, 2069–2081 (2005).
Slatter, M.A. & Gennery, A.R. Umbilical cord stem cell transplantation for primary immunodeficiencies. Expert Opin. Biol. Ther. 6, 555–565 (2006).
Gluckman, E. & Rocha, V. Cord blood transplantation: state of the art. Haematologica 94, 451–454 (2009).
Grewal, S.S., Barker, J.N., Davies, S.M. & Wagner, J.E. Unrelated donor hematopoietic cell transplantation: marrow or umbilical cord blood? Blood 101, 4233–4244 (2003).
Fasouliotis, S.J. & Schenker, J.G. Human umbilical cord blood banking and transplantation: a state of the art. Eur. J. Obstet. Gynecol. Reprod. Biol. 90, 13–25 (2000).
Van Haute, I. et al. Selecting cord blood units for storage by CD34+ cell counts. Transfusion 45, 455–457 (2005).
Cohen, Y. & Nagler, A. Umbilical cord blood transplantation–how, when and for whom? Blood Rev. 18, 167–179 (2004).
McGuckin, C.P., Basford, C., Hanger, K., Habibollah, S. & Forraz, N. Cord blood revelations—The importance of being a first born girl, big, on time and to a young mother! Early Hum. Dev. 83, 733–741 (2007).
Rabian-Herzog, C., Lesage, S. & Gluckman, E. Characterization of lymphocyte subpopulations in cord blood. Bone Marrow Transplant. 9 Suppl 1: 64–67 (1992).
D'Arena, G. et al. Flow cytometric characterization of human umbilical cord blood lymphocytes: immunophenotypic features. Haematologica 83, 197–203 (1998).
Bradford, J.A., Buller, G., Suter, M., Ignatius, M. & Beechem, J.M. Fluorescence-intensity multiplexing: simultaneous seven-marker, two-color immunophenotyping using flow cytometry. Cytometry A 61, 142–152 (2004).
Petrovic, S. et al. SLC26A7: a basolateral Cl−/HCO3 − exchanger specific to intercalated cells of the outer medullary collecting duct. Am. J. Physiol. Renal Physiol. 286, F161–F169 (2004).
Airey, J.A. et al. Human mesenchymal stem cells form Purkinje fibers in fetal sheep heart. Circulation 109, 1401–1407 (2004).
Witkovsky, P. et al. Activity-dependent phosphorylation of tyrosine hydroxylase in dopaminergic neurons of the rat retina. J. Neurosci. 24, 4242–4249 (2004).
Basford, C., Forraz, N., Habibollah, S., Hanger, K. & McGuckin, C.P. Umbilical cord blood processing using Prepacyte-CB increases haematopoietic progenitor cell availability over conventional Hetastarch separation. Cell Prolif. 42, 751–761 (2009).
Stelzer, G.T. et al. U.S.-Canadian Consensus recommendations on the immunophenotypic analysis of hematologic neoplasia by flow cytometry: standardization and validation of laboratory procedures. Cytometry 30, 214–230 (1997).
Roederer, M. et al. 8 color, 10-parameter flow cytometry to elucidate complex leukocyte heterogeneity. Cytometry 29, 328–339 (1997).
Alamo, A.L. & Melnick, S.J. Clinical application of four and five-color flow cytometry lymphocyte subset immunophenotyping. Cytometry 42, 363–370 (2000).
Keeney, M., Gratama, J.W., Chin-Yee, I.H. & Sutherland, D.R. Isotype controls in the analysis of lymphocytes and CD34+ stem and progenitor cells by flow cytometry—time to let go! Cytometry 34, 280–283 (1998).
Trummer, A., De Rop, C., Tiede, A., Ganser, A. & Eisert, R. Isotype controls in phenotyping and quantification of microparticles: a major source of error and how to evade it. Thromb. Res. 122, 691–700 (2008).
Tung, J.W., Parks, D.R., Moore, W.A. & Herzenberg, L.A. New approaches to fluorescence compensation and visualization of FACS data. Clin. Immunol. 110, 277–283 (2004).
Wagner, J.E. et al. Transplantation of unrelated donor umbilical cord blood in 102 patients with malignant and nonmalignant diseases: influence of CD34 cell dose and HLA disparity on treatment-related mortality and survival. Blood 100, 1611–1618 (2002).
McGuckin, C.P. et al. Production of stem cells with embryonic characteristics from human umbilical cord blood. Cell Prolif. 38, 245–255 (2005).
Basford, C. Umbilical cord blood processing with Prepacyte-CB increases haematopoietic progenitor cell availability over conventional Hetastarch separation. Cell Prolif. 42, 751–761 (2009).
Acknowledgements
We thank the staff at Newcastle's Royal Victoria Infirmary Woman's Directorate and Maternity Unit. We are also grateful to the One North East regional development agency of the British Government for their financial support to our laboratories. We thank I. Dimmick for his excellent advice during FACS analysis development and M. Lako for her supervision and laboratory role in the later stages of the project.
Author information
Authors and Affiliations
Contributions
C.B., C.M. and N.F. conceived the protocol. C.M. and N.F. supervised the project. C.B. performed the experiments, analyzed the data and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Basford, C., Forraz, N. & McGuckin, C. Optimized multiparametric immunophenotyping of umbilical cord blood cells by flow cytometry. Nat Protoc 5, 1337–1346 (2010). https://doi.org/10.1038/nprot.2010.88
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2010.88
This article is cited by
-
Evaluation of committed and primitive cord blood progenitors after expansion on adipose stromal cells
Cell and Tissue Research (2018)
-
Intraarterial transplantation of human umbilical cord blood mononuclear cells is more efficacious and safer compared with umbilical cord mesenchymal stromal cells in a rodent stroke model
Stem Cell Research & Therapy (2014)
-
Enrichment of Umbilical Cord Blood Mononuclears with Hemopoietic Precursors in Co-Culture with Mesenchymal Stromal Cells from Human Adipose Tissue
Bulletin of Experimental Biology and Medicine (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.