Abstract
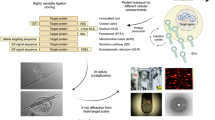
This protocol describes the method of expression cloning of heterologous proteins using Xenopus laevis oocytes and the functional characterization of membrane proteins using radiotracer assays. It can be used to isolate proteins for which sequence data is unavailable and to characterize the functions of proteins. A cDNA library is generated, that is, translated into proteins in Xenopus oocytes, and the function of these proteins is assessed by a radiotracer assay. Their large size, high degree of expression and ease of handling makes Xenopus oocytes an optimal tool for the expression and characterization of protein function when compared with traditional expression systems, such as Escherichia coli, yeast or eukaryotic cell lines. The expected results of this technique include the following: functional identification of novel proteins; molecular (kinetic) characterization of protein function; and determination of functionally relevant residues and domains of membrane proteins, including transporters, ion channels and receptors. The identification of novel proteins can take several months, whereas functional characterization in Xenopus oocytes can take 1 week.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gurdon, J., Lane, C., Woodland, H. & Marbaix, G. Use of frog eggs and oocytes for the study of messenger RNA and its translation in living cells. Nature 233, 177–182 (1971).
Hediger, M., Coady, M., Ikeda, T. & Wright, E. Expression cloning and cDNA sequencing of the Na+/glucose cotransporter. Nature 330, 379–381 (1987).
Werner, A. et al. Cloning and expression of cDNA for a Na/Pi cotransport system of kidney cortex. Proc. Natl. Acad. Sci. USA 88, 9608–9612 (1991).
Bertran, J. et al. Expression-cloning of a cDNA from rabbit kidney cortex that induces a single transport system for cystine, dibasic and neutral amino acids. Proc. Natl. Acad. Sci. USA 89, 5601–5605 (1992).
Wells, R. & Hediger, M. Cloning of a rat kidney cDNA that stimulates dibasic and neutral amino acid transport and has sequence similarity to glucosidases. Proc. Natl. Acad. Sci. USA 89, 5596–5600 (1992).
Markovich, D. et al. Two mRNA transcripts (rBAT-1 and rBAT-2) are involved in system bo,+-related amino acid transport. J. Biol. Chem. 268, 1362–1367 (1993).
Tate, S.S., Yan, N. & Udenfriend, S. Expression cloning of a Na(+)-independent neutral amino acid transporter from rat kidney. Proc. Natl. Acad. Sci. USA 89, 1–5 (1992).
Magagnin, S. et al. Expression cloning of human and rat renal cortex Na/Pi cotransport. Proc. Natl. Acad. Sci. USA 90, 5979–5983 (1993).
Markovich, D., Forgo, J., Stange, G., Biber, J. & Murer, H. Expression cloning of rat renal Na+/SO4(2−) cotransport. Proc. Natl. Acad. Sci. USA 90, 8073–8077 (1993).
Hagenbuch, B., Stieger, B., Foguet, M., Lübbert, H. & Meier, P. Functional expression cloning and characterization of the hepatocyte Na+/bile acid cotransport system. Proc. Natl. Acad. Sci. USA 88, 10629–10633 (1991).
Bissig, M., Hagenbuch, B., Stieger, B., Koller, T. & Meier, P.J. Functional expression cloning of the canalicular sulfate transport system of rat hepatocytes. J. Biol. Chem. 269, 3017–3021 (1994).
Guastella, J. et al. Cloning and expression of a rat brain GABA transporter. Science 249, 1303–1306 (1990).
Kanai, Y. & Hediger, M. Primary structure and functional characterization of a high affinity glutamate transporter. Nature 360, 467–471 (1992).
Romero, M., Kanai, Y., Gunshin, H. & Hediger, M. Expression cloning using Xenopus laevis oocytes. Methods Enzymol 296, 17–52 (1998).
Sigel, E. Use of Xenopus oocytes for the functional expression of plasma membrane proteins. J. Membr. Biol. 117, 210–221 (1990).
Tallgren, L.G. Inorganic sulphates in relation to the serum thyroxine level and in renal failure. Acta. Med. Scand. Suppl. 640, 1–100 (1980).
Van Balkom, B.W. et al. Role of cytoplasmic termini in sorting and shuttling of the aquaporin-2 water channel. Am. J. Physiol. Cell. Physiol. 286, C372–C379 (2004).
Colman, A. In Transcription and Translation: a Practical Approach (eds. Hames B.D. & Higgins S.J.) 271–302 (IRL Press Ltd, Oxford, England, 1984).
Markovich, D., Werner, A. & Murer, H. In Techniques in Molecular Medicine (eds. Hildebrandt F. & Igarashi P.) 310–318 (Springer Verlag, Heidelberg, 1999).
Smith, L.D., Xu, W. & Varnold, R.L. Oogenesis and oocyte isolation. Methods Cell Biol. 36, 45–60 (1991).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Markovich, D. Expression cloning and radiotracer uptakes in Xenopus laevis oocytes. Nat Protoc 3, 1975–1980 (2008). https://doi.org/10.1038/nprot.2008.151
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2008.151
This article is cited by
-
Evaluating pathogenicity of SLC34A3-Ser192Leu, a frequent European missense variant in disorders of renal phosphate wasting
Urolithiasis (2019)
-
Expression cloning human and rat renal cortex Na/Pi cotransporters: behind the scenes in the Murer laboratory
Pflügers Archiv - European Journal of Physiology (2019)
-
Di- and tripeptide transport in vertebrates: the contribution of teleost fish models
Journal of Comparative Physiology B (2017)
-
Na+–sulfate cotransporter SLC13A1
Pflügers Archiv - European Journal of Physiology (2014)
-
Physiological Roles of Mammalian Sulfate Transporters NaS1 and Sat1
Archivum Immunologiae et Therapiae Experimentalis (2011)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.