Abstract
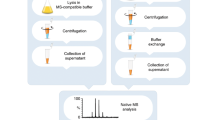
Identification of proteins with enzymatic activity by mass spectrometry (MS) and concomitant determination of function by screening enzyme activity from two-dimensional gel electrophoresis (2DE) is one of the challenges of gel-based proteomics. In this protocol, proteins are extracted from spinal cord tissue followed by 2DE with in-gel digestion and identification by matrix-assisted laser desorption/ionization. Protein spots identified as possible enzyme of interest are punched, eluted by SDS-containing Tris buffer and renatured by buffers under reductive conditions. Enzyme activity is determined using micromethods. Within about 4 weeks, a structural and functional map can be generated and MS identification can be validated, complementing immunochemical methods. 2DE separation can be seen as a prepurification step and therefore allows activity assays from minute amounts of protein as provided in a 2DE gel spot; the method may be an alternative to the time-consuming use of recombinant enzyme techniques.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout










Similar content being viewed by others
References
Chen, W.Q., Kang, S.U. & Lubec, G. Protein profiling by the combination of two independent mass spectrometry techniques. Nat. Protoc. 1, 1446–1452 (2006).
Lubec, G., Afjehi-Sadat, L., Yang, J.W. & John, J.P. Searching for hypothetical proteins: theory and practice based upon original data and literature. Prog. Neurobiol. 77, 90–127 (2005).
John, J.P. et al. Nitric oxide and oxygen radical attack on GDP-dissociation inhibitor 2 (GDI-2) in spinal cord injury of the rat. J. Proteome Res. 6, 1500–1509 (2007).
Chou, K.C. & Elrod, D.W. Prediction of enzyme family classes. J. Proteome Res. 2, 183–190 (2003).
Chou, K.C. & Cai, Y.D. Predicting enzyme family class in a hybridization space. Protein Sci. 13, 2857–2863 (2004).
Chou, K.C. Using amphiphilic pseudo amino acid composition to predict enzyme subfamily classes. Bioinformatics 21, 10–19 (2005).
Chou, K.C. & Cai, Y.D. Predicting enzyme family class in a hybridization space. Using GO-PseAA predictor to predict enzyme sub-class. Biochem. Biophys. Res. Commun. 325, 506–509 (2004).
Kuznetsova, E. et al. Enzyme genomics: application of general enzymatic screens to discover new enzymes. FEMS Microbiol. Rev. 29, 263–279 (2005).
Schmidt, A.J., Ryjenkov, D.A. & Gomelsky, M. The ubiquitous protein domain EAL is a cyclic diguanylate-specific phosphodiesterase: enzymatically active and inactive EAL domains. J. Bacteriol. 187, 4774–4781 (2005).
Prakash, P., Aruna, B., Sardesai, A.A. & Hasnain, S.E. Purified recombinant hypothetical protein coded by open reading frame Rv1885c of Mycobacterium tuberculosis exhibits a monofunctional AroQ class of periplasmic chorismate mutase activity. J. Biol. Chem. 280, 19641–19648 (2005).
Thuswaldner, S. et al. Identification, expression, and functional analyses of a thylakoid ATP/ADP carrier from Arabidopsis. J. Biol. Chem. 282, 8848–8859 (2007).
Fumic, K. et al. Functional analysis of human S-adenosylhomocysteine hydrolase isoforms SAHH-2 and SAHH-3. Eur. J. Hum. Genet. 15, 347–351 (2007).
Hager, D.A. & Burgess, R.R. Elution of proteins from sodium dodecyl sulfate-polyacrylamide gels, removal of sodium dodecyl sulfate, and renaturation of enzymatic activity: results with sigma subunit of Escherichia coli RNA polymerase, wheat germ DNA topoisomerase, and other enzymes. Anal. Biochem. 109, 76–86 (1980).
Afjehi-Sadat, L. et al. Structural and functional analysis of hypothetical proteins in mouse hippocampus from two-dimensional gel electrophoresis. J. Proteome Res. 6, 711–723 (2007).
Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein–dye binding. Anal. Biochem. 72, 248–254 (1976).
Howell, A., Dubrac, S., Noone, D., Varughese, K.I. & Devine, K. Interactions between the YycFG and PhoPR two-component systems in Bacillus subtilis: the PhoR kinase phosphorylates the non-cognate YycF response regulator upon phosphate limitation. Mol. Microbiol. 59, 1199–1215 (2006).
Genda, T., Nakamatsu, T. & Ozak, H. Purification and characterization of malate dehydrogenase from Corynebacterium glutamicum. J. Biosci. Bioeng. 95, 562–566 (2003).
Habig, W. & Racusen, D. An apparent oligomer of malate dehydrogenase from bean leaves. Plant Physiol. 53, 402–410 (1974).
Harris, D.G., Marx, D.P., Anderson, J.M., McCune, R.W. & Zimmerman, S.S. Kinetic and molecular modeling of nucleoside and nucleotide inhibition of malate dehydrogenase. Nucleosides Nucleotides Nucleic Acids 21, 813–823 (2002).
Hung, H.C., Chien, Y.C., Hsieh, J.Y., Chang, G.G. & Liu, G.Y. Functional roles of ATP-binding residues in the catalytic site of human mitochondrial NAD(P)+-dependent malic enzyme. Biochemistry 44, 12737–12745 (2005).
Yan, J.X. et al. A modified silver staining protocol for visualization of proteins compatible with matrix-assisted laser desorption/ionization and electrospray ionization-mass spectrometry. Electrophoresis 21, 3666–3672 (2000).
Yao, H. & Sem, D.S. Cofactor fingerprinting with STD NMR to characterize proteins of unknown function: identification of a rare cCMP cofactor preference. FEBS Lett. 579, 661–666 (2005).
Kleemann, R. et al. Specific reduction of insulin disulfides by macrophage migration inhibitory factor (MIF) with glutathione and dihydrolipoamide: potential role in cellular redox processes. FEBS Lett. 430, 191–196 (1998).
Acknowledgements
We are highly indebted to Professor Dr. Harald Höger (Institute for Animal Genetics, Medical University of Vienna) for supplying rat spinal cord tissues. We appreciate the technical assistance of Mrs. Maureen Cabatic-Felizardo and the general advice for use of enzyme protocols by Professor Dr. Christian Kubicek (Technical University of Vienna, Austria).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Table 1
Mass spectrometrical identification of metabolic enzymes in rat spinal cord tissue by MALDI-TOF/TOF analyses (PDF 23 kb)
Supplementary Table 2
Reagent and protocols for enzyme assays (PDF 129 kb)
Supplementary Table 3
Enzyme activities and underlying chemical reactions (PDF 22 kb)
Supplementary Table 4
Activity of metabolic enzymes identified in rat spinal cord tissue (PDF 16 kb)
Supplementary Table 5
Effects of inhibitors for malate dehydrogenase (PDF 7 kb)
Rights and permissions
About this article
Cite this article
Afjehi-Sadat, L., Lubec, G. Identification of enzymes and activity from two-dimensional gel electrophoresis. Nat Protoc 2, 2318–2324 (2007). https://doi.org/10.1038/nprot.2007.317
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.317
This article is cited by
-
Proteomic characterization and bio-informatic analysis of differentially expressed E. coli Nissle 1917 proteins with response to cocoti wine stress
3 Biotech (2017)
-
A first partial Aplysia californica proteome
Amino Acids (2011)
-
Purification of recombinant growth hormone by clear native gels for conformational analyses: preservation of conformation and receptor binding
Amino Acids (2010)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.