Abstract
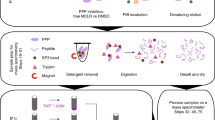
Protein phosphorylation plays important roles in various aspects of cellular events. Visualization of site-specific phosphorylation in cells is of great importance not only to analyze spatial and temporal distribution but also to investigate biological function. Now, site- and phosphorylation state-specific antibodies are widely utilized as the most powerful tools for these analyses. This protocol details a method to produce the polyclonal version of such an antibody by immunizing a synthetic phosphopeptide corresponding to a protein phosphorylated at targeted site(s). This protocol is also applicable to the production of other types of antibodies, which specifically recognize the site-specific modification, such as acetylation, methylation and proteolysis. The protocol can be completed in 2–3 months.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Graves, J.D. & Krebs, E.G. Protein phosphorylation and signal transduction. Pharmacol. Ther. 82, 111–121 (1999).
Nishizuka, Y. Intracellular signaling by hydrolysis of phospholipids and activation of protein kinase C. Science 258, 607–614 (1992).
Nurse, P. A long twentieth century of the cell cycle and beyond. Cell 100, 71–78 (2000).
Zhou, B.B. & Elledge, S.J. The DNA damage response: putting checkpoints in perspective. Nature 408, 433–439 (2000).
Bishop, A.L. & Hall, A. Rho GTPases and their effector proteins. Biochem. J. 348, 241–255 (2000).
Boyle, W.J., van der Geer, P. & Hunter, T. Phosphopeptide mapping and phosphoamino acid analysis by two-dimensional separation on thin-layer cellulose plates. Methods Enzymol. 201, 110–149 (1991).
Sternberger, L.A. & Sternberger, N.H. Monoclonal antibodies distinguish phosphorylated and nonphosphorylated forms of neurofilaments in situ. Proc. Natl. Acad. Sci. USA 80, 6126–6130 (1983).
Nishizawa, K. et al. Specific localization of phosphointermediate filament protein in the constricted area of dividing cells. J. Biol. Chem. 266, 3074–3079 (1991).
Yano, T. et al. A monoclonal antibody to the phosphorylated form of glial fibrillary acidic protein: application to a non-radioactive method for measuring protein kinase activities. Biochem. Biophys. Res. Commun. 175, 1144–1151 (1991).
Nagata, K., Izawa, I. & Inagaki, M. A decade of site- and phosphorylation state-specific antibodies: recent advances in studies of spatiotemporal protein phosphorylation. Genes Cells 6, 653–664 (2001).
Czernik, A.J. et al. Production of phosphorylation state-specific antibodies. Methods Enzymol. 201, 264–283 (1991).
White, D.A., Belyaev, N.D. & Turner, B.M. Preparation of site-specific antibodies to acetylated histones. Methods 19, 417–424 (1999).
Perez-Burgos, L. et al. Generation and characterization of methyl-lysine histone antibodies. Methods Enzymol. 376, 234–254 (2004).
Goto, H. et al. Complex formation of Plk1 and INCENP required for metaphase-anaphase transition. Nat. Cell Biol. 8, 180–187 (2006).
Carmena, M. & Earnshaw, W.C. The cellular geography of aurora kinases. Nat. Rev. Mol. Cell Biol. 4, 842–854 (2003).
Goto, H. et al. Identification of a novel phosphorylation site on histone H3 coupled with mitotic chromosome condensation. J. Biol. Chem. 274, 25543–25549 (1999).
Hendzel, M.J. et al. Mitosis-specific phosphorylation of histone H3 initiates primarily within pericentromeric heterochromatin during G2 and spreads in an ordered fashion coincident with mitotic chromosome condensation. Chromosoma 106, 348–360 (1997).
Kawajiri, A. & Inagaki, M. Approaches to study phosphorylation of intermediate filament proteins using site-specific and phosphorylation state-specific antibodies. Methods Cell Biol. 78, 353–371 (2004).
Bishop, J.D. & Schumacher, J.M. Phosphorylation of the carboxyl terminus of inner centromere protein (INCENP) by the Aurora B kinase stimulates Aurora B kinase activity. J. Biol. Chem. 277, 27577–27580 (2002).
Honda, R., Körner, R. & Nigg, E.A. Exploring the functional interactions between Aurora B, INCENP, and survivin in mitosis. Mol. Biol. Cell 14, 3325–3341 (2003).
Hsu, J.Y. et al. Mitotic phosphorylation of histone H3 is governed by Ipl1/aurora kinase and Glc7/PP1 phosphatase in budding yeast and nematodes. Cell 102, 279–291 (2000).
Goto, H., Yasui, Y., Nigg, E.A. & Inagaki, M. Aurora-B phosphorylates histone H3 at serine28 with regard to the mitotic chromosome condensation. Genes Cells 7, 11–17 (2002).
Tsujimura, K. et al. Visualization and function of vimentin phosphorylation by cdc2 kinase during mitosis. J. Biol. Chem. 269, 31097–31106 (1994).
Acknowledgements
This work was supported in part by Grants-in-Aid for Scientific Research on Priority Areas from the Ministry of Education, Culture, Sports, Science and Technology, Japan, by Grants-in-Aid for Scientific Research from Japan Society for the Promotion of Science, by a Grant-in-aid for the Third Term Comprehensive 10-Year Strategy for Cancer Control from the Ministry of Health and Welfare, Japan, by Astellas Foundation for Research on Metabolic Disorders, by Aichi Cancer Research Foundation, by The Ichiro Kanehara Foundation and by The Mochida Memorial Foundation for Medical and Pharmaceutical Research. Protocol derived from http://dx.doi.org/10.1038/nprot.2007.31.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Goto, H., Inagaki, M. Production of a site- and phosphorylation state-specific antibody. Nat Protoc 2, 2574–2581 (2007). https://doi.org/10.1038/nprot.2007.374
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.374
This article is cited by
-
GRAF1 integrates PINK1-Parkin signaling and actin dynamics to mediate cardiac mitochondrial homeostasis
Nature Communications (2023)
-
Increased Neuronal Differentiation of Neural Progenitor Cells Derived from Phosphovimentin-Deficient Mice
Molecular Neurobiology (2018)
-
Novel method for the high-throughput production of phosphorylation site-specific monoclonal antibodies
Scientific Reports (2016)
-
PLK1 regulation of PCNT cleavage ensures fidelity of centriole separation during mitotic exit
Nature Communications (2015)
-
Generation and purification of highly specific antibodies for detecting post-translationally modified proteins in vivo
Nature Protocols (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.