Abstract
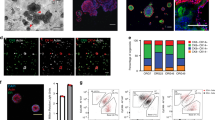
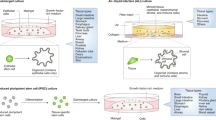
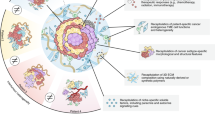
This protocol describes a strategy for the generation of 3D prostate organoid cultures from healthy mouse and human prostate cells (either bulk or FACS-sorted single luminal and basal cells), metastatic prostate cancer lesions and circulating tumor cells. Organoids derived from healthy material contain the differentiated luminal and basal cell types, whereas organoids derived from prostate cancer tissue mimic the histology of the tumor. We explain how to establish these cultures in the fully defined serum-free conditioned medium that is required to sustain organoid growth. Starting with the plating of digested tissue material, full-grown organoids can usually be obtained in ∼2 weeks. The culture protocol we describe here is currently the only one that allows the growth of both the luminal and basal prostatic epithelial lineages, as well as the growth of advanced prostate cancers. Organoids established using this protocol can be used to study many different aspects of prostate biology, including homeostasis, tumorigenesis and drug discovery.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Pienta, K.J. et al. The current status of preclinical prostate cancer animal models. Prostate 68, 629–639 (2008).
Toivanen, R., Taylor, R.A., Pook, D.W., Ellem, S.J. & Risbridger, G.P. Breaking through a roadblock in prostate cancer research: an update on human model systems. J. Steroid Biochem. Mol. Biol. 131, 122–131 (2012).
Karthaus, W.R. et al. Identification of multipotent luminal progenitor cells in human prostate organoid cultures. Cell 159, 163–175 (2014).
Gao, D. et al. Organoid cultures derived from patients with advanced prostate cancer. Cell 159, 176–187 (2014).
Sato, T. et al. Single Lgr5 stem cells build crypt-villus structures in vitro without a mesenchymal niche. Nature 459, 262–265 (2009).
Sato, T. et al. Long-term expansion of epithelial organoids from human colon, adenoma, adenocarcinoma, and Barrett's epithelium. Gastroenterology 141, 1762–1772 (2011).
Xin, L., Lukacs, R.U., Lawson, D.A., Cheng, D. & Witte, O.N. Self-renewal and multilineage differentiation in vitro from murine prostate stem cells. Stem Cells 25, 2760–2769 (2007).
Garraway, I.P. et al. Human prostate sphere-forming cells represent a subset of basal epithelial cells capable of glandular regeneration in vivo. Prostate 70, 491–501 (2010).
Niranjan, B. et al. Primary culture and propagation of human prostate epithelial cells. Methods Mol. Biol. 945, 365–382 (2013).
Höfner, T. et al. Defined conditions for the isolation and expansion of basal prostate progenitor cells of mouse and human origin. Stem Cell Reports 4, 503–518 (2015).
Liu, X. et al. ROCK inhibitor and feeder cells induce the conditional reprogramming of epithelial cells. Am. J. Pathol. 180, 599–607 (2012).
Chua, C.W. et al. Single luminal epithelial progenitors can generate prostate organoids in culture. Nat. Cell Biol. 16, 951–961 (2014).
Koo, B.K. et al. Controlled gene expression in primary Lgr5 organoid cultures. Nat. Methods 9, 81–83 (2011).
Schwank, G., Andersson-Rolf, A., Koo, B.K., Sasaki, N. & Clevers, H. Generation of BAC transgenic epithelial organoids. PLoS ONE 8, e76871 (2013).
Schwank, G. et al. Functional repair of CFTR by CRISPR/Cas9 in intestinal stem cell organoids of cystic fibrosis patients. Cell Stem Cell 13, 653–658 (2013).
Drost, J. et al. Sequential cancer mutations in cultured human intestinal stem cells. Nature 521, 43–47 (2015).
Lukacs, R.U., Goldstein, A.S., Lawson, D.A., Cheng, D. & Witte, O.N. Isolation, cultivation and characterization of adult murine prostate stem cells. Nat. Protoc. 5, 702–713 (2010).
Xin, L., Ide, H., Kim, Y., Dubey, P. & Witte, O.N. In vivo regeneration of murine prostate from dissociated cell populations of postnatal epithelia and urogenital sinus mesenchyme. Proc. Natl. Acad. Sci. USA 100, 11896–11903 (2003).
Kim, K.A. et al. Mitogenic influence of human R-spondin1 on the intestinal epithelium. Science 309, 1256–1259 (2005).
Acknowledgements
We thank members of the contributing laboratories for support. We are grateful for support from the following: The Netherlands Organisation for Scientific Research (NWO-ZonMw) VENI grant to J.D. (91614138); Koningin Wilhelmina Fonds (Dutch cancer foundation) to W.R.K. (2015-7545); the Prostate Cancer Foundation (C.L.S., Y.C.); and the CancerGenomics.nl (NWO Gravitation) program.
Author information
Authors and Affiliations
Contributions
J.D., W.R.K., Y.C., C.L.S. and H.C. conceived the study. J.D., W.R.K., D.G. and E.D. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Drost, J., Karthaus, W., Gao, D. et al. Organoid culture systems for prostate epithelial and cancer tissue. Nat Protoc 11, 347–358 (2016). https://doi.org/10.1038/nprot.2016.006
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2016.006
This article is cited by
-
Insights on Three Dimensional Organoid Studies for Stem Cell Therapy in Regenerative Medicine
Stem Cell Reviews and Reports (2024)
-
Exploring the promising potential of induced pluripotent stem cells in cancer research and therapy
Molecular Cancer (2023)
-
The role of organoids in cancer research
Experimental Hematology & Oncology (2023)
-
Gene expression in organoids: an expanding horizon
Biology Direct (2023)
-
The future of patient-derived xenografts in prostate cancer research
Nature Reviews Urology (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.