Abstract
Behavioral and pharmacotherapeutic approaches constitute two prominent strategies for treating cocaine dependence. This study investigated interactions between behavioral and pharmacological strategies in a preclinical model of cocaine vs food choice. Six rhesus monkeys, implanted with a chronic indwelling double-lumen venous catheter, initially responded under a concurrent schedule of food delivery (1-g pellets, fixed-ratio (FR) 100 schedule) and cocaine injections (0–0.1 mg/kg/injection, FR 10 schedule) during continuous 7-day treatment periods with saline or the agonist medication phenmetrazine (0.032–0.1 mg/kg/h). Subsequently, the FR response requirement for cocaine or food was varied (food, FR 100; cocaine, FR 1–100; cocaine, FR 10; food, FR 10–300), and effects of phenmetrazine on cocaine vs food choice were redetermined. Decreases in the cocaine FR or increases in the food FR resulted in leftward shifts in the cocaine choice dose-effect curve, whereas increases in the cocaine FR or decreases in the food FR resulted in rightward shifts in the cocaine choice dose-effect curve. The efficacy of phenmetrazine to decrease cocaine choice varied systematically as a function of the prevailing response requirements, such that phenmetrazine efficacy was greatest when cocaine choice was maintained by relatively low unit cocaine doses. These results suggest that efficacy of pharmacotherapies to modulate cocaine use can be influenced by behavioral contingencies of cocaine availability. Agonist medications may be most effective under contingencies that engender choice of relatively low cocaine doses.
Similar content being viewed by others
INTRODUCTION
The most recent report from the treatment episode data set estimates that 12.9% of substance abuse treatment admissions were for treatment of cocaine abuse (SAMHSA, 2011). These epidemiological data suggest that cocaine abuse remains a public health problem with no current Food and Drug Administration-approved pharmacotherapies. In general, there have been two main strategies deployed to reduce cocaine abuse. One category of strategies consists of behavioral approaches that share the general goal of reducing cocaine consumption by increasing the cost of cocaine relative to other commodities. These approaches include broad governmental policies (eg, law enforcement and taxation; Dave, 2006; Grossman, 2005; Grossman et al, 2002) as well as more targeted contingency management programs (Bigelow et al, 1981; Vocci and Montoya, 2009). The other category of strategies consists of pharmacological approaches, such as maintenance on an agonist-based pharmacotherapy, and clinical trials have demonstrated efficacy of maintenance on the dopamine/norepinephrine vs serotonin-selective monoamine releaser d-amphetamine to decrease cocaine abuse (Grabowski et al, 2001, 2004; Mooney et al, 2009). However, interactions between behavioral and pharmacological approaches in the treatment of cocaine abuse are poorly understood and understudied.
Both animal and human laboratory studies have demonstrated that simply introducing an alternative reinforcer can decrease cocaine self-administration (Carroll et al, 1989; Hart et al, 2000; Nader and Woolverton, 1991; Vosburg et al, 2010), analogous to the contingency management approaches used clinically to treat cocaine abuse. Furthermore, both preclinical and human laboratory studies have demonstrated that cocaine choice is sensitive to manipulations in the response requirement associated with cocaine or the alternative reinforcer. For example, increases in the response requirement for cocaine or decreases in the response requirement for an alternative reinforcer can reduce cocaine choice and increase choice of the alternative (Hart et al, 2000; Nader and Woolverton, 1992; Negus, 2003; Stoops et al, 2012). Reciprocally, increases in the response requirement for the alternative reinforcer or decreases in the response requirement for cocaine can increase cocaine choice and reduce choices for the alternative (Hart et al, 2000; Nader et al, 1992; Negus, 2003). Taken together, these preclinical and human laboratory studies agree that behavioral manipulations can modulate the allocation of choice between cocaine and alternative reinforcers.
Preclinical and human laboratory studies have also demonstrated that administration of some candidate pharmacotherapies can decrease cocaine self-administration. One example of this pharmacotherapy approach is studies with the dopamine/norepinephrine-selective releaser d-amphetamine. Chronic d-amphetamine treatment produced dose-dependent and sustained decreases in cocaine self-administration under second-order, progressive-ratio, and concurrent drug vs food schedules of reinforcement in rats (Chiodo et al, 2008; Thomsen et al, 2012), nonhuman primates (Czoty et al, 2011; Negus, 2003; Negus and Mello, 2003), and/or humans (Greenwald et al, 2010; Rush et al, 2010). Overall, these studies demonstrate that cocaine self-administration in general, and cocaine vs food choice in particular, is sensitive to pharmacological manipulations.
The aim of this study was to examine interactions between behavioral and pharmacological manipulations on cocaine vs food choice in rhesus monkeys. A concurrent cocaine vs food choice procedure was used for three reasons. First, the primary dependent measure (percent drug choice) is a measure of behavioral allocation rather than behavioral rate, and this type of dependent measure may be less sensitive to reinforcer-independent response rate-altering effects produced by the self-administered drug or candidate medication (Negus, 2003; Negus and Banks, 2011). Second, drug abuse can be defined as a disorder of choice (Heyman, 2009), and a goal of treatment is not only to reduce drug taking, but also to reallocate behavior to activities maintained by more adaptive reinforcers (Vocci, 2007). Finally, this procedure afforded the opportunity to introduce changes in the FR requirement for cocaine or food as behavioral manipulations. The dopamine/norepinephrine-selective releaser phenmetrazine was chosen as the agonist-based medication for two main reasons. First, we have previously shown that continuous 7-day phenmetrazine treatment decreased cocaine self-administration under a multiple second-order schedule of sequential cocaine and food availability and under a concurrent cocaine vs food choice procedure (Banks et al, 2011; Negus et al, 2009). Second, like d-amphetamine, phenmetrazine is a schedule II controlled substance that had a brief history of clinical use as an anorectic (Silverstone, 1992), and thus could be more readily extended to human laboratory or clinical trials than other experimental compounds that have not been approved for human use, such as m-fluoroamphetamine (also known as PAL-353; Banks et al, 2011; Negus et al, 2007). We hypothesized that phenmetrazine treatment would be most efficacious at reducing choice of low cocaine doses under behavioral conditions that favored choice of low as well as high cocaine doses over food (ie, when cocaine cost << food cost). Conversely, we hypothesized that phenmetrazine would be less effective under behavioral conditions that favored food choice, and under which cocaine choice was observed only at high cocaine doses.
MATERIALS AND METHODS
Animals
Studies were conducted in six adult male rhesus monkeys (Macaca mulatta) surgically implanted with a chronic indwelling double-lumen venous catheter inserted into a major vein (femoral or jugular) as described previously (Banks et al, 2011). The intravenous catheter was protected by a tether and jacket system (Lomir Biomedical, Malone, NY) that permitted monkeys to move freely in the cage. Catheter patency was periodically evaluated by intravenous (i.v.) administration of ketamine (3 mg/kg) or methohexital (2 mg/kg) through the catheter lumen and after each drug treatment that produced a rightward shift in the cocaine choice curve. The catheter was considered to be patent if i.v. administration of ketamine or methohexital produced a loss of muscle tone within 10 s. Monkeys could earn up to 50 1-g banana-flavored pellets (Grain-based Precision Primate Pellets; Test Diets, Richmond, IL) during daily experimental sessions (see below). In addition, monkeys also received daily rations of monkey biscuits (Lab Diet High Protein Monkey Biscuits; PMI Feeds, St Louis, MO). The size of these daily biscuit rations was individually determined for each monkey to maintain a healthy adult body weight as determined in collaboration with veterinary staff. These biscuit rations were delivered in the afternoons after choice sessions to minimize the effects of biscuit availability and biscuit consumption on food-maintained operant responding during choice sessions. Animals also received rations of fresh fruit 3–4 afternoons per week. Water was continuously available in each monkey's home cage, which also served as the experimental chamber (see below). A 12-h light/dark cycle was in effect (lights on from 0600 to 1800 h). Environmental enrichment consisting of foraging boards, novel treats, and video or radio programs was provided in the afternoons after behavioral sessions. All monkeys were experimentally naive at the beginning of the studies. All facilities were licensed by both the United States Department of Agriculture and the Association for Assessment and Accreditation of Laboratory Animal Care, and protocols were approved by the Institutional Animal Care and Use Committee.
Behavioral Procedures
Experimental sessions were conducted in each monkey’s home cage as previously described (Banks et al, 2011). Briefly, the front wall of the cage was equipped with a custom operant response panel that included three square response keys arranged horizontally. For this set of cocaine vs food choice studies, only responding on the left and right response keys had programmed consequences. Cages were also equipped with a pellet dispenser (Med Associates, ENV-203–1000, St Albans, VT) and two safety syringe pumps (Med Associates, PHM-108), one for each lumen of the double-lumen catheter. One pump (the ‘self-administration pump’) was used to deliver contingent cocaine injections through one lumen of the double-lumen catheter. The second pump (the ‘treatment pump’) was used to deliver saline or phenmetrazine through the second lumen. This ‘treatment’ pump was programmed to deliver 0.1 ml injections every 20 min from noon (1200 h) each day until 1100 h the next morning. Operation of operant response panels, pellet dispensers, and syringe pumps was accomplished with computers and software purchased from Med Associates.
Cocaine vs food choice sessions were conducted daily from 0900 h to 1100 h as previously described (Banks et al, 2011). The terminal choice schedule consisted of five 20-min components separated by 5-min timeout periods. During each component, the left, food-associated key was transilluminated red, and completion of the fixed-ratio (FR) requirement resulted in delivery of a 1-g food pellet. The right, cocaine-associated key was transilluminated green, and completion of the FR requirement resulted in delivery of the i.v. cocaine dose available during that component. The FR requirements for food and cocaine were manipulated as discussed below. A different cocaine dose was available during each of the five successive components (0, 0.0032, 0.01, 0.032, and 0.1 mg/kg/inj during components 1–5, respectively), and the cocaine dose was varied by manipulating the duration and resulting volume (0, 0.01, 0.03, 0.1, and 0.3 ml/inj, respectively) of each injection. Stimulus conditions on the cocaine-associated key were also varied by flashing the stimulus lights on and off in 3 s cycles, and longer flashes (with shorter interflash intervals) were associated with higher cocaine doses.
During each component, monkeys could complete up to 10 ratio requirements on the food- and cocaine-associated keys. Responding on either key reset the ratio requirement on the other key. Completion of each ratio requirement initiated a 3 s timeout, during which all stimulus lights were turned off, and responding had no scheduled consequences. If the maximum number of ratio requirements was completed before the 20-min component had elapsed, then all stimulus lights were extinguished, and responding had no scheduled consequences for the remainder of that component.
During initial training, saline was delivered through the ‘treatment’ lumen, and the response requirements were initially set at FR 100 on the food-associated key and FR 10 on the cocaine-associated key for all monkeys consistent with previous studies from our laboratory (Banks et al, 2011; Banks and Negus, 2010; Negus, 2003). Choice behavior under these conditions was considered to be stable when the lowest cocaine dose maintaining at least 80% cocaine choice varied by ⩽0.5 log units for three consecutive days. Once choice behavior stabilized by this criterion, test sessions were initially conducted to determine effects of continuous 7-day treatment with (+)-phenmetrazine (0.032–0.32 mg/kg/h) on cocaine choice. Phenmetrazine doses were examined in a counterbalanced order between monkeys, and at the conclusion of each 7-day treatment period, saline control treatment was reinstituted for at least 4 days and until cocaine choice returned and stabilized to pretest levels. As noted below, the high dose of 0.32 mg/kg/h produced evidence of nonselective behavioral disruption; consequently, this dose was studied in only four monkeys under baseline cocaine and food FR conditions, and it was not studied further. Next, FR manipulations were introduced on the food- or cocaine-associated key for at least 7 days and until choice behavior was stable. Specifically, from the baseline parameters of food FR 100/cocaine FR 10, either the food FR was held constant at FR 100 and the cocaine FR was decreased (FR 1) or increased (FR 100), or the cocaine FR was held constant at FR 10 and the food FR was decreased (FR 10) or increased (FR 300). After choice behavior was stable as defined earlier at a given FR manipulation, the effects of continuous 7-day treatment with phenmetrazine (0.032–0.1 mg/kg/h) were redetermined. Once both phenmetrazine doses had been determined at a given FR manipulation, baseline FR requirements (food FR 100/cocaine FR 10) were reinstituted until choice behavior returned to pre-FR manipulation conditions. A new FR manipulation was then instituted, and phenmetrazine effects were examined again. The sequences of FR manipulations and phenmetrazine doses were counterbalanced between monkeys. However, all FR manipulations alone and in combination with phenmetrazine for one reinforcer were determined before determining the effects of FR manipulations and phenmetrazine treatment on the other reinforcer.
Data Analysis
The primary dependent variables for each component were: (1) percent cocaine choice, defined as (number of ratio requirements (choices) completed on the cocaine-associated key÷total number of ratio requirements (choices) completed on both the cocaine- and food-associated keys) × 100, and (2) total number of ratio requirements (choices) completed from the last 3 days of each 7-day treatment period. These variables were then plotted as a function of cocaine dose. Cocaine choice dose-effect curves were analyzed using a two-way repeated-measures ANOVA with cocaine dose and treatment condition as the main factors. Additional dependent variables collected during each session included total choices, total food choices, and total cocaine choices. A significant ANOVA was followed by the Dunnett’s multiple comparisons post hoc test to compare test conditions with baseline conditions. The criterion for significance was set a priori at the 95% level of confidence (p⩽0.05). In addition, the ED50 value of the cocaine choice dose-effect curve was defined as the dose of cocaine that produced 50% cocaine choice. ED50 values were calculated for each monkey under each set of experimental conditions by interpolation when only two data points were available (one below and one above 50% cocaine choice) or by linear regression when at least three data points were available on the linear portion of the dose-effect curve. Log ED50 values were calculated for each monkey during (1) the last 3 ‘baseline’ days before introduction of each phenmetrazine treatment manipulation and (2) the last 3 days of each 7-day phenmetrazine treatment period. In cases where log ED50 values could not be determined because cocaine choice was greater or less than 50% for all cocaine doses, conservative estimates of the cocaine-choice ED50 were determined by assuming 100% cocaine choice at the next higher half-log dose (0.32 mg/kg/injection) or 0% cocaine choice at the next lower half-log dose (0.001 mg/kg/injection).
Drugs
Cocaine HCl was provided by the National Institute on Drug Abuse Drug Supply Program (Bethesda, MD). (+)-Phenmetrazine fumarate was provided by BE Blough (Research Triangle Institute, Research Triangle Park, NC). All drugs were dissolved in sterile water, and all solutions were filter-sterilized using a 0.22-μm Millipore filter (Millipore, Billerica, MA). Drug doses were calculated using the salt forms listed above.
RESULTS
Effects of Phenmetrazine Treatment on Cocaine vs Food Choice Under Baseline FR Conditions
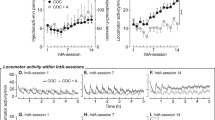
Under baseline conditions of saline treatment, food FR 100, and cocaine FR 10, monkeys primarily chose food when the unit dose of cocaine was low (0–0.01 mg/kg/injection) and almost exclusively reallocated their behavior to the cocaine-associated key during availability of higher cocaine unit doses (0.032–0.1 mg/kg/injection) (Figure 1; open circles). Figure 1 also shows the effects of phenmetrazine on cocaine choice under baseline food and cocaine FR conditions. Two-way repeated-measures ANOVA on percent cocaine choice demonstrated a significant main effect of cocaine dose (F4, 12=129.7, p<0.05), and a significant cocaine dose and phenmetrazine treatment interaction (F12, 33=2.7, p<0.05). The post hoc analysis revealed that treatment with 0.32 mg/kg/h phenmetrazine significantly decreased choice of 0.032 mg/kg/injection cocaine. For number of choices completed per component, there was a significant main effect of phenmetrazine treatment (F3, 9=6.0, p<0.05). The post hoc analysis revealed that treatment with 0.32 mg/kg/h phenmetrazine significantly decreased the number of choices completed per component when 0.0032 or 0.01 mg/kg/injection cocaine was available as the alternative to food. Furthermore, the high 0.32 mg/kg/h phenmetrazine dose was only tested in four monkeys under baseline FR conditions because it significantly decreased the total number of choices completed and produced stereotypic behaviors in some monkeys. Subsequent studies examined only the effects of the lower phenmetrazine doses of 0.032 and 0.1 mg/kg/h.
Effects of continuous 7-day treatment with phenmetrazine (0.032–0.32 mg/kg/h) on choice between cocaine and food under baseline FR conditions (Food FR 100/Coc FR 10) in rhesus monkeys (n=6), except for the 0.32 mg/kg/h phenmetrazine dose, which was tested in four monkeys. Numbers in parentheses indicate the number of subjects contributing to that data point for the 0.32/h phenmetrazine treatment; responding in the remaining monkey was eliminated. Abscissae: unit dose of cocaine in mg per kg per injection. Top ordinate: percent cocaine choice. Bottom ordinate: number of ratio requirements (choices) completed per component. All points represent mean data±SEM obtained during the last 3 days of each 7-day treatment. Filled symbols indicate statistically significant (p⩽0.05) compared with the saline condition within a given unit cocaine dose.
Effects of FR Manipulations on the Cocaine- or Food-Associated Key on Cocaine vs Food Choice
Manipulations of the FR requirement on the cocaine-associated key produced significant leftward shifts (FR 1) and rightward shifts (FR 100) in the cocaine choice dose-effect function (Figure 2). Two-way repeated-measures ANOVA on percent cocaine choice demonstrated a significant main effect of cocaine dose (F4, 12=379.4, p<0.05), FR manipulation (F2, 6=31.9, p<0.05), and a significant cocaine dose and FR manipulation interaction (F8, 24=16.5, p<0.05). The post hoc analysis revealed that decreasing the FR to 1 on the cocaine-associated key significantly increased cocaine choice at the 0.01 mg/kg/injection cocaine dose, and increasing the FR to 100 significantly decreased cocaine choice at the 0.032 mg/kg/injection cocaine dose compared with the baseline condition of cocaine FR 10. For the number of choices completed per component, there was a significant cocaine dose and FR manipulation interaction (F8, 24=2.9, p<0.05). The post hoc analysis revealed that decreasing the FR from 10 to 1 significantly increased the number of choices completed when the 0.032 mg/kg/injection cocaine dose was available, and increasing the FR from 10 to 100 significantly decreased the number of choices completed during the 0.1 mg/kg/injection cocaine dose component.
Effects of FR manipulations on the cocaine- or food-associated key on choice between cocaine and food in rhesus monkeys. Abscissae: unit dose of cocaine in mg per kg per injection. Top ordinates: percent cocaine choice. Bottom ordinates: the number of ratio requirements (choices) completed per component. All points represent mean data±SEM obtained during the 3 days preceding each phenmetrazine treatment. Filled symbols indicate statistically significant (p⩽0.05) compared with the baseline FR condition (cocaine FR 10, food FR 100) within a given unit cocaine dose.
Manipulations of the FR requirement on the food-associated key also produced significant shifts in the cocaine choice dose-effect function (Figure 2). Two-way repeated-measures ANOVA on percent cocaine choice demonstrated a significant main effect of cocaine dose (F4, 16=100.8, p<0.05), FR manipulation (F2, 8=9.6, p<0.05), and a significant interaction (F8, 32=8.3, p<0.05). The post hoc analysis revealed that increasing the food FR to 300 significantly increased choice of the 0.01 mg/kg/injection unit cocaine dose and that decreasing the food FR to 10 significantly decreased choice of the 0.032 mg/kg/injection unit cocaine dose compared with the baseline food FR 100 condition. For the number of choices completed per component, there was a significant main effect of cocaine dose (F4, 16=4.6, p<0.05), FR manipulation (F2, 8=18.2, p<0.05), and a significant interaction (F8, 32=7.9, p<0.05). The post hoc analysis revealed that increasing the food FR to 300 significantly decreased the number of choices completed per component during the first 3 components of the session when low unit cocaine doses (0–0.01 mg/kg/injection) were available as the alternative to food.
Effects of Phenmetrazine Treatment Under Conditions of Varied Cocaine FRs
The effects of phenmetrazine treatment under conditions of varied cocaine FR are shown in Figure 3. When phenmetrazine was examined under conditions where the cocaine FR was decreased from 10 to 1, two-way repeated-measures ANOVA for percent cocaine choice demonstrated a significant main effects of cocaine dose (F4, 12=156.3, p<0.05) and phenmetrazine treatment (F2, 6=5.1, p=0.05), but no significant interaction (F8, 24=2.1, p=0.07). The post hoc analysis revealed that both phenmetrazine doses significantly decreased choice of 0.01 mg/kg/injection cocaine, and 0.1 mg/kg/h phenmetrazine also significantly decreased choice of 0.032 mg/kg/injection cocaine. For the number of choices completed per component, there was no statistically significant effect.
Effects of continuous 7-day treatment with phenmetrazine (0.032–0.1 mg/kg/h) on choice between cocaine and food under different cocaine FR conditions. Abscissae: unit dose of cocaine in mg per kg per injection. Top ordinates: percent cocaine choice. Bottom ordinates: the number of ratio requirements (choices) completed per component. All points represent mean data±SEM obtained during the last 3 days of each 7-day treatment from four monkeys. Filled symbols indicate statistically significant (p⩽0.05) compared with the saline condition within a given unit cocaine dose. The graphs for the baseline FR manipulations are the same data as in Figure 1.
When phenmetrazine was examined under conditions where the cocaine FR was increased from FR 10 to FR 100 (Figure 3), two-way repeated-measures ANOVA on cocaine choice revealed a significant main effect of cocaine dose (F4, 12=81.0, p<0.05). For the number of choices completed per component, there was also a main effect of cocaine dose (F4, 12=4.8, p<0.05).
Effects of Phenmetrazine Treatment Under Conditions of Varied Food FRs
When the food FR was increased from FR 100 to FR 300 (Figure 4), two-way repeated-measures ANOVA on percent cocaine choice demonstrated a main effect of cocaine dose (F4, 16=38.7, p<0.05), phenmetrazine treatment (F2, 8=10.5, p<0.05), and a significant interaction (F8, 32=2.9, p<0.05). The post hoc analysis revealed that both doses of phenmetrazine significantly decreased choice of 0.01 mg/kg/injection cocaine. For the number of choices completed per component, there was a significant main effect of cocaine dose (F4, 16=6.6, p<0.05).
Effects of continuous 7-day treatment with phenmetrazine (0.032–0.1 mg/kg/h) on choice between cocaine and food under different food FR conditions. Abscissae: unit dose of cocaine in mg per kg per injection. Top ordinates: percent cocaine choice. Bottom ordinates: the number of ratio requirements (choices) completed per component. All points represent mean data±SEM obtained during the last 3 days of each 7-day treatment. The food FR 100 and FR 10 conditions represent data from six monkeys, whereas the food FR 300 condition represents data from five monkeys. Filled symbols indicate statistically significant (p⩽0.05) compared with the saline condition within a given unit cocaine dose. The graphs for the baseline FR manipulations are the same data as in Figure 1.
When the food FR was decreased from FR 100 to FR 10 (Figure 4), two-way repeated-measures ANOVA demonstrated a significant main effect of cocaine dose (F4, 20=37.3, p<0.05). For the number of choices completed per component, there was no statistically significant effect.
Effects of Phenmetrazine Treatment on Total Session, Food, and Cocaine Choices
Figure 5 shows the effects of phenmetrazine treatment under the different FR manipulations on mean numbers of total choices, food choices, and cocaine choices completed during each session. Under baseline FR conditions (food FR 100, cocaine FR 10), two-way repeated-measures ANOVA demonstrated a significant main effect of experimental end point (F2, 10=210.1, p<0.05). As noted above for Figure 1, there was a trend for the high dose of 0.32 mg/kg/h phenmetrazine to decrease cocaine choice, but this dose also decreased food choices and total choices in some monkeys, and was not studied further. When the cocaine FR was decreased from 10 to 1, two-way repeated-measures ANOVA demonstrated a significant main effect of experimental end point (F2, 10=9.3, p<0.05) and a significant interaction (F4, 20=9.3, p<0.05). The post hoc analysis revealed that treatment with 0.1 mg/kg/h phenmetrazine significantly increased food choices and significantly decreased cocaine choices compared with saline conditions. When the cocaine FR was increased from 10 to 100, two-way repeated-measures ANOVA demonstrated a significant main effect of experimental end point (F2, 10=9.7, p<0.05). When the food FR was increased from 100 to 300, two-way repeated-measures ANOVA demonstrated a significant main effect of experimental end point (F2, 10=11.8, p<0.05) and a significant interaction (F4, 20=3.7, p<0.05). The post hoc analysis revealed that treatment with 0.032 mg/kg/h phenmetrazine significantly increased total and food choices, and treatment with 0.1 mg/kg/h phenmetrazine significantly increased food choices. No dose of phenmetrazine significantly decreased cocaine choices. When the food FR was decreased from 100 to 10, two-way repeated-measures ANOVA demonstrated a significant main effect of experimental end point (F2, 10=111.3, p<0.05).
Effects of continuous 7-day phenmetrazine treatment (0.032–0.32 mg/kg/h) on total, food, and cocaine choices under different cocaine and food FR conditions. Abscissae: experimental end point. Ordinates: number of ratio requirements (choices) completed per session. All bars represent mean data±SEM obtained during the last 3 days of each phenmetrazine treatment or the 3 days preceding each phenmetrazine treatment. *p⩽0.05, significantly different from the saline condition.
Effects of Experimental Manipulations on Cocaine Choice ED50 Values
Phenmetrazine treatment under baseline FR conditions did not significantly increase the cocaine choice ED50 value (Supplementary Table S1). Also, consistent with our previous study examining phenmetrazine effects on cocaine choice (Banks et al, 2011), individual differences were noted (Supplementary Figure S1). In five out of six monkeys tested, at least one phenmetrazine dose increased the cocaine choice ED50 value. Manipulations of the FR requirement alone on the cocaine-associated key produced significant decreases (cocaine FR 1) and increases (cocaine FR 100) in the cocaine choice ED50 values, and these effects were consistent between monkeys (Supplementary Table S1; Supplementary Figure S1). When phenmetrazine was examined under the different cocaine FR manipulations, 0.1 mg/kg/h phenmetrazine under the cocaine FR 1 condition significantly increased the cocaine choice ED50 value (Supplementary Table S1). Manipulations of the FR requirement alone on the food-associated key also produced decreases (food FR 300) in the cocaine choice ED50 value (Supplementary Table S1); however, these effects were more variable between monkeys compared with manipulations of the cocaine FR requirement (Supplementary Figure S1). No dose of phenmetrazine under any food FR condition had a significant effect on the cocaine choice ED50 value.
DISCUSSION
This study examined the interaction between behavioral and pharmacological manipulations in a nonhuman primate model of cocaine and food choice. There were three main findings. First, the efficacy of phenmetrazine to decrease cocaine choice under baseline FR conditions was similar to the results of our previous study examining effects of different monoamine releasers with varying pharmacological selectivity to release dopamine/norepinephrine vs serotonin (Banks et al, 2011). Second, also in agreement with previous studies (Nader et al, 1992; Negus, 2003; Woolverton and English, 1997), cocaine choice could be modulated by changes in the response requirement for cocaine relative to the alternative food reinforcer. Finally, cocaine choice was most sensitive to phenmetrazine under conditions when the relative response requirement for cocaine was low, the cocaine choice dose-effect curve was shifted leftward, and cocaine choice was maintained by low cocaine doses. Conversely, cocaine choice was insensitive to phenmetrazine treatment when the relative response requirement for cocaine was high, the cocaine choice dose-effect curve was shifted rightward, and cocaine choice was maintained only by high cocaine doses. Overall, these preclinical findings suggest that the efficacy of candidate pharmacotherapies to decrease cocaine choice and produce a reciprocal increase in choice of an alternative nondrug reinforcer is influenced by the contingencies of cocaine availability.
Baseline Cocaine Choice and Effects of FR Manipulations
Intravenous cocaine maintained a dose-dependent increase in choice vs an alternative food reinforcer, consistent with previous studies by our laboratory and others (Banks et al, 2011; Czoty et al, 2005; Nader et al, 1992; Negus, 2003). As expected, manipulations in the response requirement that increased the relative price of cocaine injections vs food pellets significantly decreased cocaine choice, whereas response requirement manipulations that decreased the relative price of cocaine injections vs food pellets significantly increased cocaine choice. These results are consistent with previous nonhuman primate (Czoty et al, 2005; Nader et al, 1992; Negus, 2003; Woolverton et al, 1997) and human laboratory (Greenwald and Hursh, 2006; Stoops et al, 2012) studies examining the behavioral determinants of drug choice. Moreover, our preclinical results may be analogous to epidemiological evidence for the price sensitivity of drug consumption, which provides a rationale for both governmental policies and more targeted contingency management strategies designed to reduce drug consumption by increasing drug price or facilitating access to nondrug alternatives (Dunlop et al, 2011; Grossman, 2005; Xu and Chaloupka, 2011). Notably, though, in the present study relatively large changes in the response requirement produced relatively modest shifts in cocaine choice. For example, a 100-fold increase in the cocaine FR from FR 1 to FR 100 was associated with only a 3.7-fold increase in the cocaine choice ED50 value. Moreover, this 100-fold increase in cocaine price was not sufficient to decrease choice of the highest cocaine dose (0.1 mg/kg/injection), and is consistent with previous results from our laboratory where the cocaine FR had to be increased to 1000 to significantly decrease choice maintained by this cocaine dose (Negus, 2003). Thus, although behavioral manipulations can clearly influence drug consumption, the cost to society (eg, for enforcement of drug laws) or to individuals (eg, for contingency management services) for achieving and maintaining increases in drug price sufficient to reduce consumption can be substantial (Chisholm et al, 2006). The challenges associated with behavioral approaches to controlling drug consumption provide one rationale for exploring pharmacotherapies as an alternative and/or complementary approach.
Effects of Phenmetrazine on Cocaine Choice
Under baseline FR conditions (FR 100 food; FR 10 cocaine), phenmetrazine produced a significant decrease in cocaine choice, but only at a phenmetrazine dose that also significantly disrupted overall rates of behavior. Nonetheless, at least one dose of phenmetrazine did increase the cocaine choice ED50 value in five out of the six monkeys under these baseline conditions. Moreover, the results of this study are in agreement with earlier findings (Banks et al, 2011; Negus, 2003) that phenmetrazine and other dopamine/norepinephrine-selective releasers display modest but significant efficacy for decreasing cocaine choice. When the efficacy of phenmetrazine to decrease cocaine choice was examined under different behavioral conditions, phenmetrazine was most efficacious in decreasing cocaine choice when the FR requirement was 1 for cocaine or the FR requirement was 300 for food, and cocaine choice was consequently maintained by relatively low cocaine doses.
This is the first study to evaluate the effects of a candidate pharmacotherapy on cocaine vs food choice under different behavioral conditions; however, the results of this study agree with previous studies using other schedules of reinforcement that self-administration of lower unit doses of cocaine is more vulnerable than responding maintained by higher cocaine doses to various manipulations including treatment with candidate medications. For example, both the selective dopamine uptake inhibitor GBR12909 (Glowa et al, 1995) and the monoamine releaser d-amphetamine (Negus et al, 2003) were more potent to reduce self-administration maintained by lower than higher unit doses of cocaine under multiple schedules of sequential cocaine and food availability in rhesus monkeys.
Unit price is a term related to unit dose that has also been invoked as a potentially important determinant of drug self-administration (Hursh, 1993). Unit price in preclinical drug self-administration studies is defined as the ratio of response requirement (eg, FR value) divided by unit dose, and unit price analysis theorizes that drug consumption will be constant for any combinations of response requirement and unit dose that yield identical unit prices. For example, unit price analysis predicts identical levels of drug consumption for a given unit dose of drug at a given FR value (eg, 0.01 mg/kg/inj at FR 1) and a 10-fold higher unit dose of the same drug at a 10-fold higher FR value (eg, 0.1 mg/kg at FR 10), because in both cases, the unit price is the same (unit price=100). Experimental data generally support the predictions of unit price analysis for self-administration of cocaine and other drugs, although responding maintained by low cocaine doses was more sensitive to increases in response requirement (ie, more ‘elastic’) than would be predicted by simple unit price analysis (Hursh and Winger, 1995). Unit price has also been considered as a determinant of pharmacotherapy effects on drug self-administration. For example, one study concluded that efficacy of GBR12909 to reduce cocaine self-administration increased as the unit price for cocaine increased (Stafford et al, 2000). However, this positive correlation between GBR12909 efficacy and cocaine unit price was apparent in only two of three monkeys tested, and additionally, ‘high prices’ were usually achieved by using small unit doses rather than by using large response requirements. In this study, unit price was not a predictor of phenmetrazine efficacy to reduce cocaine choice. For example, phenmetrazine effectively reduced choice of 0.01 mg/kg/inj cocaine under a FR 1 response requirement (unit price=100), but it did not affect choice of a 10-fold higher dose of 0.1 mg/kg/inj cocaine under FR requirements up to FR 100 (unit prices up to 1000). Overall, unit cocaine dose appeared to be more important than unit cocaine price as a determinant of phenmetrazine effects on cocaine choice.
Implications
Results of this study demonstrate that both behavioral and pharmacological manipulations can significantly decrease cocaine choice and increase choice of an alternative nondrug reinforcer. Furthermore, the circumstances under which behavioral or pharmacological strategies reduce cocaine choice may have broader implications for preclinical research and medications development for cocaine dependence. First, a main goal of any treatment strategy should be to not only decrease drug-taking behavior, but also reallocate behavior to activities maintained by more adaptive reinforcers (Vocci, 2007). Thus, in preclinical studies that aspire to evaluate pharmacological or behavioral treatment strategies for drug dependence, choice procedures can play a critical role in the preclinical evaluation process to determine whether a given experimental manipulation produces this critical reallocation of behavior (Haney and Spealman, 2008; Negus et al, 2011). Second, although dopamine/norepinephrine-selective releasers have consistently demonstrated efficacy to decrease cocaine choice in nonhuman primates (Banks et al, 2011; Negus, 2003; present study), human laboratory studies (Greenwald et al, 2010; Rush et al, 2010), and clinical trials (Grabowski et al, 2001; Mooney et al, 2009), the magnitude of this therapeutic effect is limited by untoward effects and lower than the magnitude of effect that can potentially be achieved with behavioral economic approaches. However, the present results suggest that phenmetrazine or related compounds may nonetheless be therapeutically useful under circumstances in which cocaine is relatively inexpensive, cocaine consumption is maintained by relatively low cocaine doses, and behavioral options are limited. Furthermore, although phenmetrazine efficacy was reduced in the presence of effective behavioral manipulations, phenmetrazine treatment also did not lessen the effect of the behavioral manipulations, nor did phenmetrazine increase cocaine choice. Thus, combining behavioral and pharmacological strategies may be useful under specific clinical conditions. Human laboratory and clinical studies are clearly needed to more accurately define the specific clinical conditions where this combination approach may be most effective.
References
Banks ML, Blough BE, Negus SS (2011). Effects of monoamine releasers with varying selectivity for releasing dopamine/norepinephrine versus serotonin on choice between cocaine and food in rhesus monkeys. Behav Pharmacol 22: 824–836.
Banks ML, Negus SS (2010). Effects of extended cocaine access and cocaine withdrawal on choice between cocaine and food in rhesus monkeys. Neuropsychopharmacology 35: 493–504.
Bigelow GE, Stitzer ML, Griffiths RR, Liebson IA (1981). Contingency management approaches to drug self-administration and drug abuse: efficacy and limitations. Addict Behav 6: 241–252.
Carroll ME, Lac ST, Nygaard SL (1989). A concurrently available nondrug reinforcer prevents the acquisition or decreases the maintenance of cocaine-reinforced behavior. Psychopharmacology (Berl) 97: 23–29.
Chiodo K, Läck C, Roberts D (2008). Cocaine self-administration reinforced on a progressive ratio schedule decreases with continuous d-amphetamine treatment in rats. Psychopharmacology (Berl) 200: 465–473.
Chisholm D, Doran C, Shibuya K, Rehm J (2006). Comparative cost-effectiveness of policy instruments for reducing the global burden of alcohol, tobacco and illicit drug use. Drug Alcohol Rev 25: 553–565.
Czoty PW, Gould RW, Martelle JL, Nader MA (2011). Prolonged attenuation of the reinforcing strength of cocaine by chronic d-amphetamine in rhesus monkeys. Neuropsychopharmacology 36: 539–547.
Czoty PW, McCabe C, Nader MA (2005). Assessment of the relative reinforcing strength of cocaine in socially housed monkeys using a choice procedure. J Pharmacol Exp Ther 312: 96–102.
Dave D (2006). The effects of cocaine and heroin price on drug-related emergency department visits. J Health Econ 25: 311–333.
Dunlop SM, Perez D, Cotter T (2011). Australian smokers’ and recent quitters’ responses to the increasing price of cigarettes in the context of a tobacco tax increase. Addiction 106: 1687–1695.
Glowa JR, Wojnicki FHE, Matecka D, Bacher JD, Mansbach RS, Balster RL et al (1995). Effects of dopamine reuptake inhibitors on food- and cocaine-maintained responding: I. Dependence on unit dose of cocaine. Exp Clin Psychopharmacol 3: 219–231.
Grabowski J, Rhoades H, Schmitz J, Stotts A, Daruzska LA, Creson D et al (2001). Dextroamphetamine for cocaine-dependence treatment: a double-blind randomized clinical trial. J Clin Psychopharmacol 21: 522–526.
Grabowski J, Rhoades H, Stotts A, Cowan K, Kopecky C, Dougherty A et al (2004). Agonist-like or antagonist-like treatment for cocaine dependence with methadone for heroin dependence: two double-blind randomized clinical trials. Neuropsychopharmacology 29: 969–981.
Greenwald MK, Hursh SR (2006). Behavioral economic analysis of opioid consumption in heroin-dependent individuals: effects of unit price and pre-session drug supply. Drug Alcohol Depend 85: 35–48.
Greenwald MK, Lundahl LH, Steinmiller CL (2010). Sustained release d-amphetamine reduces cocaine but not “speedball”-seeking in buprenorphine-maintained volunteers: a test of dual-agonist pharmacotherapy for cocaine/heroin polydrug abusers. Neuropsychopharmacology 35: 2624–2637.
Grossman M (2005). Individual behaviours and substance use: the role of price. Adv Health Econ Health Serv Res 16: 15–39.
Grossman M, Chaloupka FJ, Shim K (2002). Illegal drug use and public policy. Health Aff (Millwood) 21: 134–145.
Haney M, Spealman R (2008). Controversies in translational research: drug self-administration. Psychopharmacology (Berl) 199: 403–419.
Hart CL, Haney M, Foltin RW, Fischman MW (2000). Alternative reinforcers differentially modify cocaine self-administration by humans. Behav Pharmacol 11: 87–91.
Heyman GH (2009) Addiction: A Disorder of Choice. Harvard University Press: Cambridge.
Hursh SR (1993). Behavioral economics of drug self-administration: an introduction. Drug Alcohol Depend 33: 165–172.
Hursh SR, Winger G (1995). Normalized demand for drugs and other reinforcers. J Exp Anal Behav 64: 373–384.
Mooney ME, Herin DV, Schmitz JM, Moukaddam N, Green CE, Grabowski J (2009). Effects of oral methamphetamine on cocaine use: a randomized, double-blind, placebo-controlled trial. Drug Alcohol Depend 101: 34–41.
Nader MA, Woolverton WL (1991). Effects of increasing the magnitude of an alternative reinforcer on drug choice in a discrete-trials choice procedure. Psychopharmacology (Berl) 105: 169–174.
Nader MA, Woolverton WL (1992). Effects of increasing response requirement on choice between cocaine and food in rhesus monkeys. Psychopharmacology (Berl) 108: 295–300.
Negus SS (2003). Rapid assessment of choice between cocaine and food in rhesus monkeys: effects of environmental manipulations and treatment with d-amphetamine and flupenthixol. Neuropsychopharmacology 28: 919–931.
Negus SS, Banks ML (2011). Making the right choice: lessons from drug discrimination for research on drug reinforcement and drug self-administration. In: Glennon RA, Young R, (eds). Drug Discrimination: Applications to Medicinal Chemistry and Drug Studies 1st edn. John Wiley and Sons: Holboken, NJ. pp 361–388.
Negus SS, Baumann MH, Rothman RB, Mello NK, Blough BE (2009). Selective suppression of cocaine- versus food-maintained responding by monoamine releasers in rhesus monkeys: benzylpiperazine, (+)phenmetrazine, and 4-benzylpiperidine. J Pharmacol Exp Ther 329: 272–281.
Negus SS, Mello NK (2003). Effects of chronic d-amphetamine treatment on cocaine- and food-maintained responding under a second-order schedule in rhesus monkeys. Drug Alcohol Depend 70: 39–52.
Negus SS, Mello NK, Blough BE, Baumann MH, Rothman RB (2007). Monoamine releasers with varying selectivity for dopamine/norepinephrine versus serotonin release as candidate “agonist” medications for cocaine dependence: studies in assays of cocaine discrimination and cocaine self-administration in rhesus monkeys. J Pharmacol Exp Ther 320: 627–636.
Rush CR, Stoops WW, Sevak RJ, Hays LR (2010). Cocaine choice in humans during d-amphetamine maintenance. J Clin Psychopharmacol 30: 152–159.
SAMHSA (2011) Results from the 2010 National Survey on Drug Use and Health: Summary of National Findings. Substance Abuse and Mental Health Services Administration: Rockville.
Silverstone T (1992). Appetite suppressants. A review. Drugs 43: 820–836.
Stafford D, Rice KC, Lewis DB, Glowa JR (2000). Response requirements and unit dose modify the effects of GRB 12909 on cocaine-maintained behavior. Exp Clin Psychopharmacol 8: 539–548.
Stoops WW, Lile JA, Glaser PE, Hays LR, Rush CR (2012). Alternative reinforcer response cost impacts cocaine choice in humans. Prog Neuropsychopharmacol Biol Psychiatry 36: 189–193.
Thomsen M, Barrett A, Negus SS, Caine SB (2012). Cocaine - food choice in rats: environmental manipulations and effects of amphetamine. Behav Pharmacol (in press).
Vocci FJ (2007). Can replacement therapy work in the treatment of cocaine dependence? And what are we replacing anyway? Addiction 102: 1888–1889.
Vocci FJ, Montoya ID (2009). Psychological treatments for stimulant misuse, comparing and contrasting those for amphetamine dependence and those for cocaine dependence. Curr Opin Psychiatry 22: 263–268.
Vosburg SK, Haney M, Rubin E, Foltin RW (2010). Using a novel alternative to drug choice in a human laboratory model of a cocaine binge: a game of chance. Drug Alcohol Depend 110: 144–150.
Woolverton WL, English JA (1997). Further analysis of choice between cocaine and food using the unit price model of behavioral economics. Drug Alcohol Depend 49: 71–78.
Xu X, Chaloupka FJ (2011). The effects of prices on alcohol use and its consequences. Alcohol Res Health 34: 236–245.
Acknowledgements
We appreciate the technical assistance of Jennifer Gough and Crystal Reyns. This work was funded by National Institutes of Heath Grants R01-DA26946 and R01-DA012790 from the National Institute on Drug Abuse, National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Dr Banks declares that during the past 3 years, he has received compensation as a collaborator with the pharmaceutical companies Abbott and Purdue for projects related to opioid pharmacology and analgesic drug development. Dr Negus declares that during the past 3 years, he has received compensation as a consultant for or collaborator with the pharmaceutical companies Abbott, Alkermes, Argolyn, Grunenthal, and Limerick Biopharma for projects related to opioid pharmacology, analgesic drug development, or assessment of abuse liability. Both authors declare that the present study was not related to this professional relationship and should not be perceived as constituting a potential conflict of interest. Dr Blough declares no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Neuropsychopharmacology website
Supplementary information
Rights and permissions
About this article
Cite this article
Banks, M., Blough, B. & Stevens Negus, S. Interaction Between Behavioral and Pharmacological Treatment Strategies to Decrease Cocaine Choice in Rhesus Monkeys. Neuropsychopharmacol 38, 395–404 (2013). https://doi.org/10.1038/npp.2012.193
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/npp.2012.193
Keywords
This article is cited by
-
The dopamine, serotonin and norepinephrine releasing activities of a series of methcathinone analogs in male rat brain synaptosomes
Psychopharmacology (2019)
-
Repeated 7-Day Treatment with the 5-HT2C Agonist Lorcaserin or the 5-HT2A Antagonist Pimavanserin Alone or in Combination Fails to Reduce Cocaine vs Food Choice in Male Rhesus Monkeys
Neuropsychopharmacology (2017)
-
Effects of Environmental Manipulations and Treatment with Bupropion and Risperidone on Choice between Methamphetamine and Food in Rhesus Monkeys
Neuropsychopharmacology (2015)
-
Use of Preclinical Drug Vs. Food Choice Procedures to Evaluate Candidate Medications for Cocaine Addiction
Current Treatment Options in Psychiatry (2015)
-
Effects of the dopamine/norepinephrine releaser phenmetrazine on cocaine self-administration and cocaine-primed reinstatement in rats
Psychopharmacology (2015)