Abstract
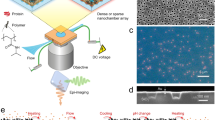
The selectivity and speed of many biological transport processes1 transpire from a ‘reduction of dimensionality’2 that confines diffusion to one or two dimensions instead of three3. This behaviour remains highly sought after on polymeric surfaces4 as a means to expedite diffusional search processes in molecular engineered systems. Here, we have reconstituted the two-dimensional diffusion of colloidal particles on a molecular brush surface. The surface is composed of phenylalanine-glycine nucleoporins (FG Nups)5—intrinsically disordered proteins that facilitate selective transport through nuclear pore complexes in eukaryotic cells6. Local and ensemble-level experiments involving optical trapping using a photonic force microscope7 and particle tracking by video microscopy8, respectively, reveal that 1-µm-sized colloidal particles bearing nuclear transport receptors9 called karyopherins can exhibit behaviour that varies from highly localized to unhindered two-dimensional diffusion. Particle diffusivity is controlled by varying the amount of free karyopherins in solution, which modulates the multivalency of Kap-binding sites within the molecular brush10. We conclude that the FG Nups resemble stimuli-responsive11 molecular ‘velcro’, which can impart ‘reduction of dimensionality’ as a means of biomimetic transport control in artificial environments.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Alberts, B., Johnson, A., Lewis, J., Raff, M., Roberts, K. & Walter, P. Molecular Biology of the Cell 4th edn (Garland, 2002).
Adam, G. & Delbrück, M. in Structural Chemistry and Molecular Biology (eds Rich, A. & Davidson, N.) 198–215 (W.H. Freeman, 1968).
Berg, O. G. & Vonhippel, P. H. Diffusion-controlled macromolecular interactions. Annu. Rev. Biophys. Biophys. Chem. 14, 131–160 (1985).
Santer, S. & Ruhe, J. Motion of nano-objects on polymer brushes. Polymer 45, 8279–8297 (2004).
Denning, D. P., Patel, S. S., Uversky, V., Fink, A. L. & Rexach, M. Disorder in the nuclear pore complex: the FG repeat regions of nucleoporins are natively unfolded. Proc. Natl Acad. Sci. USA 100, 2450–2455 (2003).
Grunwald, D., Singer, R. H. & Rout, M. Nuclear export dynamics of RNA–protein complexes. Nature 475, 333–341 (2011).
Jeney, S., Mor, F., Koszali, R., Forro, L. & Moy, V. T. Monitoring ligand–receptor interactions by photonic force microscopy. Nanotechnology 21, 255102 (2010).
Dettmer, S. L., Keyser, U. F. & Pagliara, S. Local characterization of hindered Brownian motion by using digital video microscopy and 3D particle tracking. Rev. Sci. Instrum. 85, 23708 (2014).
Chook, Y. M. & Sueel, K. E. Nuclear import by karyopherin-betas: recognition and inhibition. Biochim. Biophys. Acta 1813, 1593–1606 (2011).
Kapinos, L. E., Schoch, R. L., Wagner, R. S., Schleicher, K. D. & Lim, R. Y. H. Karyopherin-centric control of nuclear pores based on molecular occupancy and kinetic analysis of multivalent binding with FG-nucleoporins. Biophys. J. 106, 1751–1762 (2014).
Stuart, M. A. C. et al. Emerging applications of stimuli-responsive polymer materials. Nature Mater. 9, 101–113 (2010).
Whitesides, G. M. The origins and the future of microfluidics. Nature 442, 368–373 (2006).
Van den Heuvel, M. G. L. & Dekker, C. Motor proteins at work for nanotechnology. Science 317, 333–336 (2007).
Peters, R. Translocation through the nuclear pore complex: selectivity and speed by reduction-of-dimensionality. Traffic 6, 421–427 (2005).
Elbaum, M. Polymers in the pore. Science 314, 766–767 (2006).
Jovanovic-Talisman, T. et al. Artificial nanopores that mimic the transport selectivity of the nuclear pore complex. Nature 457, 1023–1027 (2009).
Kowalczyk, S. W. et al. Single-molecule transport across an individual biomimetic nuclear pore complex. Nature Nanotech. 6, 433–438 (2011).
Lim, R. Y. H. et al. Nanomechanical basis of selective gating by the nuclear pore complex. Science 318, 640–643 (2007).
Patel, S. S., Belmont, B. J., Sante, J. M. & Rexach, M. F. Natively unfolded nucleoporins gate protein diffusion across the nuclear pore complex. Cell 129, 83–96 (2007).
Mammen, M., Choi, S. K. & Whitesides, G. M. Polyvalent interactions in biological systems: implications for design and use of multivalent ligands and inhibitors. Angew. Chem. Int. Ed. 37, 2755–2794 (1998).
Isgro, T. A. & Schulten, K. Binding dynamics of isolated nucleoporin repeat regions to importin-beta. Structure 13, 1869–1879 (2005).
Paradise, A., Levin, M. K., Korza, G. & Carson, J. H. Significant proportions of nuclear transport proteins with reduced intracellular mobilities resolved by fluorescence correlation spectroscopy. J. Mol. Biol. 365, 50–65 (2007).
Happel, J. & Brenner, H. Low Reynolds Number Hydrodynamics (Martinus Nijhoff, 1983).
Perl, A. et al. Gradient-driven motion of multivalent ligand molecules along a surface functionalized with multiple receptors. Nature Chem. 3, 317–322 (2011).
Martinez-Veracoechea, F. J. & Frenkel, D. Designing super selectivity in multivalent nano-particle binding. Proc. Natl Acad. Sci. USA 108, 10963–10968 (2011).
Bormuth, V., Varga, V., Howard, J. & Schaffer, E. Protein friction limits diffusive and directed movements of kinesin motors on microtubules. Science 325, 870–873 (2009).
Yang, W. D. & Musser, S. M. Nuclear import time and transport efficiency depend on importin beta concentration. J. Cell Biol. 174, 951–961 (2006).
Tu, L. C., Fu, G., Zilman, A. & Musser, S. M. Large cargo transport by nuclear pores: implications for the spatial organization of FG-nucleoporins. EMBO J. 32, 3220–3230 (2013).
Jacobson, K., Ishihara, A. & Inman, R. Lateral diffusion of proteins in membranes. Annu. Rev. Physiol. 49, 163–175 (1987).
Eichmann, S. L., Meric, G., Swavola, J. C. & Bevan, M. A. Diffusing colloidal probes of protein–carbohydrate interactions. Langmuir 29, 2299–2310 (2013).
Uversky, V. N. Intrinsically disordered proteins from A to Z. Int. J. Biochem. Cell Biol. 43, 1090–1103 (2011).
Pierres, A., Benoliel, A. M. & Bongrand, P. Measuring the lifetime of bonds made between surface-linked molecules. J. Biol. Chem. 270, 26586–26592 (1995).
Srinivasan, N. & Kumar, S. Ordered and disordered proteins as nanomaterial building blocks. Nanomed. Nanobiotechnol. 4, 204–218 (2012).
Acknowledgements
The authors thank R.L. Schoch, M. Grimm and F.M. Mor for assistance and discussions. R.Y.H.L., K.D.S. and L.E.K. acknowledge support from the National Centre of Competence in Research ‘Molecular Systems Engineering’, the Swiss National Science Foundation, the Biozentrum and the Swiss Nanoscience Institute. S.J. acknowledges support from the Swiss National Science Foundation (SNF grant nos R'Equip 206021_121396 and 200021_143703). S.L.D. acknowledges the German Academic Exchange Service and the German National Academic Foundation. S.P. and U.F.K. acknowledge support from an ERC starting grant. S.P. also acknowledges support from the Leverhulme and Newton Trust through an Early Career Fellowship.
Author information
Authors and Affiliations
Contributions
K.D.S. conceived and carried out the experiments, wrote the manuscript, and analysed and interpreted the data. S.L.D. developed the tracking software and analysed the data. S.P. developed the tracking software and analysed the data. L.E.K. contributed proteins and interpreted data. U.F.K. analysed and interpreted data. S.J. designed and built the experimental set-up, conceived the experiment and the PFM data analysis protocol. R.Y.H.L. conceived the experiment, interpreted data and wrote the manuscript. All authors contributed to, discussed and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary Information (PDF 2142 kb)
Supplementary Movie 1
Supplementary Movie 1 (MOV 100 kb)
Supplementary Movie 2
Supplementary Movie 2 (MOV 102 kb)
Supplementary Movie 3
Supplementary Movie 3 (MOV 113 kb)
Rights and permissions
About this article
Cite this article
Schleicher, K., Dettmer, S., Kapinos, L. et al. Selective transport control on molecular velcro made from intrinsically disordered proteins. Nature Nanotech 9, 525–530 (2014). https://doi.org/10.1038/nnano.2014.103
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2014.103
This article is cited by
-
A designer FG-Nup that reconstitutes the selective transport barrier of the nuclear pore complex
Nature Communications (2021)
-
DNA origami scaffold for studying intrinsically disordered proteins of the nuclear pore complex
Nature Communications (2018)
-
Molecular velcro in Flatland
Nature Nanotechnology (2014)