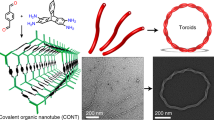
Abstract
Amphiphilic molecules—molecules that have both hydrophobic and hydrophilic properties—can self-assemble in water to form diverse structures such as micelles, vesicles and tubes1,2,3, and these nanostructures can be used for delivering drugs4,5, stabilizing membrane proteins6 or as nanoreactors7. We have previously shown that lipids can self-organize on the surface of single-walled carbon nanotubes into regular ring-shaped assemblies8. Here we show that these lipid assemblies can be polymerized and isolated from the nanotube template by application of an electric field. We also demonstrate that these assemblies are monodispersed, water-soluble, and can dissolve various hydrophobic rylene dyes, fullerenes and membrane proteins. The stability of these constructs and their diverse applications will be useful in the fields of cosmetics, medicine and material sciences.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Chen, D. & Jiang, M. Strategies for constructing polymeric micelles and hollow spheres in solution via specific intermolecular interactions. Acc. Chem. Res. 38, 494–502 (2005).
Shimizu, T., Masuda, M. & Minamikawa, H. Supramolecular nanotubes architectures based on amphiphilic molecules. Chem. Rev. 105, 1401–1443 (2005).
Zhou, Y. & Shimizu, T. Lipid nanotubes: A unique template to create diverse one-dimensional nanostructures. Chem. Mater. 20, 625–633 (2008).
Ferrari, M. Cancer nanotechnology: Opportunities and challenges. Nature Rev. Cancer 5, 161–171 (2005).
Duncan, R. The dawning era of polymer therapeutics. Nature Rev. Drug Discov. 2, 347–360 (2003).
Sanders, C. R., Hoffmann, A. K., Gray, D. N., Keyes, M. H. & Ellis, C. D. French Swimwear for membrane proteins. ChemBioChem 5, 423–426 (2004).
Dwars, T., Paetzold, E. & Oehme, G. Reactions in micellar systems. Angew. Chem. Int. Ed. 44, 7174–7199 (2005).
Richard, C., Balavoine, F., Schultz, P., Ebbesen, T. W. & Mioskowski, C. Supramolecular self-assembly of lipid derivatives on carbon nanotubes. Science 300, 775–778 (2003).
Ji, Q., Kamiya, S., Jung, J.-H. & Shimizu, T. Self-assembly of glycolipids on silica nanotube templates yielding hybrid nanotubes with concentric organic and inorganic layers. J. Mater. Chem. 15, 743–748 (2005).
Wang, Y., Angelatos, A. S. & Caruso, F. Template synthesis of nanostructured materials via layer-by-layer assembly. Chem. Mater. 20, 848–858 (2008).
Artyukhin, A. B. et al. Functional one-dimensional lipid bilayers on carbon nanotubes templates. J. Am. Chem. Soc. 127, 7538–7542 (2005).
Nish, A., Hwang, J.-Y., Doig, J. & Nicholas, R. J. Highly selective dispersion of single-walled carbon nanotubes using aromatic polymers. Nature Nanotech. 2, 640–646 (2007).
Zheng, M. et al. DNA-assisted dispersion and separation of carbon nanotubes. Nature Mater. 2, 338–342 (2003).
Balavoine, F. et al. Helical crystallization of proteins on carbon nanotubes: A first step towards the development of new biosensors. Angew. Chem. Int. Ed. 38, 1912–1915 (1999).
Qiao, R. & Ke, P. C. Lipid–carbon nanotube self-assembly in aqueous solution. J. Am. Chem. Soc. 128, 13656–13657 (2006).
Wu, Y. et al. Coating single-walled carbon nanotubes with phospholipids. J. Phys. Chem. B 110, 2475–2478 (2006).
Yurekli, K., Mitchell, C. A. & Krishnamoorti, R. Small-angle neutron scattering from surfactant-assisted aqueous dispersions of carbon nanotubes. J. Am. Chem. Soc. 126, 9902–9903 (2004).
Islam, M. F., Rojas, E., Bergey, D. M., Johnson, A. T. & Yodh, A. G. High weight fraction surfactant solubilization of single-wall carbon nanotubes in water. Nano Lett. 3, 269–273 (2003).
Ke, P. U. Fiddling the string of carbon nanotubes with amphipiles. Phys. Chem. Chem. Phys. 9, 439–447 (2007).
Matarredona, O. et al. Dispersion of single-walled carbon nanotubes in aqueous solutions of the anionic surfactant NaDDBS. J. Phys. Chem. B 107, 13357–13367 (2003).
O'Connell, M. J. et al. Reversible water-solubilization of single-walled carbon nanotubes by polymer wrapping. Chem. Phys. Lett. 342, 265–271 (2001).
Fabian, J. & Zahradnik, R. The search for highly coloured organic compounds. Angew. Chem. Int. Ed. 28, 677–694 (1989).
Lee, S. K. et al. Electrochemistry, spectroscopy and electrogenerated chemiluminescence of perylene, terrylene and quaterrylene diimides in aprotic solution. J. Am. Chem. Soc. 121, 3513–3520 (1999).
Liu, S., Lu, Y.-J., Kappes, M. M. & Ibers, J. A. The structure of the C60 molecule: X-ray crystal structure determination of a twin at 110 K. Science 254, 408–410 (1991).
Georgakilas, V. et al. Supramolecular self-assembled fullerene nanostructures. Proc. Natl Acad. Sci. USA 99, 5075–5080 (2002).
Sanders, C. R. & Scott Prosser, R. Bicelles: A model membrane system for all seasons? Structure 6, 1227–1234 (1998).
Schafmeister, C. E., Miercke, L. J. & Stroud, R. M. Structure at 2.5 Å of a designed peptide that maintains solubility of membrane proteins. Science 262, 734–738 (1993).
McGregor, C.-L. et al. Lipopeptide detergents designed for the structural study of membrane proteins. Nature Biotechnol. 21, 171–176 (2003).
Popot, J.-L. et al. Amphipol: Polymeric surfactants for membrane biology research. Cell. Mol. Life Sci. 60, 1559–1574 (2003).
Sanders, C. R. & Myers, J. K. Disease-related misassembly of membrane proteins. Annu. Rev. Biophys. Biomol. Struct. 33, 25–51 (2004).
Braun, V. & Mahren, S. Transmembrane transcriptional control (surface signalling) of the Escherichia coli Fec type. FEMS Microbiol. Rev. 29, 673–684 (2005).
Schalk, I. J. et al. Iron-free pyoverdin binds to its outer membrane receptor FpvA in Pseudomonas aeruginosa: A new mechanism for membrane iron transport. Mol. Microbiol. 39, 351–361 (2001).
Rigaud, J.-L. et al. Bio-beads: An efficient strategy for two-dimensional crystallization of membrane proteins. J. Struct. Biol. 118, 226–235 (1997).
Author information
Authors and Affiliations
Contributions
P.S. performed the TEM experiments. H.C. performed the protein solubilization experiments. S.M. and C.M. conceived and designed the experiments.
Corresponding author
Ethics declarations
Competing interests
S. R., P. S. and C. M. have filed a patent application on the process described in this work.
Supplementary information
Supplementary Information
Supplementary Information (PDF 582 kb)
Rights and permissions
About this article
Cite this article
Thauvin, C., Rickling, S., Schultz, P. et al. Carbon nanotubes as templates for polymerized lipid assemblies. Nature Nanotech 3, 743–748 (2008). https://doi.org/10.1038/nnano.2008.318
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2008.318
This article is cited by
-
Influences and mechanisms of nanofullerene on the horizontal transfer of plasmid-encoded antibiotic resistance genes between E. coli strains
Frontiers of Environmental Science & Engineering (2020)
-
Nanotubes-/nanowires-based, microfluidic-integrated transistors for detecting biomolecules
Microfluidics and Nanofluidics (2010)