Abstract
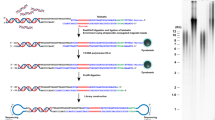
Expression of telomerase, the specialized reverse transcriptase that adds 5′-TTAGGG-3′ repeats to the ends of human chromosomes, is upregulated in ≥85% of human cancers and tumor cell lines. We describe a direct primer-extension activity assay1,2 for human telomerase that displays sensitivity to ∼106 telomerase-positive cells, making the method suitable for use with standard cell culture–based research (Fig. 1). Telomerase is first immunoaffinity purified from cell lysate using an antibody to telomerase and captured using protein G–agarose beads (Steps 14–17). Then telomerase is dissociated from the beads using excess peptide antigen (Step 19). A second affinity purification exploits the stable binding interaction between human telomerase and the telomeric DNA substrate 5′-(TTAGGG)3-3′ (dissociation half-life ≥ 10 h at 23 °C)3. Modifying neutravidin beads with 5′-biotin-CTAGACCTGTCATCA(TTAGGG)3-3′ (Step 5) generates an affinity reagent that captures >90% of immunopurified telomerase4 (Step 22), providing highly enriched telomerase bound to its DNA substrate in a volume of 20 μl of beads. Addition of assay buffer results in extension of the bead-immobilized DNA substrate (Step 24). Telomerase extension products are released by heating in denaturing formamide buffer to disrupt the avidin-biotin interaction, and then separated and visualized by standard techniques.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Morin, G.B. The human telomere terminal transferase enzyme is a ribonucleoprotein that synthesizes TTAGGG repeats. Cell 59, 521–529 (1989).
Sun, D. et al. Regulation of catalytic activity and processivity of human telomerase. Biochemistry 38, 4037–4044 (1999).
Wallweber, G., Gryzanov, S., Pongracz, K. & Pruzan, R. Interaction of human telomerase with its primer substrate. Biochemistry 42, 589–600 (2003).
Cohen, S.B. et al. Protein composition of catalytically active human telomerase from immortal cells. Science 315, 1850–1853 (2007).
Kim, N.W. et al. Specific association of human telomerase activity with immortal cells and cancer. Science 266, 2011–2015 (1994).
Shay, J.W. & Bacchetti, S. A survey of telomerase activity in human cancer. Eur. J. Cancer 33, 787–791 (1997).
Wright, W.E., Shay, J.W. & Piatyszek, M.A. Modifications of a telomeric repeat amplification protocol (TRAP) result in increased reliability, linearity and sensitivity. Nucleic Acids Res. 23, 3794–3795 (1995).
Acknowledgements
We thank members of the laboratory of T. Bryan for assistance with telomerase methods. This research was supported by the Carcinogenesis Fellowship of the Cancer Council New South Wales and the National Health and Medical Research Council of Australia.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
A provisional patent application describing the epitope sequence of the hTERT antibody has been filed (S.B.C.; New South Wales, Australia).
Supplementary information
Supplementary Text and Figures
Supplementary Figure 1 and Supplementary Methods (PDF 910 kb)
Rights and permissions
About this article
Cite this article
Cohen, S., Reddel, R. A sensitive direct human telomerase activity assay. Nat Methods 5, 355–360 (2008). https://doi.org/10.1038/nmeth.f.209
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.f.209
This article is cited by
-
CDK1 dependent phosphorylation of hTERT contributes to cancer progression
Nature Communications (2020)
-
Modular ssDNA binding and inhibition of telomerase activity by designer PPR proteins
Nature Communications (2018)
-
Inhibition of telomerase RNA decay rescues telomerase deficiency caused by dyskerin or PARN defects
Nature Structural & Molecular Biology (2016)
-
A novel two-step genome editing strategy with CRISPR-Cas9 provides new insights into telomerase action and TERT gene expression
Genome Biology (2015)
-
Telomeric G-quadruplexes are a substrate and site of localization for human telomerase
Nature Communications (2015)